Advanced Manufacturing Of Trans-Polyhydroxy Diphenyl Ethylene For Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for bioactive polyphenols, specifically trans-polyhydroxy diphenyl ethylene derivatives such as trans-resveratrol and piceatannol. Patent CN101481300B introduces a groundbreaking preparation method that addresses the critical bottlenecks of traditional synthesis, offering a pathway that is both economically viable and technically superior for large-scale manufacturing. This innovation leverages a modified Perkin reaction followed by a specific decarboxylation and isomerization sequence to construct the trans-stilbene skeleton with exceptional selectivity. The significance of this technology lies in its ability to bypass the cumbersome hydroxyl protection and deprotection steps that have historically plagued the production of these sensitive molecules. By utilizing readily available hydroxy-substituted benzaldehydes and phenylacetic acids, the process establishes a direct line to high-value intermediates used in nutraceuticals and pharmaceutical formulations. 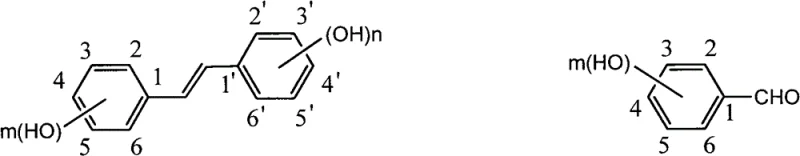
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyhydroxy stilbenes has relied heavily on methods such as the Wittig reaction, Wittig-Horner reaction, or Heck coupling, all of which present substantial drawbacks for industrial application. These conventional pathways often demand strictly anhydrous conditions and cryogenic operations, which escalate energy consumption and require specialized equipment that increases capital expenditure. Furthermore, the presence of multiple hydroxyl groups on the aromatic rings typically necessitates complex protection and deprotection strategies to prevent side reactions, adding multiple synthetic steps that erode overall yield and atom economy. The use of expensive transition metal catalysts in Heck reactions or phosphorus reagents in Wittig protocols also introduces significant cost burdens and environmental concerns regarding waste disposal. Consequently, these traditional methods struggle to meet the rigorous cost and scalability requirements of modern commercial supply chains, often resulting in inconsistent batch quality and prolonged lead times for procurement teams.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a streamlined two-step sequence that fundamentally simplifies the manufacturing landscape for these complex intermediates. By employing a Perkin condensation between hydroxy-substituted benzaldehydes and phenylacetic acids in the presence of acetic anhydride and triethylamine, the method constructs the carbon skeleton under relatively mild thermal conditions ranging from 90°C to 150°C. This eliminates the need for extreme temperatures or pressures, thereby reducing operational risks and energy costs significantly. The subsequent decarboxylation and isomerization step, catalyzed by metallic copper in an organic base solvent, drives the formation of the thermodynamically stable trans-isomer with high selectivity. This direct route avoids the use of protecting groups entirely, which not only shortens the synthetic timeline but also minimizes the generation of chemical waste. The result is a process that is inherently safer, more cost-effective, and far more amenable to scale-up than any preceding technology in this domain.
Mechanistic Insights into Perkin Condensation and Copper-Catalyzed Isomerization
The core of this synthetic strategy relies on the precise orchestration of the Perkin reaction to form the initial cis-diphenyl ethylene skeleton without compromising the integrity of the phenolic hydroxyl groups. In this mechanism, the acetic anhydride acts as both a solvent and a dehydrating agent, facilitating the condensation between the aldehyde and the activated methylene group of the phenylacetic acid derivative. The triethylamine catalyst promotes the formation of the enolate intermediate, which attacks the carbonyl carbon of the aldehyde to form the beta-hydroxy acid intermediate, followed by dehydration to yield the acrylic acid derivative. 
