Advanced Silver Oxide Catalysis for Scalable Benzothiophene Production and Commercial Supply
Advanced Silver Oxide Catalysis for Scalable Benzothiophene Production and Commercial Supply
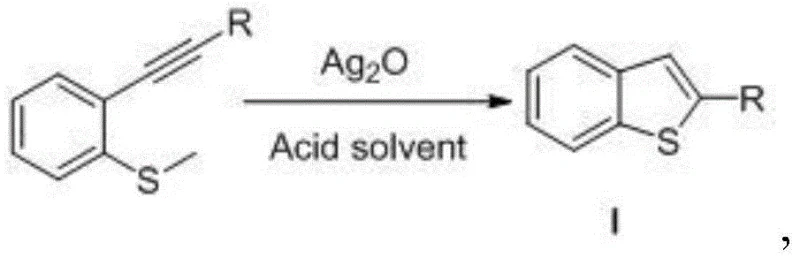
The landscape of heterocyclic compound synthesis is undergoing a significant transformation driven by the need for greener, more cost-effective methodologies that do not compromise on yield or purity. A pivotal development in this sector is detailed in patent CN110483475B, which introduces a novel method for preparing benzothiophene compounds utilizing a silver oxide catalytic system. Benzothiophenes are critical scaffolds in modern medicinal chemistry and materials science, serving as essential building blocks for liquid crystal polymers, organic electronic materials, and potent agrochemical agents. The traditional reliance on harsh conditions or expensive noble metals has long been a bottleneck for procurement teams seeking reliable supply chains. This new approach leverages the unique catalytic properties of silver oxide in an acidic medium to facilitate the intramolecular cyclization of 2-ethynyl methyl sulfide derivatives, offering a robust alternative that addresses both economic and environmental concerns in fine chemical manufacturing.
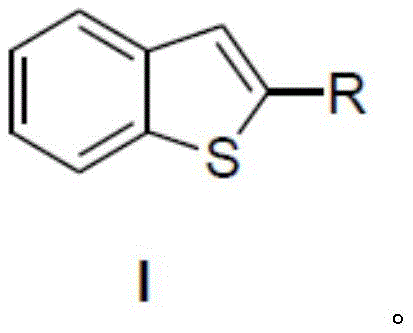
This technological breakthrough is particularly relevant for R&D directors focusing on the scalability of complex pharmaceutical intermediates. By shifting away from transition metals that require rigorous removal steps to meet ppm-level specifications, this silver-catalyzed route simplifies the downstream purification process. The general structure of the target compounds, as illustrated in the patent, allows for significant diversification at the R-position, enabling the synthesis of a wide array of derivatives tailored for specific biological activities or material properties. For a reliable benzothiophene supplier, mastering this specific catalytic cycle means being able to offer a broader portfolio of high-purity intermediates while maintaining competitive pricing structures that are increasingly demanded by global generic drug manufacturers and agrochemical formulators.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiophene cores has relied heavily on methods that present substantial operational and environmental challenges. Conventional routes often employ palladium or copper catalysts mediated by electrophilic halogen sources such as iodine, bromine, or N-bromosuccinimide (NBS). While these methods can achieve high yields, they generate significant quantities of halogen-containing waste streams that are environmentally hazardous and costly to treat, violating the principles of green chemistry that modern chemical plants strive to uphold. Furthermore, alternative approaches utilizing gold(I) catalysts, while effective, involve prohibitively expensive metal inputs that drastically inflate the cost of goods sold (COGS), making them less viable for large-scale commercial production where margin compression is a constant pressure. Other radical-mediated cyclizations often suffer from poor selectivity and the formation of complex by-product mixtures, necessitating resource-intensive purification protocols that reduce overall throughput and extend lead times for high-purity intermediates.
The Novel Approach
The methodology disclosed in CN110483475B represents a paradigm shift by utilizing silver oxide (Ag2O) as a cost-effective and efficient catalyst in the presence of an acidic solvent. This novel approach effectively activates the 2-ethynyl methyl sulfide precursor under mild conditions, typically at room temperature, thereby eliminating the need for energy-intensive heating or cryogenic cooling. The use of simple carboxylic acids like acetic acid or trifluoroacetic acid not only serves as the reaction medium but also plays a crucial role in protonating intermediates to drive the cyclization forward. This strategy circumvents the need for expensive ligands or toxic halogenating agents, resulting in a cleaner reaction profile. For procurement managers, this translates directly into cost reduction in heterocyclic compound manufacturing, as the raw material costs are significantly lower and the waste disposal burden is minimized. The simplicity of the workup procedure, involving basic neutralization and standard extraction, further enhances the operational efficiency, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ag2O-Catalyzed Cyclization
The mechanistic pathway of this silver oxide-catalyzed transformation is rooted in the ability of the silver cation to coordinate with the alkyne moiety of the substrate, increasing its electrophilicity and facilitating nucleophilic attack by the adjacent sulfur atom. In the presence of an acidic solvent, the reaction environment promotes the formation of a vinyl-silver intermediate, which subsequently undergoes protodemetalation to yield the final benzothiophene ring system. This mechanism is distinct from radical pathways, offering superior control over regioselectivity and minimizing the formation of polymeric side products. The acidic medium ensures that the sulfur nucleophile remains sufficiently reactive while stabilizing the transition states involved in the ring closure. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters, as the interplay between the silver catalyst loading and the acidity of the solvent dictates the reaction kinetics. The patent data indicates that maintaining a weakly acidic environment is crucial; strong mineral acids might lead to substrate decomposition, whereas insufficient acidity results in sluggish reaction rates.
