Advanced Selective Nitration Technology for Scalable 5-Nitrobenzofuran Intermediate Manufacturing
Advanced Selective Nitration Technology for Scalable 5-Nitrobenzofuran Intermediate Manufacturing
The pharmaceutical and agrochemical industries continuously demand high-purity heterocyclic building blocks, particularly benzofuran and benzothiophene derivatives nitrated at specific positions. Patent CN1379754A introduces a groundbreaking methodology for synthesizing these valuable intermediates, specifically targeting the challenging 5-nitro substitution pattern. This technology pivots away from traditional, waste-intensive pathways, offering a streamlined approach that begins with the preparation of novel nitroaromatic compounds of general formula (I'). 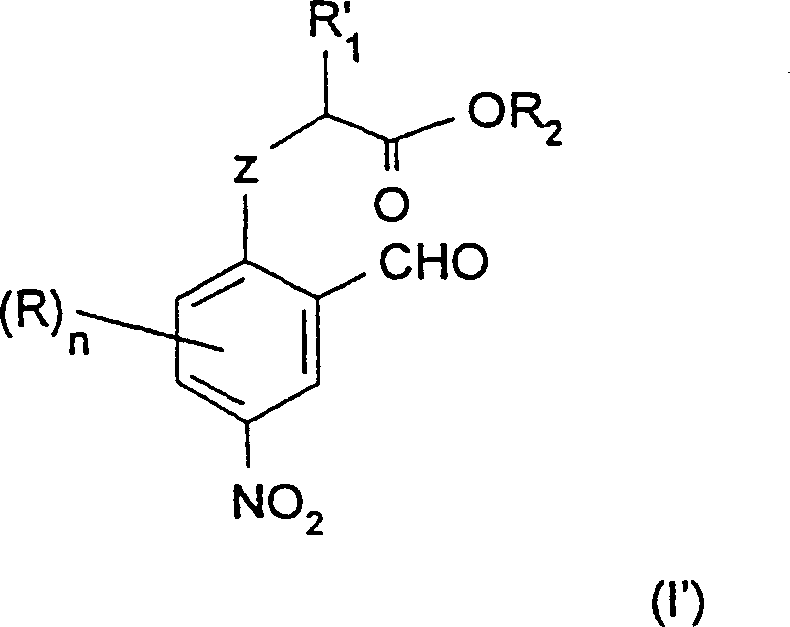 . The core innovation lies in a highly selective nitration strategy performed on O- or S-alkylated precursors, which ensures the nitro group is installed precisely at the 4-position relative to the oxygen or sulfur atom. This precision is paramount for downstream cyclization into 2-alkyl-5-nitrobenzofurans, a structural motif prevalent in numerous bioactive molecules. For R&D directors seeking robust synthetic routes, this patent provides a detailed blueprint for achieving superior regioselectivity without the need for complex protecting group manipulations typically associated with phenolic substrates.
. The core innovation lies in a highly selective nitration strategy performed on O- or S-alkylated precursors, which ensures the nitro group is installed precisely at the 4-position relative to the oxygen or sulfur atom. This precision is paramount for downstream cyclization into 2-alkyl-5-nitrobenzofurans, a structural motif prevalent in numerous bioactive molecules. For R&D directors seeking robust synthetic routes, this patent provides a detailed blueprint for achieving superior regioselectivity without the need for complex protecting group manipulations typically associated with phenolic substrates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-nitrobenzofuran derivatives has relied heavily on methodologies described in earlier literature, such as European patent EP-A-0471609. These conventional routes often employ Wittig-type reactions involving phosphonium salts, specifically reacting 2-hydroxy-5-nitro-benzyltriphenylphosphonium bromide with acyl chlorides. While chemically valid, this approach presents significant logistical and economic hurdles for large-scale manufacturing. The preparation of the phosphonium salt itself requires stoichiometric amounts of triphenylphosphine and benzyl bromides, generating substantial phosphorus-containing waste that complicates environmental compliance and waste treatment protocols. Furthermore, the use of pyridine as a base and solvent adds to the toxicity profile and cost of goods. From a supply chain perspective, the reliance on specialized phosphorous reagents introduces volatility in raw material pricing and availability. Additionally, the purification of the final product from triphenylphosphine oxide byproducts can be tedious, often requiring extensive chromatography or recrystallization steps that reduce overall yield and throughput efficiency in a commercial plant setting.
