Scalable Synthesis of High-Purity Curcumin Analogues via Metal-Free Ozonolysis and Recrystallization
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for synthesizing bioactive compounds. Patent CN112374976B introduces a groundbreaking methodology for the preparation of a specific curcumin analogue, chemically defined as (1E,4E)-1,5-bis(2-methoxyphenyl)pent-1,4-dien-3-one. This compound, structurally represented below, holds significant potential in medicinal chemistry due to its pharmacological properties similar to natural curcumin but with enhanced synthetic accessibility. The patent outlines a novel three-step synthetic strategy that fundamentally shifts away from traditional extraction methods or complex catalytic systems, offering a streamlined approach that prioritizes operational simplicity and high purity.
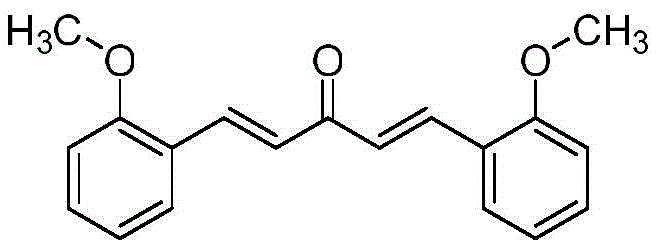
Unlike previous methods that rely on hazardous reagents or difficult purification techniques, this invention leverages ozonolysis and precise recrystallization protocols to achieve a final product purity exceeding 99%. For R&D directors and procurement managers alike, this represents a critical advancement in reliable curcumin analogue supplier capabilities, ensuring that the material can be produced consistently without the bottlenecks associated with silica gel column chromatography or the safety risks of pyrophoric metals. The following analysis dissects the technical merits and commercial implications of this patented technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of curcumin and its derivatives has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Prior art, such as US20060276536, relied on diethyl ether and hexane mixtures for recrystallization; however, diethyl ether is a controlled substance in many jurisdictions due to its high flammability and explosion risk, making it unsuitable for mass production environments. Furthermore, other existing methods, like those described in WO2011029359, utilize metallic sodium dissolved in methanol as a base, introducing severe safety hazards regarding water sensitivity and potential explosions. Beyond safety, the purification processes in these conventional routes often necessitate silica gel column chromatography, which generates substantial amounts of solid waste and solvent consumption, drastically inflating production costs and complicating waste management logistics for supply chain heads.

Moreover, comparative data within the patent highlights the inefficiency of alternative synthetic routes. For instance, Comparative Example 1 demonstrates a pathway involving thioester formation using DCC and DMAP, followed by reduction with triethylsilane and Pd/C. This multi-step sequence not only involves expensive precious metal catalysts but also resulted in a dismal yield of only 18.2% for the intermediate aldehyde, requiring rigorous column chromatography purification. Such low efficiency and high material intensity render these traditional methods economically unviable for cost reduction in pharmaceutical intermediate manufacturing, creating a clear demand for a more direct and waste-minimized approach.
The Novel Approach
The methodology disclosed in CN112374976B presents a paradigm shift by employing a concise three-step sequence that eliminates the need for precious metal catalysts and halogenated solvents. As illustrated in the reaction scheme below, the process begins with the ozonolysis of o-hydroxytrans-cinnamic acid to generate salicylaldehyde, followed by a methylation step to produce 2-methoxybenzaldehyde, and concludes with a Claisen-Schmidt condensation with acetone. This route is designed for industrial feasibility, utilizing common solvents like acetonitrile and methanol, and avoiding the use of controlled or highly hazardous reagents. The strategic choice of reagents ensures that the reaction process is easy to operate, significantly lowering the barrier for commercial scale-up of complex pharmaceutical intermediates.
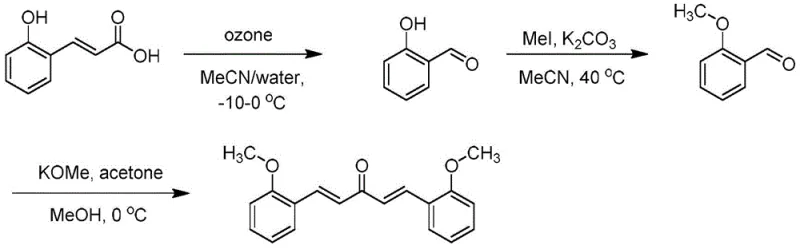
Crucially, this novel approach integrates purification strategies directly into the reaction design, bypassing the need for silica gel column chromatography entirely. The intermediates are purified via vacuum distillation and simple extraction, while the final product is isolated through recrystallization from methanol, yielding crystalline solids with purity greater than 99%. This simplification of downstream processing not only reduces the generation of solid waste and liquid effluents but also shortens the overall production cycle time. For supply chain professionals, this translates to enhanced supply chain reliability and the ability to respond rapidly to market demands without the delays typically associated with complex purification workflows.
