Scalable Synthesis of 4-Hydroxy Thiazolidine Esters via Mild DABCO Catalysis for Industrial Applications
Scalable Synthesis of 4-Hydroxy Thiazolidine Esters via Mild DABCO Catalysis for Industrial Applications
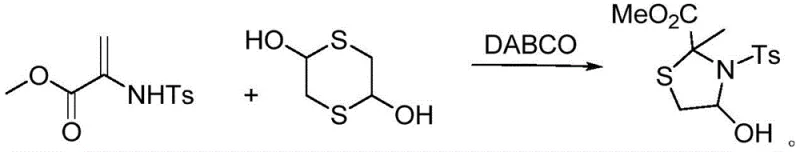
The pharmaceutical and agrochemical industries continuously seek robust synthetic methodologies for constructing complex heterocyclic scaffolds, particularly thiazolidine derivatives which serve as critical building blocks for bioactive molecules. Patent CN113603655A introduces a groundbreaking preparation method for 4-hydroxy-2-methyl-3-(benzenesulfonyl)thiazolidine-2-carboxylic acid methyl ester, a sophisticated intermediate with significant potential in the development of next-generation pesticides and therapeutic agents. This innovation leverages a mild, organocatalytic cyclization strategy that operates efficiently at room temperature, circumventing the harsh conditions typically associated with heterocycle synthesis. By utilizing commercially available 1,4-dithio-2,5-diol and a sulfonamide-functionalized acrylate, the process achieves exceptional conversion rates without the need for expensive transition metal catalysts or energy-intensive heating protocols. For R&D directors and process chemists, this represents a paradigm shift towards greener, more sustainable manufacturing practices that do not compromise on molecular complexity or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the thiazolidine core has been fraught with synthetic challenges that hinder efficient commercial production. Traditional routes often rely on the cyclization of aryl isothiocyanates with dithiols under microwave irradiation or the reaction of dithiols with acyl azides requiring elevated temperatures. These legacy methods impose severe constraints on process scalability, primarily due to the requirement for specialized equipment capable of handling high-energy inputs and the inherent safety risks associated with thermal instability of azide reagents. Furthermore, conventional protocols frequently necessitate the use of costly ligands or transition metal catalysts to drive the reaction to completion, which introduces significant downstream purification burdens to remove trace metal impurities that are strictly regulated in final API or agrochemical products. The reliance on harsh conditions also limits the functional group tolerance, often leading to side reactions and complex impurity profiles that reduce overall process yield and increase waste generation.
The Novel Approach
In stark contrast to these cumbersome traditional techniques, the methodology disclosed in CN113603655A utilizes a simple yet highly effective organocatalytic system driven by triethylenediamine (DABCO). This novel approach enables the direct cyclization of 1,4-dithio-2,5-diol with methyl 2-((4-methylphenyl)sulfonamido)acrylate under ambient conditions, eliminating the need for external heating or microwave assistance. The reaction proceeds smoothly in common organic solvents such as chloroform, dichloromethane, or toluene, demonstrating remarkable versatility and operational simplicity. By avoiding extreme temperatures and hazardous reagents, this new pathway significantly enhances the safety profile of the manufacturing process while simultaneously improving the atom economy. The mild nature of the reaction preserves the integrity of sensitive functional groups, such as the methyl ester and the sulfonyl moiety, ensuring that the final product retains high structural fidelity suitable for subsequent derivatization steps in drug discovery pipelines.
Mechanistic Insights into DABCO-Catalyzed Thiazolidine Formation
The success of this synthetic route lies in the unique mechanistic role played by the DABCO catalyst, which facilitates the nucleophilic attack and subsequent ring closure through a concerted base-catalyzed pathway. The reaction initiates with the activation of the nucleophilic sulfur centers on the 1,4-dithio-2,5-diol, enhancing their reactivity towards the electron-deficient double bond of the acrylate substrate. DABCO, acting as a non-nucleophilic base, likely assists in deprotonating intermediate species or stabilizing the transition state, thereby lowering the activation energy barrier for the cyclization event. This catalytic cycle allows the reaction to proceed rapidly at room temperature, a feat that is difficult to achieve with uncatalyzed thermal methods. The presence of the tosyl group on the nitrogen atom further influences the electronic environment of the forming ring, stabilizing the developing negative charge during the cyclization process and directing the regioselectivity to favor the formation of the desired 2-methyl-4-hydroxy thiazolidine scaffold over potential isomeric byproducts.
Impurity control is another critical aspect where this mechanism offers distinct advantages over prior art. In traditional high-temperature syntheses, thermal degradation of the starting materials or the product often leads to the formation of polymeric tars or decomposed sulfur-containing species that are difficult to separate. However, the room temperature operation of the DABCO-catalyzed process minimizes thermal stress on the reaction mixture, effectively suppressing these degradation pathways. The high selectivity observed, with yields reaching up to 96% in optimized examples, suggests that the kinetic pathway favored by the catalyst is highly specific, leaving very little room for competing side reactions. This intrinsic selectivity simplifies the downstream purification process, often allowing for straightforward isolation techniques such as column chromatography or potentially crystallization upon further process optimization, which is vital for maintaining high purity specifications required by regulatory bodies for agrochemical and pharmaceutical intermediates.