Impurity control is another critical aspect where this mechanism offers distinct advantages. Unlike halogen-mediated methods that can leave residual iodine or bromine impurities difficult to remove, the silver oxide method primarily generates inorganic silver salts as by-products, which are easily removed during the aqueous workup or filtration steps. The mild reaction temperature (15-30°C) prevents thermal degradation of sensitive functional groups such as aldehydes or nitro groups, which are often present in advanced intermediates. This tolerance for diverse functional groups expands the utility of the method for synthesizing specialized derivatives required in crop protection and drug discovery. The high conversion rates reported, often exceeding 90%, suggest that the catalytic cycle is highly efficient, with minimal catalyst deactivation. For quality assurance teams, this implies a more consistent impurity profile across different batches, simplifying the validation process for regulatory filings and ensuring that the final API or agrochemical active meets stringent purity specifications without requiring multiple recrystallization steps.
How to Synthesize Benzothiophene Efficiently
Implementing this synthesis route in a pilot or production plant requires careful attention to the stoichiometry of the catalyst and the choice of solvent to maximize yield and minimize cycle time. The patent outlines a straightforward protocol where the substrate, silver oxide, and acidic solvent are combined and stirred at ambient temperatures, removing the need for complex reactor heating or cooling jackets. Detailed standardized synthetic steps see the guide below, which encapsulates the optimized conditions derived from extensive experimental screening. This operational simplicity reduces the training burden on plant operators and lowers the risk of batch failures due to procedural errors. By adhering to the specified catalyst loading ranges of 1-10 mol%, manufacturers can fine-tune the process to balance reaction speed against catalyst cost, ensuring economic viability at multi-ton scales.
- Charge a reaction vessel with 2-ethynyl methyl sulfide derivatives and silver oxide catalyst (1-10 mol%) in an acidic solvent such as acetic acid.
- Stir the mixture at room temperature (15-30°C) for approximately 1.5 to 3 hours to ensure complete conversion.
- Quench the reaction with aqueous sodium hydroxide, extract with dichloromethane, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement strategists, the adoption of this silver oxide-catalyzed technology offers tangible benefits that extend beyond mere chemical efficiency. The shift towards this methodology addresses several pain points associated with the sourcing of heterocyclic building blocks, particularly regarding cost volatility and supply continuity. By utilizing commodity-grade reagents like silver oxide and acetic acid, the dependency on scarce or geopolitically sensitive precious metals is reduced. This stability in raw material sourcing is crucial for maintaining uninterrupted production schedules, especially in a global market prone to logistical disruptions. Furthermore, the simplified purification workflow reduces the consumption of solvents and silica gel, contributing to a leaner manufacturing process that aligns with sustainability goals and reduces the overall carbon footprint of the supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive gold catalysts and toxic halogenating agents results in a direct decrease in raw material expenditures. Additionally, the mild reaction conditions significantly lower energy consumption costs associated with heating and cooling, while the simplified workup reduces labor and waste treatment expenses. These cumulative savings allow for a more competitive pricing structure without compromising on the quality of the final intermediate, providing a strategic advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: The use of widely available and stable reagents ensures that production is not held hostage by the supply constraints of exotic ligands or specialized catalysts. The robustness of the reaction across a wide range of substrates means that a single production line can be adapted to manufacture various benzothiophene derivatives, increasing asset utilization and flexibility. This adaptability is key to responding quickly to changing market demands and ensuring timely delivery of critical intermediates to downstream customers.
- Scalability and Environmental Compliance: The absence of hazardous halogen waste simplifies environmental compliance and reduces the cost of effluent treatment. The reaction's ability to proceed efficiently at room temperature makes it inherently safer and easier to scale from kilogram to multi-ton quantities without encountering heat transfer limitations. This scalability ensures that the technology can support commercial volume requirements, providing a secure source of supply for long-term contracts and large-scale API manufacturing campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver oxide catalytic method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this technology. Understanding these nuances helps stakeholders make informed decisions about integrating this synthetic route into their existing portfolios.
Q: What are the advantages of using Silver Oxide over traditional Palladium or Gold catalysts?
A: Silver oxide offers a significantly lower cost profile compared to expensive gold catalysts and avoids the generation of hazardous halogenated waste associated with iodine-mediated palladium cyclizations, aligning better with green chemistry principles.
Q: What represents the optimal reaction conditions for this cyclization?
A: The patent specifies that using acetic acid or trifluoroacetic acid as the solvent at room temperature (25°C) with a catalyst loading of 5-8 mol% provides the best balance of reaction rate and yield, often exceeding 90%.
Q: Can this method be applied to substrates with diverse electronic properties?
A: Yes, the method demonstrates broad substrate scope, successfully tolerating various substituents including alkyl groups, halogens, nitro groups, and aldehydes on the aromatic ring without significant loss in efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to meet the evolving needs of the global pharmaceutical and agrochemical industries. Our technical team has thoroughly analyzed the potential of the silver oxide-catalyzed route described in CN110483475B and is well-positioned to leverage this knowledge for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzothiophene intermediate we deliver meets the highest standards of quality and consistency required for regulatory submissions.
We invite you to collaborate with us to explore how this cost-effective and environmentally friendly synthesis method can enhance your supply chain resilience. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in heterocyclic chemistry can drive value and innovation in your upcoming projects.