Mechanistic Insights into Ozonolysis and Aldol Condensation
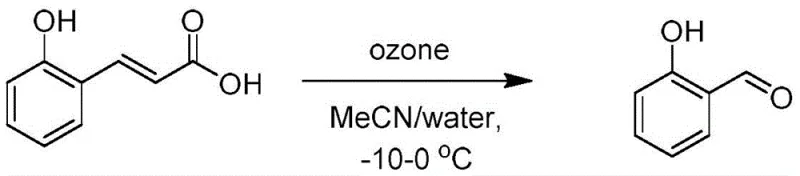
The cornerstone of this synthetic strategy is the initial ozonolysis step, which serves as a clean and efficient oxidative cleavage method. In this transformation, o-hydroxytrans-cinnamic acid is subjected to ozone gas in a mixed solvent system of acetonitrile and water at temperatures between -10 and 0°C. This specific condition allows for the selective cleavage of the carbon-carbon double bond to form salicylaldehyde without over-oxidation to the carboxylic acid, a common side reaction in less controlled oxidative environments. The use of an ice-salt bath ensures precise thermal control, which is vital for managing the exothermic nature of ozonolysis and preventing the decomposition of the unstable ozonide intermediates. Following the reaction, the mixture is sparged with oxygen to decompose residual ozonides, and the product is isolated via vacuum distillation, a technique that offers superior separation efficiency compared to extraction alone.
Subsequent steps leverage well-established organic transformations optimized for high yield and minimal impurity formation. The methylation of salicylaldehyde utilizes methyl iodide and potassium carbonate in acetonitrile at 40°C, achieving yields of approximately 90% when the molar ratio is optimized to 1:3:3. This SN2 reaction is robust and scalable, producing 2-methoxybenzaldehyde which can be used directly in the next step after simple aqueous workup. The final condensation with acetone, catalyzed by potassium methoxide in methanol, proceeds through a classic aldol mechanism followed by dehydration to form the conjugated enone system. The stereochemistry is controlled to favor the (1E,4E) isomer, and the use of methanol as both solvent and recrystallization medium ensures that the final crystal lattice is formed with high fidelity, effectively excluding structural impurities and isomers.
How to Synthesize (1E,4E)-1,5-bis(2-methoxyphenyl)pent-1,4-dien-3-one Efficiently
The synthesis of this high-value curcumin analogue is achieved through a meticulously optimized three-step protocol that balances reaction kinetics with practical work-up procedures. The process begins with the low-temperature ozonolysis of the cinnamic acid precursor, followed by a room-temperature methylation, and concludes with a base-mediated condensation. Each step has been refined to maximize yield while minimizing the need for complex purification equipment, making it ideal for facilities aiming for reducing lead time for high-purity pharmaceutical intermediates. The detailed standardized synthesis steps, including specific reagent quantities, temperature profiles, and isolation techniques, are outlined in the guide below to ensure reproducibility and quality control.
- Perform ozonolysis on o-hydroxytrans-cinnamic acid in a MeCN/water mixture at -10 to 0°C to yield salicylaldehyde, followed by vacuum distillation.
- Conduct methylation of salicylaldehyde using methyl iodide and potassium carbonate in acetonitrile at 40°C to obtain 2-methoxybenzaldehyde.
- React 2-methoxybenzaldehyde with acetone using potassium methoxide as a base in methanol, followed by recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the technological innovations embedded in this patent address several critical pain points traditionally associated with the manufacturing of fine chemical intermediates. By eliminating the reliance on precious metal catalysts such as palladium and avoiding the use of chromatography columns, the process inherently lowers the raw material costs and operational expenditures. The removal of expensive catalytic systems means that procurement teams can source more abundant and cost-effective reagents, while the simplified purification workflow reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in waste disposal and solvent recovery. This economic efficiency is further compounded by the high yield and purity of the final product, which minimizes the loss of valuable material during production.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts like Pd/C and expensive coupling reagents such as DCC drastically reduces the bill of materials for each batch. Furthermore, the avoidance of silica gel column chromatography removes a significant cost center related to stationary phase materials and the large volumes of solvents required for elution. This streamlined approach allows for a more lean manufacturing process where resources are focused on conversion rather than purification, resulting in a significantly lower cost of goods sold (COGS) for the final active ingredient.
- Enhanced Supply Chain Reliability: The use of stable, non-hazardous reagents like potassium carbonate and potassium methoxide, as opposed to dangerous metallic sodium or controlled ethers, ensures that the supply chain is not vulnerable to regulatory restrictions or safety-related shutdowns. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring specialized high-pressure or cryogenic setups beyond standard chillers, enhances the continuity of supply. This reliability is crucial for maintaining consistent inventory levels and meeting the stringent delivery schedules demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, generating minimal solid waste and avoiding halogenated solvents that pose environmental and health risks. The ability to purify intermediates via distillation and the final product via recrystallization makes the process highly scalable from pilot plant to full commercial production without the engineering challenges associated with scaling up chromatographic separations. This environmental compliance not only reduces the regulatory burden but also aligns with the sustainability goals of modern chemical enterprises, facilitating easier permitting and community acceptance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this curcumin analogue. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and quality standards. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own drug development pipelines or manufacturing processes.
Q: Why is ozonolysis preferred over traditional oxidants for this synthesis?
A: Ozonolysis eliminates the need for expensive and toxic heavy metal oxidants, significantly reducing environmental pollution and simplifying waste treatment protocols compared to traditional methods.
Q: How is high purity (>99%) achieved without silica gel chromatography?
A: The process utilizes vacuum distillation for intermediates and a specific methanol recrystallization technique for the final product, effectively removing impurities while avoiding the high costs and waste associated with column chromatography.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method avoids dangerous reagents like metallic sodium and管制 solvents like diethyl ether, utilizing stable conditions and simple work-up procedures that are highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Curcumin Analogue Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN112374976B for producing high-quality curcumin analogues. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle ozonolysis reactions safely and efficiently, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the >99% purity benchmark required for pharmaceutical applications. We are committed to delivering excellence in every gram we produce.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free and chromatography-free route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of this critical pharmaceutical intermediate.
