Advanced DABCO-Catalyzed Synthesis of Thiazolidine Intermediates for Commercial Scale-Up
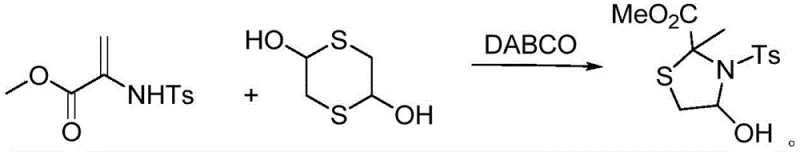
The chemical industry is witnessing a significant paradigm shift towards sustainable and efficient synthetic methodologies, particularly in the realm of heterocyclic compound manufacturing. Patent CN113603655B, published in late 2022, introduces a groundbreaking preparation method for 4-hydroxy-2-methyl-3-(benzenesulfonyl)thiazolidine-2-carboxylic acid methyl ester, a sophisticated intermediate with profound implications for both pharmaceutical and agrochemical sectors. This patent details a novel organocatalytic cyclization strategy that leverages the unique properties of 1,4-dithio-2,5-diol and a sulfonamide-functionalized acrylate derivative. By utilizing triethylenediamine (DABCO) as a mild yet effective catalyst, the invention successfully circumvents the severe limitations associated with conventional thiazolidine synthesis, such as the reliance on expensive transition metals or energy-intensive microwave irradiation. For R&D directors and process chemists, this represents a critical advancement in accessing complex sulfur-nitrogen heterocycles with high atom economy and operational simplicity. The ability to conduct this transformation at ambient temperature not only enhances safety profiles but also drastically reduces the carbon footprint of the manufacturing process, aligning perfectly with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the thiazolidine scaffold, a privileged structure found in numerous bioactive molecules ranging from antibiotics to insecticides, has been fraught with synthetic challenges. Traditional pathways often necessitate the use of aryl isothiocyanates reacting with dithiols under microwave radiation, or the employment of hazardous acyl azides which require elevated temperatures to drive the cyclization forward. These legacy methods suffer from significant drawbacks, including the requirement for specialized equipment capable of handling high-energy inputs and the generation of toxic byproducts that complicate downstream processing. Furthermore, many existing protocols rely on precious metal catalysts or intricate ligand systems that are not only cost-prohibitive for large-scale production but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). The harsh thermal conditions often lead to the decomposition of sensitive functional groups, resulting in lower yields and a complex impurity profile that demands rigorous and expensive purification steps. Consequently, the commercial viability of thiazolidine-based candidates has frequently been hindered by these inefficient and environmentally burdensome synthetic routes.
The Novel Approach
In stark contrast to these archaic techniques, the methodology disclosed in CN113603655B offers a streamlined and robust alternative that redefines efficiency in heterocyclic synthesis. The core innovation lies in the direct cyclization of commercially available 1,4-dithio-2,5-diol with methyl 2-((4-methylphenyl)sulfonamido)acrylate, mediated by a catalytic amount of DABCO. This approach eliminates the need for external heating, proceeding smoothly at room temperature over a period of approximately 12 hours. The reaction demonstrates exceptional tolerance to various organic solvents, with chloroform identified as the optimal medium, although alternatives like dichloromethane and toluene remain viable. By avoiding extreme thermal stress and hazardous reagents, this novel route ensures the integrity of the molecular framework is maintained throughout the transformation. The simplicity of the work-up procedure, involving standard solvent removal followed by column chromatography, further underscores the practical utility of this method for industrial applications. This shift from high-energy, high-risk processes to a mild, ambient-temperature protocol marks a substantial leap forward in process chemistry, offering a reliable pathway for the reliable thiazolidine intermediate supplier market.
Mechanistic Insights into DABCO-Catalyzed Cyclization
The mechanistic elegance of this transformation is rooted in the dual functionality of DABCO as a nucleophilic catalyst and a base. In the initial phase of the reaction, the tertiary amine centers of DABCO likely activate the electron-deficient double bond of the acrylate substrate through nucleophilic attack or hydrogen bonding interactions, thereby increasing its electrophilicity. Simultaneously, the thiol groups of the 1,4-dithio-2,5-diol are primed for nucleophilic attack. The proximity of the two sulfur atoms in the dithiol backbone facilitates an intramolecular cyclization event once the initial intermolecular addition occurs. This cascade leads to the formation of the five-membered thiazolidine ring with high regioselectivity. The mild basicity of DABCO is sufficient to deprotonate the intermediate species without promoting unwanted side reactions such as polymerization or hydrolysis, which are common pitfalls in acrylate chemistry. This delicate balance of reactivity ensures that the reaction proceeds with high fidelity, converting the starting materials into the desired 4-hydroxy-2-methyl-3-(benzenesulfonyl)thiazolidine-2-carboxylic acid methyl ester with minimal formation of oligomeric byproducts.
From an impurity control perspective, the room temperature condition is paramount. Thermal degradation pathways, which often generate difficult-to-remove tars or decomposition products in traditional heating methods, are effectively suppressed. The specific stoichiometry employed, typically a 1:1 molar ratio of the dithiol to the acrylate, prevents the accumulation of unreacted starting materials that could complicate purification. Moreover, the use of an organocatalyst rather than a transition metal means there is no risk of metal leaching, a critical consideration for pharmaceutical intermediates where residual metal limits are strictly regulated by agencies like the FDA and EMA. The resulting product exhibits a clean spectral profile, as evidenced by the sharp NMR signals and high-resolution mass spectrometry data provided in the patent examples. This high level of chemical purity directly translates to reduced processing costs and shorter cycle times in a manufacturing setting, providing a distinct competitive advantage for producers adopting this technology.