How to Synthesize 4-Hydroxy-2-methyl-3-(benzenesulfonyl)thiazolidine-2-carboxylic Acid Methyl Ester Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow the optimized protocol which emphasizes precise stoichiometry and controlled addition rates to maximize yield. The process begins by dissolving the acrylate precursor and the DABCO catalyst in the chosen solvent, followed by the gradual introduction of the dithiol to manage exothermicity, although the reaction is generally mild. Maintaining the reaction at room temperature for a duration of approximately 12 hours ensures complete conversion without risking product decomposition. Detailed standardized operating procedures regarding specific mixing speeds, quenching methods, and workup parameters are essential for reproducibility and are outlined in the technical guide below.
- Charge methyl 2-((4-methylphenyl)sulfonamido)acrylate and triethylenediamine (DABCO) into a reaction vessel with chloroform solvent.
- Add 1,4-dithio-2,5-diol to the mixture and stir at room temperature for approximately 12 hours to ensure complete cyclization.
- Upon completion, perform rotary evaporation to remove solvents and purify the crude residue via column chromatography to isolate the high-purity thiazolidine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers transformative benefits that directly impact the bottom line and operational resilience. The primary advantage stems from the utilization of commodity chemicals as starting materials; 1,4-dithio-2,5-diol and sulfonamide acrylates are readily available from multiple global suppliers, reducing the risk of single-source dependency and ensuring a stable supply of raw materials. This accessibility contrasts sharply with older methods that might require custom-synthesized, expensive isothiocyanates or unstable azides, which often suffer from long lead times and volatile pricing. By shifting to a platform based on stable, bulk-available feedstocks, procurement managers can negotiate better contracts and secure long-term supply agreements that protect against market fluctuations.
- Cost Reduction in Manufacturing: The economic implications of switching to this DABCO-catalyzed method are profound, primarily driven by the elimination of costly inputs and energy consumption. Since the reaction proceeds at room temperature, there is no requirement for energy-intensive heating mantles, oil baths, or microwave reactors, leading to a drastic reduction in utility costs per kilogram of product produced. Furthermore, the catalyst itself, triethylenediamine, is an inexpensive, commodity organic base, removing the financial burden associated with precious metal catalysts like palladium or rhodium and their requisite ligands. The high yield reported in the patent implies that less raw material is wasted to side products, effectively lowering the cost of goods sold (COGS) by maximizing the output from every batch of input materials.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the robustness and simplicity of this chemical process. The tolerance for various solvents, including chloroform, dichloromethane, toluene, and THF, provides manufacturing flexibility; if one solvent faces a shortage or regulatory restriction, the process can be adapted to use an alternative without re-validating the entire chemistry. Additionally, the mild reaction conditions reduce the wear and tear on reactor vessels and associated equipment, minimizing unplanned downtime for maintenance. The simplicity of the workup, involving standard evaporation and chromatography, means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without requiring specialized infrastructure, thereby diversifying the supply base.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is inherently safer and more environmentally compliant due to the absence of hazardous thermal conditions. The room temperature operation eliminates the risk of thermal runaway, a critical safety consideration when scaling exothermic reactions, thus reducing the need for complex engineering controls or specialized pressure-rated reactors. From an environmental standpoint, the avoidance of heavy metal catalysts simplifies waste stream management, as there is no need for expensive and complex heavy metal scavenging steps to meet stringent discharge limits. This aligns perfectly with modern green chemistry initiatives and helps companies meet their sustainability goals by reducing the overall environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiazolidine synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this route for potential licensing or in-house production. Understanding these nuances is crucial for assessing the feasibility of integrating this method into existing manufacturing portfolios.
Q: What is the optimal catalyst loading for this thiazolidine cyclization?
A: According to patent CN113603655A, the optimal catalyst loading for triethylenediamine (DABCO) is approximately 10 mole% relative to the dithiol substrate, balancing reaction rate and cost efficiency.
Q: Can the reaction solvent be substituted for large-scale production?
A: Yes, while chloroform is preferred for optimal yield, the patent indicates that dichloromethane, diethyl ether, toluene, or THF can also be utilized, offering flexibility for process safety and environmental compliance during scale-up.
Q: How does temperature affect the stability of the thiazolidine product?
A: The reaction is best conducted at room temperature; increasing the temperature to 50°C leads to product decomposition and significantly lower yields, highlighting the thermal sensitivity of the heterocyclic system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy Thiazolidine Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced heterocyclic intermediates like the 4-hydroxy-2-methyl-3-(benzenesulfonyl)thiazolidine derivative in the development of high-performance agrochemicals and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for global regulatory submissions. Our commitment to quality assurance ensures that clients receive materials with consistent impurity profiles, facilitating smoother downstream processing and final product registration.
We invite procurement leaders and R&D heads to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our expertise in process optimization and supply chain management, we can help you identify opportunities to reduce total landed costs while securing a reliable source of this critical intermediate. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and samples for your evaluation, allowing you to validate the superior quality and performance of our materials in your own applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
