Advanced Synthesis of Anti-Influenza Quinoline Derivatives for Commercial Scale-Up
Advanced Synthesis of Anti-Influenza Quinoline Derivatives for Commercial Scale-Up
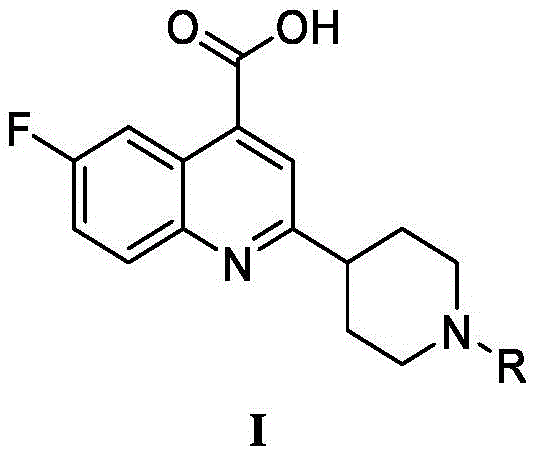
The global pharmaceutical landscape is constantly evolving in response to emerging viral threats, particularly influenza strains such as H1N1. Patent CN114105947A introduces a significant breakthrough in the field of antiviral drug development by disclosing a novel class of quinoline derivatives characterized by a 4-carboxylic acid-quinoline ring framework. These compounds exhibit potent inhibitory activity against the influenza A/WSN/33 virus, addressing the critical need for new therapeutic agents beyond existing neuraminidase inhibitors. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a robust platform for developing next-generation anti-influenza medications with improved efficacy profiles.
The core innovation lies not only in the biological activity of the final compounds but also in the elegance and efficiency of the synthetic methodology employed. Unlike traditional routes that often rely on harsh conditions or precious metal catalysts, this patent outlines a streamlined approach utilizing readily available starting materials. The structural novelty of these derivatives, specifically the substitution patterns at the 2-position and 6-position of the quinoline nucleus, allows for extensive structure-activity relationship (SAR) exploration. This flexibility is crucial for optimizing pharmacokinetic properties and ensuring a strong pipeline of candidate molecules for clinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized quinoline carboxylic acids has been plagued by several inherent challenges that impact both cost and environmental sustainability. Conventional methodologies frequently involve multi-step sequences requiring stringent anhydrous conditions, cryogenic temperatures, or the use of toxic organic solvents that are difficult to recover. Furthermore, many established routes depend on transition metal catalysis, which introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing these trace metals to meet regulatory standards often necessitates additional purification steps, such as specialized scavenging resins or repeated recrystallizations, which drastically reduce overall yield and increase production lead times.
Another significant bottleneck in traditional quinoline synthesis is the handling of reactive intermediates that may be unstable or hazardous on a large scale. Processes involving strong acids or bases in non-aqueous media can lead to safety concerns regarding exothermic reactions and waste disposal. The generation of substantial amounts of saline waste and the inability to recycle solvents efficiently contribute to a high environmental footprint. For supply chain managers, these factors translate into volatile pricing and potential disruptions in the availability of key intermediates, making long-term planning for commercial manufacturing increasingly difficult.
The Novel Approach
The methodology described in patent CN114105947A offers a transformative solution to these longstanding issues by employing a direct condensation reaction in an aqueous alkaline environment. This novel approach utilizes 5-fluoroisatin (Compound II) and a substituted acetyl-piperidine derivative (Compound III) as the primary building blocks. By conducting the reaction in a 10% aqueous alkali solution under reflux conditions, the process eliminates the need for expensive anhydrous solvents and complex inert atmosphere setups. The simplicity of the reaction conditions allows for a significant reduction in energy consumption and operational complexity, directly addressing the need for cost reduction in pharmaceutical intermediate manufacturing.
Moreover, the workup procedure is remarkably straightforward, involving simple acidification to precipitate the product, followed by washing and drying. This avoids the need for column chromatography or extensive solvent extraction, which are typically resource-intensive. The ability to recycle organic solvents used in the preparation of the intermediate Compound III further enhances the green chemistry profile of this route. For manufacturers aiming for commercial scale-up of complex pharmaceutical intermediates, this process offers a clear pathway to high-volume production with minimal waste generation and superior safety characteristics compared to legacy methods.
Mechanistic Insights into Base-Catalyzed Condensation
The chemical transformation at the heart of this technology is a base-catalyzed condensation that constructs the quinoline ring system while simultaneously installing the critical carboxylic acid functionality. The reaction mechanism likely proceeds through the deprotonation of the alpha-carbon of the acetyl group in Compound III by the hydroxide ions present in the alkaline solution. This generates a nucleophilic enolate species that attacks the carbonyl carbon of the isatin derivative (Compound II). Subsequent intramolecular cyclization and dehydration steps lead to the formation of the aromatic quinoline core. The use of a strong base, such as potassium hydroxide or sodium hydroxide, in a molar ratio of 1:3 to 1:5 relative to Compound II ensures complete conversion and drives the equilibrium towards the desired product.