The Novel Approach
In stark contrast, the methodology disclosed in CN1379754A offers a completely different synthetic logic that bypasses the need for phosphonium chemistry entirely. The novel approach centers on the strategic protection of the phenolic hydroxyl group prior to nitration. By converting the starting salicylaldehyde derivative into an O-alkylated ether (Formula II), the electronic properties of the aromatic ring are modulated to favor para-substitution during the subsequent nitration step. 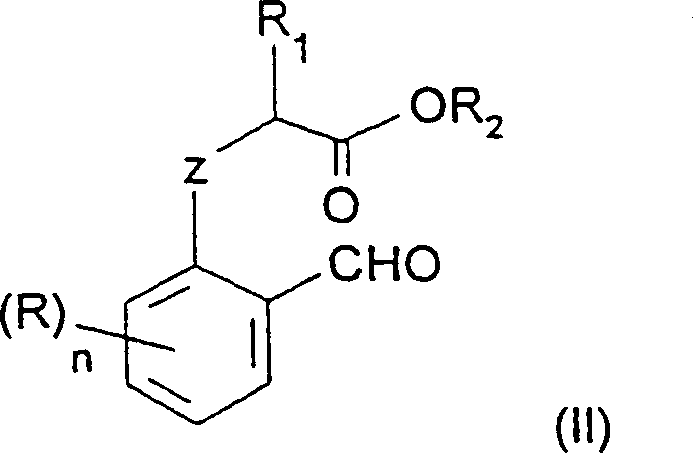 . This transformation is achieved using a standard nitrating mixture of nitric acid and sulfuric acid, reagents that are ubiquitous and cost-effective in the fine chemical industry. The key discovery is that selective nitration at the 4-position is only feasible when the starting material is an O- or S-alkylated compound; free phenols fail to provide this selectivity. This insight allows manufacturers to utilize commodity chemicals like salicylaldehyde and alpha-halo esters, drastically simplifying the supply chain. The resulting nitroaromatic intermediate can then be seamlessly converted into the target heterocycle through saponification and cyclization, offering a cleaner, more direct path to high-value API intermediates with significantly reduced impurity profiles.
. This transformation is achieved using a standard nitrating mixture of nitric acid and sulfuric acid, reagents that are ubiquitous and cost-effective in the fine chemical industry. The key discovery is that selective nitration at the 4-position is only feasible when the starting material is an O- or S-alkylated compound; free phenols fail to provide this selectivity. This insight allows manufacturers to utilize commodity chemicals like salicylaldehyde and alpha-halo esters, drastically simplifying the supply chain. The resulting nitroaromatic intermediate can then be seamlessly converted into the target heterocycle through saponification and cyclization, offering a cleaner, more direct path to high-value API intermediates with significantly reduced impurity profiles.
