Scalable Synthesis of 4-Hydroxy-2-Methyl-3-(Benzenesulfonyl)Thiazolidine-2-Carboxylic Acid Methyl Ester via Ambient Catalysis
Scalable Synthesis of 4-Hydroxy-2-Methyl-3-(Benzenesulfonyl)Thiazolidine-2-Carboxylic Acid Methyl Ester via Ambient Catalysis
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics and crop protection agents. A recent technological breakthrough documented in patent CN113603655B introduces a highly efficient preparation method for 4-hydroxy-2-methyl-3-(benzenesulfonyl)thiazolidine-2-carboxylic acid methyl ester, a sophisticated thiazolidine derivative with significant potential in medicinal chemistry. This innovation addresses long-standing challenges in heterocyclic synthesis by utilizing a mild, organocatalytic approach that operates effectively at room temperature, thereby circumventing the energy-intensive and hazardous conditions typically associated with traditional ring-closure methodologies. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards greener, more cost-effective manufacturing protocols that do not compromise on chemical purity or structural integrity.
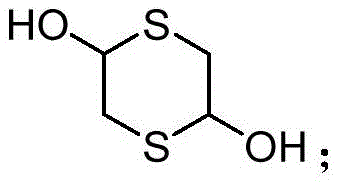
Thiazolidine compounds constitute a vital class of five-membered heterocycles containing both sulfur and nitrogen atoms, known for their diverse biological activities ranging from anti-inflammatory and anticancer properties to potent insecticidal effects. The ability to introduce a thiazolidinyl group into various molecular architectures often enhances the bioactivity of the parent compound, making these structures highly coveted in drug discovery pipelines. However, the historical difficulty in synthesizing these cores with high regioselectivity and yield has often bottlenecked development projects. The methodology outlined in CN113603655B leverages the unique reactivity of 1,4-dithiane-2,5-diol, a readily accessible commodity chemical, reacting it with functionalized acrylates to forge the thiazolidine ring system with exceptional precision and minimal waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazolidine derivatives relying on 1,4-dithio-2,5-diol precursors has been plagued by significant technical hurdles that hinder industrial adoption. Conventional literature often describes protocols requiring harsh reaction conditions, such as high-energy microwave irradiation or elevated thermal temperatures, which pose safety risks and increase operational expenditures. Furthermore, many existing routes depend on the use of expensive transition metal catalysts or specialized ligands that are not only costly to procure but also necessitate rigorous downstream purification steps to remove trace metal residues to meet stringent pharmaceutical standards. Alternative methods involving aryl isothiocyanates or acyl azides often suffer from poor atom economy and generate hazardous byproducts, complicating waste management and environmental compliance. These factors collectively contribute to extended lead times and inflated production costs, making the reliable supply of high-purity thiazolidine intermediates a persistent challenge for global supply chains.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in patent CN113603655B utilizes a streamlined, one-pot cyclization strategy driven by triethylenediamine (DABCO) as an inexpensive and non-toxic organocatalyst. This method allows the reaction between 1,4-dithiane-2,5-diol and methyl 2-((4-methylphenyl)sulfonamido)acrylate to proceed smoothly at room temperature, eliminating the need for external heating sources or pressure vessels. The reaction kinetics are optimized to reach completion within a standard 12-hour window, achieving impressive yields of up to 96% without the formation of significant polymeric byproducts or decomposition species. By employing chloroform as the preferred solvent, the process ensures excellent solubility of reactants while facilitating easy product isolation through simple rotary evaporation and column chromatography. This paradigm shift not only simplifies the operational workflow but also drastically reduces the carbon footprint associated with the manufacturing of these valuable fine chemical intermediates.
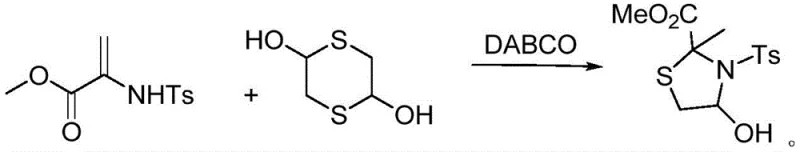
Mechanistic Insights into DABCO-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the dual activation capability of the DABCO catalyst, which facilitates the nucleophilic attack of the sulfur atoms in 1,4-dithiane-2,5-diol onto the electron-deficient double bond of the acrylate substrate. The reaction initiates with the coordination of the catalyst, which enhances the nucleophilicity of the dithiol species, promoting a conjugate addition that sets the stage for subsequent intramolecular cyclization. This pathway avoids the high-energy transition states required in thermal cyclizations, thereby preserving the stereochemical integrity of the sensitive hydroxyl and ester functionalities present in the molecule. The mild conditions prevent the retro-aldol type decompositions or elimination reactions that often plague thiazolidine synthesis at elevated temperatures, ensuring that the final product retains its structural fidelity and biological potential.