Understanding the kinetics of this reaction is vital for process optimization. The patent specifies a reaction time of 40 to 50 hours under reflux, indicating that while the thermodynamics favor the product, the kinetic barrier requires sustained thermal energy to overcome. This extended reaction time allows for the gradual consumption of starting materials, minimizing the formation of side products that could arise from rapid, uncontrolled reactions. The choice of base is also flexible, with options including lithium hydroxide, sodium hydride, and alkoxides, providing manufacturers with the ability to tailor the process based on local supply chain availability and cost considerations without compromising the integrity of the final molecular structure.
Purity control is inherently built into this mechanistic pathway. The precipitation of the final product upon acidification to pH 1 acts as a self-purification step, as many organic impurities remain soluble in the acidic aqueous medium or are removed during the subsequent washing stages. The high HPLC purity values reported (often exceeding 99.8%) suggest that the reaction is highly selective. This selectivity reduces the burden on downstream processing teams, allowing them to focus on crystallization optimization rather than impurity profiling and removal. For R&D teams, this means faster turnaround times for generating high-quality batches for biological testing and stability studies.
How to Synthesize Quinoline Derivatives Efficiently
The synthesis of these high-value antiviral intermediates is divided into two distinct stages: the preparation of the piperidine-containing ketone intermediate (Compound III) and the final condensation to form the quinoline scaffold (Compound I). The first stage involves the alkylation of 1-(piperidin-4-yl)ethanone (Compound V) with various aryl halides (Compound IV) in the presence of a base and an organic solvent. This step allows for the diversification of the R-group on the piperidine nitrogen, enabling the creation of a library of analogues with varying lipophilicity and electronic properties. The second stage brings together the diversified intermediate with the fluorinated isatin core to complete the molecular architecture.
- Prepare intermediate Compound III by reacting 1-(piperidin-4-yl)ethanone (Compound V) with various aryl halides (Compound IV) in organic solvent with base.
- Dissolve 5-fluoroisatin (Compound II) and intermediate Compound III in 10% aqueous alkali solution.
- Reflux the mixture for 40-50 hours, cool, acidify to pH 1, filter, wash, and dry to obtain the target quinoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages for procurement managers and supply chain heads focused on efficiency and reliability. The primary driver for cost optimization is the elimination of precious metal catalysts and the reliance on commodity chemicals like aqueous alkalis and common organic solvents. This shift significantly lowers the raw material cost per kilogram of the final intermediate. Additionally, the simplified workup procedure reduces the consumption of auxiliary materials such as silica gel or specialized filtration media, further contributing to overall cost reduction in API manufacturing. The robustness of the process also implies lower rates of batch failure, ensuring a more predictable cost structure.
Supply chain reliability is enhanced by the use of widely available starting materials. Compound V and the various aryl halides (Compound IV) are commercially sourced or easily synthesized from bulk chemicals, reducing dependency on single-source suppliers for exotic reagents. The ability to use a variety of bases (KOH, NaOH, K2CO3) provides flexibility; if the price of one reagent spikes due to market volatility, the process can be adjusted to use an alternative without requiring a complete re-validation of the synthetic route. This adaptability is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates in a fluctuating global market.
- Cost Reduction in Manufacturing: The process replaces expensive catalytic systems with inexpensive aqueous bases, drastically cutting reagent costs. The avoidance of chromatographic purification in favor of simple precipitation and filtration reduces labor and material expenses associated with downstream processing. Furthermore, the potential for solvent recovery and reuse in the intermediate synthesis step minimizes waste disposal fees and raw material procurement costs, leading to substantial overall savings.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals and avoiding specialized catalysts, the risk of supply disruption is minimized. The flexibility in base selection allows procurement teams to source materials locally, reducing logistics lead times and import dependencies. The high yield and purity consistency ensure that production schedules can be met reliably, supporting continuous manufacturing operations without unexpected delays caused by quality failures or reprocessing.
- Scalability and Environmental Compliance: The use of water as the primary reaction medium for the final step aligns with green chemistry principles, simplifying environmental permitting and waste treatment. The process generates less hazardous waste compared to traditional organic synthesis routes, lowering compliance costs. The straightforward scale-up from gram to kilogram scales, evidenced by the patent examples, demonstrates that the technology is ready for immediate industrial implementation without significant engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these quinoline derivatives. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis route?
A: The process utilizes inexpensive aqueous alkali solutions instead of expensive transition metal catalysts, significantly reducing raw material costs and simplifying purification.
Q: What is the purity profile of the final compounds?
A: The patented method consistently yields products with HPLC purity exceeding 99.8%, minimizing the need for complex chromatographic purification steps.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of recyclable solvents and simple workup procedures (acidification and filtration) makes the process highly scalable and environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivatives Supplier
As the demand for effective antiviral agents continues to grow, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from pilot plant to full-scale manufacturing. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of quinoline derivatives meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with our technical team to explore the full potential of this synthetic route for your specific antiviral programs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of how implementing this process can optimize your budget. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your project requirements, ensuring a successful and efficient path to market.