How to Synthesize 4-hydroxy-2-methyl-3-(benzenesulfonyl)thiazolidine-2-carboxylic acid methyl ester Efficiently
The execution of this synthesis is designed for operational ease, requiring standard laboratory glassware and commonly available reagents. The process begins with the dissolution of the sulfonamide acrylate precursor and the DABCO catalyst in the chosen organic solvent, followed by the addition of the dithiol component. The mixture is then allowed to stir under ambient conditions, eliminating the need for oil baths, heating mantles, or microwave reactors. Monitoring the reaction progress can be achieved through standard thin-layer chromatography (TLC) or HPLC methods. Once the conversion is deemed complete, typically within the 10 to 14-hour window, the solvent is removed under reduced pressure. The crude solid is then subjected to flash column chromatography to isolate the pure product. This straightforward protocol minimizes the technical expertise required for operation, making it accessible for a wide range of production facilities.
- Prepare the reaction mixture by dissolving methyl 2-((4-methylphenyl)sulfonamido)acrylate and DABCO catalyst in chloroform (CHCl3) at room temperature.
- Add 1,4-dithio-2,5-diol to the solution and maintain stirring at room temperature for approximately 12 hours to ensure complete cyclization.
- Upon completion, remove the solvent via rotary evaporation and purify the crude residue using column chromatography to isolate the high-purity thiazolidine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented methodology offers compelling economic and logistical benefits. The primary driver of cost reduction lies in the substitution of expensive, specialized reagents with commodity chemicals. Both 1,4-dithio-2,5-diol and the acrylate precursor are readily accessible from global chemical suppliers, ensuring a stable and resilient supply chain that is less susceptible to market volatility. The elimination of precious metal catalysts removes a significant line item from the bill of materials, while also obviating the need for costly scavenging resins or filtration steps required to meet heavy metal specifications. Furthermore, the energy savings derived from conducting the reaction at room temperature are substantial when scaled to multi-ton production volumes, directly impacting the utility costs associated with manufacturing. These factors combine to create a highly cost-competitive process for cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The economic model of this synthesis is fundamentally superior to legacy methods due to the drastic simplification of the reagent list. By relying on DABCO, an inexpensive and widely produced organocatalyst, the process avoids the price fluctuations associated with palladium, rhodium, or other transition metals. Additionally, the high yield reported in the patent examples, reaching up to 96 percent, maximizes the throughput of raw materials, minimizing waste disposal costs. The simplified work-up procedure reduces the consumption of silica gel and solvents during purification, further driving down the variable costs per kilogram of product. This efficiency allows manufacturers to offer high-purity thiazolidine derivatives at a more aggressive price point without compromising margins.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust, non-hazardous starting materials. Unlike acyl azides, which are potentially explosive and subject to strict transportation regulations, the reagents used in this process are stable and easy to handle. This reduces the regulatory burden and insurance costs associated with logistics. The flexibility in solvent choice—allowing for the use of chloroform, dichloromethane, or toluene—provides procurement teams with the agility to switch suppliers based on availability and pricing without disrupting the production schedule. This adaptability is crucial for maintaining reducing lead time for high-purity thiazolidine intermediates in a dynamic global market.
- Scalability and Environmental Compliance: The inherent safety of the room-temperature process facilitates seamless commercial scale-up of complex heterocyclic intermediates. There is no need for specialized high-pressure or high-temperature reactors, allowing production to take place in standard stainless steel vessels. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and effluent management, ensuring compliance with increasingly stringent environmental regulations. The high atom economy of the cyclization reaction means less chemical waste is generated per unit of product, aligning with corporate sustainability goals and reducing the carbon tax liabilities often associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of using DABCO over traditional catalysts for thiazolidine synthesis?
A: Unlike traditional methods that often require expensive transition metal catalysts, complex ligands, or harsh conditions like microwave radiation, the DABCO-catalyzed method operates at room temperature. This significantly lowers energy consumption, eliminates the need for costly metal removal steps, and simplifies the overall operational workflow, making it highly suitable for large-scale manufacturing.
Q: How does this synthesis method impact the purity profile of the final intermediate?
A: The mild reaction conditions (room temperature) prevent thermal decomposition of sensitive functional groups, which is a common issue in high-temperature protocols. Furthermore, the high selectivity of the organocatalytic cyclization minimizes the formation of side products, resulting in a cleaner crude reaction mixture that requires less intensive purification to achieve stringent pharmaceutical purity specifications.
Q: Is this process scalable for industrial production of agrochemical intermediates?
A: Yes, the process is highly scalable. It utilizes commercially available, low-cost raw materials and avoids hazardous reagents like acyl azides. The absence of extreme temperatures or pressures reduces engineering constraints on reactor design, facilitating a smoother transition from laboratory benchtop synthesis to multi-ton commercial production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-hydroxy-2-methyl-3-(benzenesulfonyl)thiazolidine-2-carboxylic acid methyl ester Supplier
As the demand for advanced heterocyclic building blocks continues to surge in the development of next-generation agrochemicals and therapeutics, securing a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge synthetic methodologies like the DABCO-catalyzed cyclization described in CN113603655B to deliver superior value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale industrial output. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the exacting standards required for global regulatory filings.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development timelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our advanced manufacturing capabilities can support your long-term strategic goals. Let us be your trusted ally in navigating the complexities of fine chemical synthesis and delivering high-quality intermediates to the global market.