Mechanistic Insights into Selective Electrophilic Aromatic Substitution
The success of this synthetic route hinges on a deep understanding of electrophilic aromatic substitution mechanisms, specifically how substituent effects dictate regioselectivity. In the precursor molecule (Formula II), the alkoxy group (-OR) acts as a strong activating group and an ortho/para director. However, the steric hindrance at the ortho positions, particularly adjacent to the bulky aldehyde and alkyl ester side chains, kinetically favors attack at the para position relative to the oxygen atom. 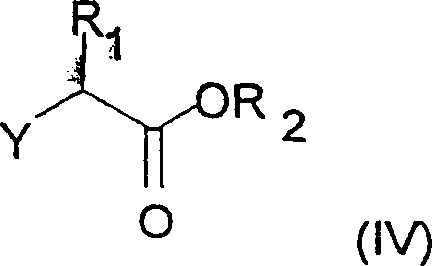 . When the nitronium ion (NO2+) is generated in situ from the sulfuric-nitric acid mixture, it attacks the electron-rich aromatic ring. Because the hydroxyl group has been masked as an ether, the strong hydrogen bonding interactions that often complicate the nitration of free phenols are eliminated. This prevents the formation of unwanted ortho-nitro isomers or oxidation byproducts that typically plague phenolic nitrations. The patent specifies that the reaction proceeds optimally at temperatures between -10°C and 20°C, a range that balances reaction rate with the need to suppress poly-nitration or decomposition of the sensitive aldehyde functionality. This mechanistic control ensures that the nitro group ends up exactly where it is needed for the subsequent ring-closing step, effectively pre-installing the 5-nitro motif of the final benzofuran system.
. When the nitronium ion (NO2+) is generated in situ from the sulfuric-nitric acid mixture, it attacks the electron-rich aromatic ring. Because the hydroxyl group has been masked as an ether, the strong hydrogen bonding interactions that often complicate the nitration of free phenols are eliminated. This prevents the formation of unwanted ortho-nitro isomers or oxidation byproducts that typically plague phenolic nitrations. The patent specifies that the reaction proceeds optimally at temperatures between -10°C and 20°C, a range that balances reaction rate with the need to suppress poly-nitration or decomposition of the sensitive aldehyde functionality. This mechanistic control ensures that the nitro group ends up exactly where it is needed for the subsequent ring-closing step, effectively pre-installing the 5-nitro motif of the final benzofuran system.
Furthermore, the impurity control mechanism is inherently built into the choice of starting materials and reaction conditions. The use of alpha-halo esters (Formula IV) with specific leaving groups like bromine or chlorine allows for clean SN2 alkylation of the phenoxide anion generated from salicylaldehyde (Formula III). The patent highlights that using bases like potassium carbonate or sodium carbonate in polar aprotic solvents such as DMF or DMAC facilitates this coupling with high conversion. Any unreacted starting material or side products from the alkylation step are generally easier to separate from the ether intermediate than the complex mixtures arising from Wittig reactions. During the nitration phase, the strict control of acid concentration (50% to 98% sulfuric acid) and the molar ratio of nitrating agent (1.0 to 1.2 equivalents) minimizes the formation of dinitro species. This precision in stoichiometry and condition control translates directly to a cleaner crude product, reducing the burden on downstream purification units and enhancing the overall mass balance of the process, which is a critical metric for process chemists evaluating technology transfer feasibility.
How to Synthesize 5-Nitrobenzofuran Intermediates Efficiently
The synthesis of these high-value intermediates follows a logical three-stage sequence that is amenable to batch processing in standard multipurpose chemical reactors. The process begins with the O-alkylation of a 2-hydroxybenzaldehyde derivative with an alpha-halo carboxylic acid ester, establishing the carbon backbone and protecting the phenol. Following isolation, the ether intermediate undergoes the critical selective nitration step using mixed acids to install the nitro group at the 4-position. Finally, the ester is hydrolyzed, and the resulting acid undergoes intramolecular cyclization in the presence of a carboxylic anhydride to form the benzofuran ring. This streamlined workflow eliminates the need for cryogenic conditions beyond standard cooling and avoids the use of moisture-sensitive organometallic reagents, making it highly suitable for facilities equipped with standard glass-lined or stainless steel reactors.
- Perform O-alkylation of 2-hydroxybenzaldehyde with an alpha-halo ester using a base like potassium carbonate in a polar solvent such as DMF.
- Conduct selective nitration of the resulting ether using a nitric acid and sulfuric acid mixture at low temperatures (-10°C to 20°C) to target the 4-position.
