Advanced Synthesis of 5-Nitrobenzofuran Derivatives via Selective Nitration Technology
Advanced Synthesis of 5-Nitrobenzofuran Derivatives via Selective Nitration Technology
The pharmaceutical and agrochemical industries continuously demand high-purity heterocyclic building blocks, particularly benzofuran and benzothiophene derivatives nitrated at specific positions. Patent CN1268601C introduces a groundbreaking methodology for producing these complex structures through novel nitroaromatic intermediates. This technology addresses the longstanding challenge of regioselective nitration on activated aromatic rings, offering a robust pathway to 2-alkyl-5-nitrobenzofurans. By utilizing a unique sequence of O-alkylation followed by controlled nitration, manufacturers can achieve superior purity profiles compared to legacy Wittig-based syntheses. The core innovation lies in the preparation of intermediates of general formula (I'), which serve as versatile precursors for downstream cyclization.
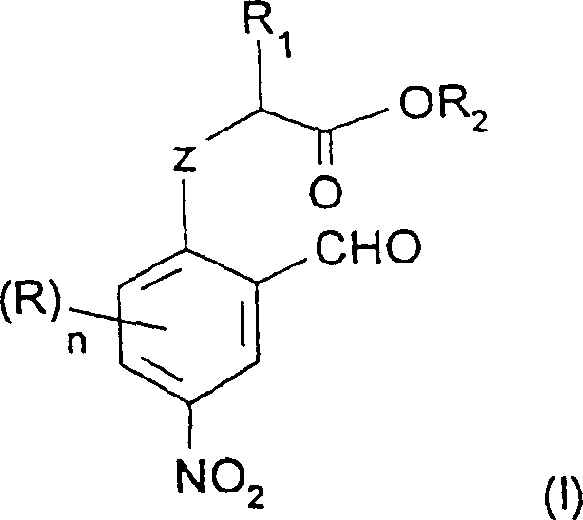
For procurement specialists and supply chain directors, this patent represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. The process relies on readily available starting materials such as salicylaldehyde and alpha-halo esters, avoiding the expensive phosphonium salts required in older European patents like EP-A-0471609. Furthermore, the ability to perform key alkylation steps in aqueous media using phase transfer catalysis aligns with modern green chemistry initiatives, potentially lowering waste disposal costs. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic advantages is crucial for securing long-term supply continuity for active pharmaceutical ingredients (APIs) targeting various therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-nitrobenzofuran derivatives has relied heavily on Wittig olefination strategies, as exemplified by earlier art such as EP-A-0471609. These conventional routes typically involve the reaction of 2-hydroxy-5-nitro-benzyltriphenylphosphonium bromide with acyl chlorides. While chemically feasible, this approach suffers from significant economic and operational drawbacks. The requirement for triphenylphosphine generates substantial amounts of triphenylphosphine oxide waste, which is notoriously difficult and costly to remove from the final product stream. Additionally, the preparation of the phosphonium salt itself involves multiple steps and hazardous alkylating agents like benzyl bromide. From a supply chain perspective, the reliance on stoichiometric amounts of expensive phosphorus reagents creates volatility in production costs and complicates the purification process, often necessitating extensive chromatography or recrystallization steps that reduce overall yield.
The Novel Approach
In stark contrast, the methodology disclosed in CN1268601C utilizes a completely different synthetic logic that bypasses the need for phosphorus chemistry entirely. The novel approach begins with the O-alkylation of a 2-hydroxybenzaldehyde derivative, followed by a highly selective nitration at the 4-position relative to the oxygen atom. This sequence ensures that the nitro group is installed precisely where needed for the subsequent formation of the 5-nitrobenzofuran core. The process leverages the directing effects of the alkoxy group to achieve regioselectivity that is unattainable with free phenols. By shifting the nitration step to occur on the O-alkylated intermediate rather than the final heterocycle or a phosphonium salt, the process minimizes side reactions and simplifies the isolation of the desired isomer. This strategic rearrangement of synthetic steps results in a cleaner reaction profile and facilitates easier scale-up for commercial production of complex pharmaceutical intermediates.
Mechanistic Insights into Selective Para-Nitration
The heart of this technological advancement lies in the precise control of electrophilic aromatic substitution. When a phenolic substrate contains a free hydroxyl group, nitration often leads to a mixture of ortho and para products, or even over-nitration, due to the strong activating nature of the OH group. However, the patent demonstrates that converting the phenol into an ether (O-alkylation) modulates this reactivity. The alkoxy group remains an ortho/para director, but steric hindrance at the ortho position (adjacent to the aldehyde and the alkoxy chain) combined with the specific reaction conditions favors attack at the para position. The use of a nitronium ion (NO2+) source generated in situ from nitric acid and sulfuric acid provides the necessary electrophilic strength while maintaining control.
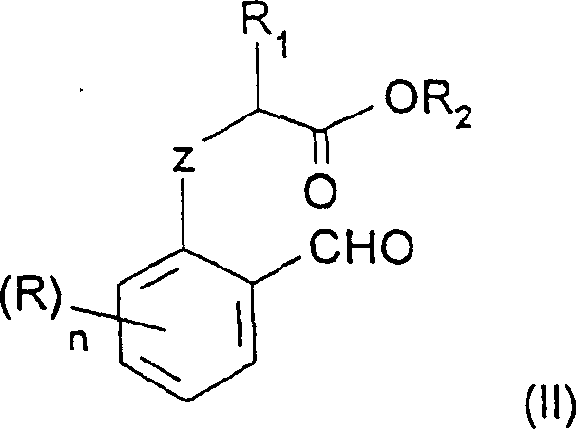
Crucially, the reaction conditions specified—temperatures ranging from -10°C to 20°C, preferably -5°C to 10°C—are vital for maintaining this selectivity. At higher temperatures, the kinetic control is lost, leading to potential isomerization or decomposition. The presence of concentrated sulfuric acid serves not only to generate the nitronium ion but also to protonate the aldehyde carbonyl, slightly deactivating the ring against over-reaction. This delicate balance allows for the isolation of the 4-nitro isomer in high purity, as evidenced by experimental data showing titration concentrations exceeding 96%. For R&D directors, this implies that impurity profiles can be tightly managed without resorting to complex downstream purification, ensuring that the high-purity pharmaceutical intermediates meet stringent regulatory specifications for downstream API synthesis.
