Advanced Heck Coupling Strategy for Commercial Scale-up of Trans-Resveratrol Intermediates
Advanced Heck Coupling Strategy for Commercial Scale-up of Trans-Resveratrol Intermediates
The pharmaceutical and nutraceutical industries are constantly seeking more efficient, sustainable, and cost-effective routes for the synthesis of high-value bioactive compounds like trans-resveratrol. A groundbreaking approach detailed in patent CN102976938A introduces a novel method for obtaining trans-resveratrol through a Heck cross-coupling reaction mediated by ionic liquids. This technology represents a significant departure from traditional synthetic pathways, offering a green, environmentally friendly alternative that utilizes ionic liquids to facilitate the coupling reaction. The method is characterized by its simplicity, high total yield, and low production costs, making it exceptionally convenient for post-reaction treatment and industrial application. By leveraging the unique properties of ionic liquids and transition metal salts, this process addresses critical bottlenecks in the manufacturing of this potent antioxidant, positioning it as a highly viable candidate for large-scale commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trans-resveratrol has relied heavily on extraction from natural plant sources or chemical synthesis via Wittig-Horner and Perkin condensation reactions. Extraction methods suffer from inherently low yields due to the minimal content of resveratrol in plants like Polygonum cuspidatum, alongside issues with uncertain impurity profiles and massive waste generation. On the chemical synthesis front, the Wittig-Horner reaction, while an improvement over the classic Wittig reaction, still necessitates the use of toxic trialkyl phosphites and involves hazardous Arbuzov rearrangement steps that require violent reaction conditions. Furthermore, these traditional routes often struggle with stereoselectivity, producing mixtures of (E) and (Z) isomers that are notoriously difficult to separate, thereby complicating purification and reducing overall process efficiency. The reliance on expensive and toxic catalysts, such as ruthenium-based Grubbs catalysts in metathesis reactions, further exacerbates cost and environmental concerns, limiting their suitability for industrial-scale operations.
The Novel Approach
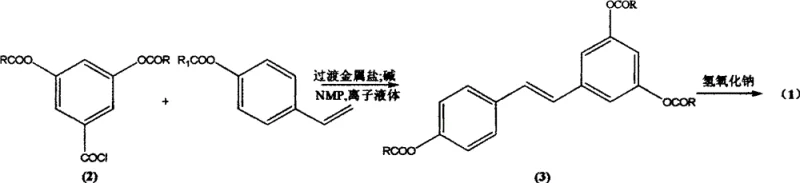
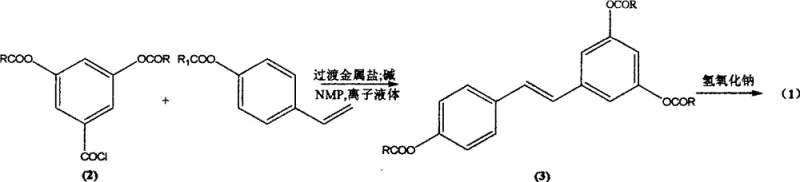
In stark contrast to these legacy methods, the innovative process described in the patent utilizes a Heck cross-coupling reaction that completely abandons the need for classical phosphine ligands and harsh condensation conditions. By employing readily available starting materials such as 3,5-diacetoxybenzoyl chloride and p-acetoxystyrene, the method achieves the construction of the stilbene skeleton with exceptional stereocontrol. The core innovation lies in the substitution of traditional volatile organic solvents with ionic liquids, which not only accelerates the reaction rate but also allows for operation at milder temperatures ranging from 40°C to 100°C. This approach effectively eliminates the formation of unwanted (Z)-isomers, ensuring the direct acquisition of the desired (E)-configuration with high selectivity. The result is a streamlined synthesis that bypasses complex purification steps, yielding a highly purified intermediate that can be easily converted to the final target product through simple saponification.
Mechanistic Insights into Ionic Liquid-Mediated Heck Coupling
The mechanistic foundation of this synthesis rests on the unique interaction between the transition metal catalyst, the substrate, and the ionic liquid medium. In this system, N-methyl-2-pyrrolidone (NMP) serves a dual purpose: acting as a co-solvent and effectively replacing expensive phosphine ligands to stabilize the active palladium species. The ionic liquid, typically a 1-butyl-3-methylimidazolium salt such as [Bmim][BF4], creates a highly organized polar environment that facilitates the oxidative addition of the aryl halide to the metal center. This specific solvation shell enhances the electrophilicity of the substrate and stabilizes the transition state of the migratory insertion step, which is crucial for forming the carbon-carbon double bond. The absence of bulky phosphine ligands reduces steric hindrance around the metal center, allowing for a more efficient catalytic cycle that proceeds rapidly even at lower catalyst loadings, typically in the range of 1:2000 to 1:10000 relative to the substrate.
Impurity control in this process is intrinsically linked to the thermodynamic stability of the product within the ionic liquid matrix. The reaction conditions favor the formation of the trans-(E) isomer, which is thermodynamically more stable than the cis-(Z) counterpart. The ionic liquid environment likely suppresses the isomerization pathways that lead to the Z-isomer, a common issue in homogeneous catalysis in conventional organic solvents. Additionally, the use of inorganic bases like tripotassium phosphate or cesium carbonate ensures efficient deprotonation of the intermediate without introducing nucleophilic side reactions that could degrade the sensitive acetoxy protecting groups. The final saponification step is equally critical, as it cleanly removes the acetyl groups under mild alkaline conditions, preventing the degradation of the stilbene backbone and ensuring the final product meets stringent purity specifications of greater than 99% as determined by HPLC analysis.