- Execute saponification of the ester group followed by cyclization using a carboxylic anhydride like acetic anhydride to close the furan ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology represents a significant opportunity to optimize the cost structure and reliability of the supply chain for benzofuran-based active ingredients. The shift away from phosphonium-based chemistry removes the dependency on triphenylphosphine, a reagent subject to price fluctuations and supply constraints due to its widespread use in various organic syntheses. Instead, the process relies on salicylaldehyde and alpha-halo esters, which are commodity chemicals produced at massive scales globally, ensuring a stable and competitive pricing environment. The elimination of phosphorus waste also translates to substantial cost reduction in manufacturing by lowering wastewater treatment fees and simplifying regulatory compliance regarding hazardous waste disposal. Moreover, the high selectivity of the nitration step reduces the formation of isomeric impurities, which means less material is lost during purification crystallizations, thereby improving the effective yield per ton of raw material input.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the replacement of expensive, stoichiometric reagents with catalytic or inexpensive bulk chemicals. By avoiding the Wittig reaction, the process eliminates the generation of triphenylphosphine oxide, a solid waste product that is difficult and costly to dispose of. The use of simple mineral acids for nitration and common carbonates for alkylation further drives down the raw material bill. Additionally, the ability to perform the alkylation in aqueous media using phase transfer catalysts offers an alternative green chemistry pathway that reduces solvent consumption and recovery costs, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Diversifying the synthetic route mitigates risks associated with single-source suppliers of specialized reagents. Since the key building blocks like salicylaldehyde and nitric acid are available from multiple global vendors, the risk of supply disruption is minimized. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic failure, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturing, preventing costly downtime or delays in drug product launches due to intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-gram to kilogram examples within the patent literature without requiring exotic equipment. The exothermic nature of the nitration is manageable with standard jacketed reactors and controlled dosing strategies. From an environmental standpoint, the reduction in heavy metal or phosphorus load in the effluent simplifies the permitting process for new manufacturing lines. The potential to recycle solvents like dichloromethane or DMF, and the use of water-soluble salts that are easily separated, aligns with modern green chemistry principles, enhancing the sustainability profile of the supply chain and meeting the increasingly stringent ESG criteria of major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective nitration technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the process viability. Understanding these details helps stakeholders assess the fit of this technology within their existing manufacturing capabilities and quality systems. Engaging with these technical specifics early in the evaluation process can accelerate the timeline for technology transfer and commercial adoption.
Q: Why is selective nitration critical for 5-nitrobenzofuran synthesis?
A: Direct nitration of free phenols often leads to mixtures of ortho and para products. By protecting the hydroxyl group via alkylation first, the nitro group is directed exclusively to the para position relative to the oxygen, which corresponds to the 5-position in the final benzofuran ring, ensuring high regioselectivity and purity.
Q: What are the advantages of this route over the traditional Wittig reaction?
A: Traditional methods often rely on phosphonium salts and Wittig olefination, which generate significant phosphorus waste and require expensive reagents. This new pathway utilizes simple alkylation and electrophilic aromatic substitution, utilizing cheaper starting materials like salicylaldehyde and alpha-halo esters, significantly reducing raw material costs and waste treatment burdens.
Q: Can this process be scaled for industrial production?
A: Yes, the process is designed for scalability. The nitration step uses standard mixed acid conditions manageable in stainless steel reactors with temperature control, and the alkylation can be performed in common polar solvents or even under phase transfer catalysis in aqueous media, facilitating large-scale manufacturing without exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitrobenzofuran Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our team of expert process chemists has extensively analyzed the methodology described in CN1379754A and possesses the technical capability to implement this selective nitration route at commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless and risk-mitigated. Our facility is equipped with state-of-the-art corrosion-resistant reactors capable of handling mixed acid nitrations safely, along with rigorous QC labs that enforce stringent purity specifications to meet the demanding requirements of global regulatory agencies.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced chemistry for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data from our pilot batches and comprehensive route feasibility assessments. Let us demonstrate how our mastery of complex heterocyclic synthesis can drive value, reduce lead time for high-purity pharmaceutical intermediates, and secure your supply chain against future market volatilities.