How to Synthesize 5-Nitrobenzofuran Intermediates Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and robustness, making it ideal for technology transfer from laboratory to pilot plant. The process is generally divided into three distinct operational phases: the initial alkylation to protect the phenol and introduce the side chain, the critical nitration step to install the nitro functionality, and the final cyclization to close the furan ring. Each step utilizes common industrial reagents and standard unit operations such as filtration, extraction, and distillation. The flexibility of the method allows for variations in the alkyl chain length (R1) and the heteroatom (Z = O or S), providing a platform technology for generating diverse libraries of benzofuran and benzothiophene derivatives. Detailed standardized synthesis steps follow below to guide process engineers in replicating these results.
- Perform O-alkylation of 2-hydroxybenzaldehyde with an alpha-halo ester using a base and optionally a phase transfer catalyst in aqueous or organic media.
- Conduct selective nitration of the O-alkylated intermediate at the 4-position using a nitronium ion source in sulfuric acid at low temperatures (-10°C to 20°C).
- Execute saponification of the ester group followed by cyclization using a carboxylic acid anhydride to yield the final 5-nitrobenzofuran derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits beyond mere chemical elegance. The primary advantage stems from the elimination of expensive and waste-generating phosphorus reagents. By replacing the Wittig pathway with an alkylation-nitration-cyclization sequence, the material cost of goods sold (COGS) is significantly reduced. The starting materials, such as salicylaldehyde and alpha-bromo esters, are commodity chemicals available from multiple global suppliers, reducing supply risk and enhancing negotiation leverage. Furthermore, the ability to perform the alkylation in aqueous media using phase transfer catalysts drastically reduces the volume of organic solvents required, leading to lower solvent recovery costs and a smaller environmental footprint. This aligns with increasingly strict environmental regulations, ensuring long-term operational compliance.
- Cost Reduction in Manufacturing: The removal of triphenylphosphine and its associated waste streams eliminates a major cost center in the production of nitro-aromatics. The new process relies on catalytic amounts of iodide salts or quaternary ammonium salts which can potentially be recycled, further driving down variable costs. Additionally, the high yields reported in the patent examples, such as over 90% for the nitration step and nearly 99% for the alkylation, mean that less raw material is wasted per kilogram of finished product. This efficiency translates directly to improved margins and more competitive pricing for the final high-purity OLED material or API intermediate.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents like benzyltriphenylphosphonium bromide creates single points of failure in the supply chain. In contrast, the reagents for this novel process—sulfuric acid, nitric acid, acetic anhydride, and simple aldehydes—are produced at massive scales globally. This ubiquity ensures that production schedules are not disrupted by shortages of niche chemicals. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic loss of selectivity, further enhances reliability. This stability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing for just-in-time delivery models.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids hazardous exotherms that are difficult to manage in large reactors. The nitration is conducted at low temperatures with controlled addition rates, a standard practice in fine chemical manufacturing. Moreover, the shift towards aqueous alkylation reduces the generation of halogenated organic waste, simplifying wastewater treatment. The final cyclization step uses acetic anhydride, which produces acetic acid as a byproduct—a substance that is easily recoverable and reusable. These factors collectively support the commercial scale-up of complex polymer additives or drug substances while meeting rigorous environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitration technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on process feasibility and product quality. Understanding these details is essential for technical teams evaluating the integration of this route into existing manufacturing lines.
Q: How does this nitration method improve regioselectivity compared to traditional methods?
A: The patent describes that O- or S-alkylation of the phenolic substrate prior to nitration blocks the ortho position and strongly directs the nitronium electrophile to the para position (4-position), achieving high selectivity that is difficult with free phenols.
Q: What are the key advantages of the alkylation step described in CN1268601C?
A: The process allows for O-alkylation in aqueous media using phase transfer catalysts, eliminating the need for large volumes of organic solvents and enabling easier workup and potential catalyst recycling, which significantly reduces environmental impact.
Q: Can this process be scaled for commercial production of API intermediates?
A: Yes, the reaction conditions utilize common industrial reagents like sulfuric acid, nitric acid, and acetic anhydride. The exothermic nitration is controlled at low temperatures, and the cyclization uses standard heating, making it highly adaptable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitrobenzofuran Supplier
The technological potential of CN1268601C is immense, offering a clearer path to high-value heterocyclic compounds used in modern medicine and materials science. NINGBO INNO PHARMCHEM stands ready to leverage this chemistry as your trusted partner. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the infrastructure to translate this patent into reality. Our facilities are equipped with corrosion-resistant reactors suitable for nitration chemistry and advanced purification systems to ensure stringent purity specifications. Our rigorous QC labs employ state-of-the-art HPLC and NMR analysis to guarantee that every batch of 5-nitrobenzofuran intermediate meets the exacting standards required by global regulatory bodies.
We invite you to discuss how this optimized synthetic route can benefit your specific project requirements. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis comparing this method against your current supply chain. Contact us today to request specific COA data and route feasibility assessments. By collaborating with NINGBO INNO PHARMCHEM, you secure not just a chemical supplier, but a strategic partner dedicated to innovation, quality, and supply chain resilience in the competitive landscape of fine chemical intermediates.