How to Synthesize 3,4',5-Triacetoxy Stilbene Efficiently
The synthesis of the key intermediate, 3,4',5-triacetoxy stilbene, serves as the cornerstone of this entire production route. The process begins by charging a reactor with the acyl chloride and vinylbenzene derivatives under an inert nitrogen atmosphere to prevent oxidation of the sensitive palladium catalyst. The reaction is conducted in the presence of the ionic liquid and NMP, with the temperature carefully ramped to optimize the coupling efficiency while minimizing side reactions. Following the coupling, the workup procedure is remarkably straightforward; the reaction mixture is cooled, and the solid product precipitates upon the addition of water, allowing for simple filtration. This eliminates the need for energy-intensive distillation or column chromatography, significantly reducing the operational complexity and solvent consumption associated with traditional pharmaceutical manufacturing. For the complete standardized operating procedure and specific parameter optimization, please refer to the detailed guide below.
- Prepare the reaction mixture by combining 3,5-diacetoxybenzoyl chloride and p-acetoxystyrene in an ionic liquid solvent such as [Bmim][BF4] with a transition metal salt catalyst.
- Add N-methyl-2-pyrrolidone (NMP) as a ligand substitute and an inorganic base, then heat the mixture to 40-100°C for 0.1 to 2 hours to facilitate the Heck cross-coupling reaction.
- Filter the reaction mixture to recover the catalyst, hydrolyze the acetoxy groups via saponification with potassium hydroxide, and recrystallize the final product to obtain pure trans-resveratrol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. The elimination of expensive phosphine ligands and the drastic reduction in transition metal catalyst loading translate into substantial raw material cost savings. Traditional Heck couplings often require stoichiometric or near-stoichiometric amounts of costly ligands to maintain catalyst activity, whereas this system operates efficiently with minimal metal salt input. Furthermore, the ability to recover and recycle the ionic liquid solvent creates a closed-loop system that minimizes waste disposal costs and reduces the dependency on volatile organic solvents, which are subject to fluctuating market prices and increasingly strict environmental regulations. These factors combine to create a robust economic model that supports long-term price stability for high-purity trans-resveratrol intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of high-cost reagents with commodity chemicals. By utilizing inexpensive transition metal salts instead of complex organometallic catalysts and avoiding toxic phosphites, the direct material cost is significantly lowered. The high yield of the coupling step, reported to be between 99% and 100%, ensures that raw material utilization is maximized, reducing the cost per kilogram of the final API intermediate. Additionally, the simplified workup procedure reduces labor and utility costs associated with solvent recovery and waste treatment, contributing to a leaner and more competitive manufacturing cost structure.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials such as acetoxy-substituted benzoyl chlorides and styrenes, which are produced by numerous global chemical suppliers. This diversifies the supplier base and mitigates the risk of shortages associated with specialized reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and base types, ensures consistent production output even with minor variations in raw material quality. This reliability is crucial for maintaining continuous supply lines to downstream pharmaceutical manufacturers who require just-in-time delivery of critical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable due to its heterogeneous nature and the ease of catalyst separation by simple filtration. This avoids the engineering challenges associated with removing trace metals from homogeneous solutions, which often require complex scavenging resins or extensive washing steps. The use of non-volatile, non-flammable ionic liquids enhances plant safety by reducing fire hazards and VOC emissions, facilitating compliance with stringent environmental, health, and safety (EHS) standards. This green chemistry profile not only future-proofs the manufacturing site against tightening regulations but also appeals to end-customers seeking sustainably sourced ingredients for their nutraceutical and pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid-mediated Heck coupling technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this route for their own production pipelines and for procurement specialists assessing the quality and consistency of the supply.
Q: What are the primary advantages of using ionic liquids in this Heck coupling synthesis?
A: The use of ionic liquids like [Bmim][BF4] significantly enhances reaction rates and allows for lower reaction temperatures compared to traditional organic solvents. Furthermore, ionic liquids are non-volatile, non-flammable, and can be recycled, which drastically reduces waste generation and aligns with green chemistry principles for industrial production.
Q: How does this method improve stereoselectivity compared to Wittig-Horner reactions?
A: Traditional Wittig-Horner reactions often produce a mixture of E and Z isomers, requiring difficult purification steps. This novel Heck coupling method utilizes specific reaction conditions and the steric environment of the ionic liquid to achieve high stereoselectivity for the thermodynamically stable (E)-isomer, resulting in a product purity greater than 99% without complex chromatographic separation.
Q: Is the transition metal catalyst recoverable in this process?
A: Yes, one of the key economic benefits of this process is the ease of catalyst recovery. Since the transition metal salt is insoluble in the reaction system under the specific conditions used, it can be separated from the product simply by filtration. This eliminates the need for expensive and toxic metal scavenging resins often required in homogeneous catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Resveratrol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ionic liquid-mediated Heck coupling for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust, commercial-scale manufacturing operations. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are realized in actual plant output. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of trans-resveratrol meets the exacting standards required by the global pharmaceutical and nutraceutical markets.
We invite you to collaborate with us to optimize your supply chain for trans-resveratrol and related stilbene derivatives. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our mastery of green chemistry and process intensification, we can help you reduce lead times and secure a stable supply of high-purity intermediates. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