Following the skeleton construction, the transformation from the cis-acrylic acid derivative to the trans-polyhydroxy diphenyl ethylene is achieved through a copper-catalyzed decarboxylation and isomerization process. Under elevated temperatures between 180°C and 220°C in solvents like quinoline or DBU, the carboxyl group is eliminated as carbon dioxide, while the double bond undergoes isomerization to the more stable trans-configuration. The metallic copper plays a pivotal role in facilitating electron transfer and stabilizing the transition state during decarboxylation, ensuring high conversion rates. This mechanistic pathway is particularly advantageous because it leverages thermodynamic control to ensure high trans-selectivity, thereby reducing the burden on downstream purification processes. 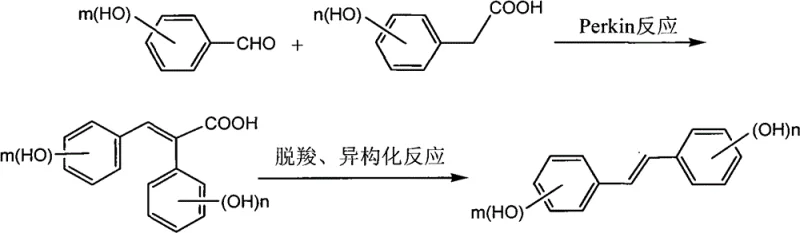
How to Synthesize Trans-Polyhydroxy Diphenyl Ethylene Efficiently
The operational execution of this synthesis is designed for maximum efficiency, beginning with the precise mixing of hydroxy-substituted benzaldehyde and phenylacetic acid in a molar ratio optimized for high conversion. The reaction mixture is heated in acetic anhydride with triethylamine, where temperature control is critical to balance reaction rate and byproduct formation. After the initial condensation, the intermediate acrylic acid derivative is isolated and subjected to the second step involving metallic copper and an organic base solvent at high temperatures. The final product is recovered through simple acidification and recrystallization, avoiding the need for complex chromatographic separation.
- Conduct Perkin reaction between hydroxy-substituted benzaldehyde and phenylacetic acid using acetic anhydride and triethylamine at 90-150°C to form the cis-skeleton.
- Perform decarboxylation and isomerization on the acrylic acid derivative using metal Cu and organic base solvent at 180-220°C.
- Purify the final trans-polyhydroxy diphenyl ethylene product through recrystallization using alcohol-water or ethyl acetate-petroleum ether mixtures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic advantages that directly impact the bottom line and operational resilience. The elimination of protection and deprotection steps significantly reduces the number of unit operations required, which in turn lowers labor costs and decreases the consumption of auxiliary chemicals and solvents. The use of commodity chemicals such as hydroxy-substituted benzaldehydes and phenylacetic acids ensures a stable and reliable supply of raw materials, mitigating the risk of shortages that often plague specialized reagent markets. Furthermore, the simplicity of the purification process, which relies on recrystallization rather than column chromatography, allows for much larger batch sizes to be processed without a proportional increase in processing time or equipment footprint.
- Cost Reduction in Manufacturing: The removal of hydroxyl protection groups eliminates the need for expensive silylating or acylating agents and the subsequent reagents required to remove them, leading to substantial cost savings in raw material procurement. Additionally, the high atom economy of the Perkin reaction ensures that a greater proportion of the starting mass is converted into the final product, reducing waste disposal costs and improving overall process efficiency. The ability to operate at moderate temperatures in the first step also reduces energy consumption compared to cryogenic methods, further driving down utility expenses in large-scale production facilities.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials like substituted benzaldehydes and phenylacetic acids, manufacturers can diversify their supplier base and avoid single-source dependencies on exotic catalysts or reagents. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, ensuring consistent output even when sourcing from different vendors. This stability is crucial for maintaining continuous production schedules and meeting the strict delivery timelines required by downstream pharmaceutical clients who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The avoidance of column chromatography in favor of recrystallization makes this process inherently scalable, as crystallization tanks can be easily enlarged to meet increasing demand without complex engineering changes. The reduction in solvent usage and the elimination of phosphorus or heavy metal waste streams associated with alternative methods simplify wastewater treatment and regulatory compliance. This environmental friendliness not only reduces the cost of waste management but also aligns with the increasing corporate sustainability goals of global pharmaceutical companies seeking green chemistry partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity on its feasibility and advantages over legacy methods. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is essential for R&D teams evaluating the technology for technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: Why is the Perkin reaction preferred over Wittig reaction for this synthesis?
A: The Perkin reaction avoids the need for strict anhydrous conditions and expensive phosphorus reagents required in Wittig reactions, significantly reducing raw material costs and operational complexity.
Q: How is high trans-selectivity achieved without protection groups?
A:
Q: What are the purification advantages of this method?
A: The method allows for simple recrystallization purification instead of complex column chromatography, which drastically improves scalability and reduces solvent waste in commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Polyhydroxy Diphenyl Ethylene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, leveraging advanced technologies like the one described in CN101481300B to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trans-polyhydroxy diphenyl ethylene meets the highest standards required for pharmaceutical and nutraceutical applications. Our commitment to quality and consistency makes us the preferred choice for companies seeking a long-term strategic partner in the fine chemical sector.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific product portfolio and cost structure. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic improvements this technology offers for your supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that you have all the necessary information to make informed sourcing decisions. Let us help you engineer a more efficient and cost-effective supply chain for your critical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