From an impurity control perspective, the specificity of this organocatalytic system is paramount for producing pharmaceutical-grade intermediates. The use of a stoichiometric balance between the dithiol and the acrylate, specifically a 1:1 molar ratio, minimizes the presence of unreacted starting materials that could complicate purification. Moreover, the avoidance of strong acids or bases prevents the hydrolysis of the methyl ester group or the cleavage of the sulfonamide protecting group, which are common side reactions in less selective protocols. The resulting crude product is remarkably clean, allowing for straightforward purification via standard silica gel chromatography to achieve the high purity levels required for downstream coupling reactions. This level of control over the reaction profile is essential for maintaining a consistent quality profile across different production batches, a key requirement for regulatory compliance in the life sciences sector.
How to Synthesize 4-Hydroxy-2-Methyl-3-(Benzenesulfonyl)Thiazolidine-2-Carboxylic Acid Methyl Ester Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings, requiring only standard glassware and ambient environmental controls. The protocol begins with the dissolution of the sulfonamide acrylate and the DABCO catalyst in chloroform, followed by the controlled addition of the dithiol reactant to initiate the exothermic cyclization process. Operators are advised to maintain strict temperature monitoring to ensure the reaction remains within the optimal room temperature range, as deviations can impact the final yield and purity profile. After the stipulated reaction time, the workup involves simple solvent removal and chromatographic separation, making this an ideal candidate for technology transfer to manufacturing sites lacking specialized high-pressure equipment.
- Dissolve methyl 2-((4-methylphenyl)sulfonamido)acrylate and triethylenediamine (DABCO) catalyst in chloroform solvent under ambient conditions.
- Add 1,4-dithiane-2,5-diol to the reaction mixture and stir continuously for 12 hours to ensure complete cyclization.
- Upon completion, remove the solvent via rotary evaporation and purify the crude residue using column chromatography to isolate the high-purity thiazolidine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic advantages that extend beyond mere technical feasibility. The reliance on commodity chemicals such as 1,4-dithiane-2,5-diol and DABCO ensures a stable and resilient supply base, mitigating the risks associated with sourcing exotic or single-source reagents. The elimination of expensive metal catalysts translates directly into reduced raw material costs and simplified waste disposal protocols, as there is no need for costly heavy metal scavenging steps or specialized hazardous waste treatment. Furthermore, the ambient temperature operation significantly lowers energy consumption compared to reflux or microwave-assisted processes, contributing to a more sustainable and economically viable manufacturing model that aligns with modern corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of precious metal catalysts with inexpensive organic amines and the reduction of energy inputs required for heating. By operating at room temperature, facilities can avoid the capital expenditure associated with high-temperature reactors and the ongoing operational costs of energy consumption. Additionally, the high yield and selectivity of the reaction minimize the loss of valuable starting materials, ensuring that the cost per kilogram of the final intermediate is significantly optimized. This efficiency allows for competitive pricing strategies in the global market for fine chemical intermediates, providing a distinct margin advantage for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing dependency on complex logistics for temperature-sensitive reagents or specialized catalytic systems. Since the raw materials are widely available from multiple global suppliers, the risk of supply disruption due to geopolitical or logistical issues is markedly decreased. The simplicity of the post-reaction workup also accelerates the production cycle time, enabling faster turnaround from order placement to product delivery. This agility is crucial for meeting the dynamic demands of pharmaceutical clients who require just-in-time delivery of critical intermediates to maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is straightforward due to the absence of exothermic runaway risks associated with high-energy reactions. The mild conditions allow for the use of standard stainless steel or glass-lined reactors without the need for specialized pressure ratings, facilitating rapid capacity expansion. From an environmental standpoint, the process generates minimal hazardous waste, and the use of chloroform, while requiring careful handling, is a well-established solvent with established recovery and recycling protocols in modern chemical plants. This alignment with green chemistry principles simplifies regulatory permitting and environmental auditing, ensuring long-term operational compliance and reducing the administrative burden on EHS teams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiazolidine synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the process capabilities and limitations. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is DABCO preferred over traditional metal catalysts for this thiazolidine synthesis?
A: DABCO (triethylenediamine) serves as an efficient organocatalyst that eliminates the need for expensive transition metals, thereby simplifying downstream purification and reducing heavy metal contamination risks in the final API intermediate.
Q: What are the critical reaction conditions to maintain high yield in this process?
A: Maintaining room temperature is crucial; increasing the temperature to 50°C significantly decreases yield due to product decomposition, while lowering it to 0°C drastically extends reaction time without improving conversion efficiency.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the use of commercially available raw materials, mild ambient conditions, and simple workup procedures makes this route highly amenable to large-scale commercial manufacturing without requiring specialized high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy-2-Methyl-3-(Benzenesulfonyl)Thiazolidine-2-Carboxylic Acid Methyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality heterocyclic intermediates to drive innovation in drug discovery and agrochemical development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials regardless of their project phase. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thiazolidine derivative we produce adheres to the highest international standards for pharmaceutical and fine chemical applications.
We invite forward-thinking organizations to collaborate with us to leverage this advanced synthetic technology for their specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your overall budget. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in complex chemical synthesis and supply chain solutions.
