Advanced Synthesis of Sulfonic Acid-Containing Sulfur Ylides for Commercial Epoxidation
The chemical industry is constantly seeking safer, more efficient pathways for synthesizing critical intermediates, particularly for the pharmaceutical and agrochemical sectors. Patent CN111116441B introduces a groundbreaking synthetic method for sulfur ylides containing sulfonic acid groups, addressing long-standing challenges in epoxidation chemistry. This technology replaces hazardous, volatile reagents like dimethyl sulfide with a robust, water-soluble sulfonate system that can be efficiently recycled. By integrating a unique recovery loop, the process not only mitigates environmental risks associated with toxic sulfur compounds but also drastically simplifies downstream purification. For manufacturers of high-purity oxirane derivatives, this represents a significant leap forward in process safety and economic viability. The ability to recover the sulfonate backbone transforms what was once a linear, waste-generating process into a circular, sustainable manufacturing cycle. This report analyzes the technical depth and commercial potential of this innovation for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of epoxy compounds via sulfur ylides has relied heavily on dimethyl sulfide or dimethyl sulfoxide as primary precursors. These traditional reagents present severe operational hazards; dimethyl sulfide is a toxic substance with a low boiling point and a notoriously foul odor, posing significant risks to worker safety and requiring expensive containment infrastructure. While some alternatives like p-propoxyanisole sulfide have been explored to replace dimethyl sulfide, they introduce new processing bottlenecks. Specifically, the byproduct generated alongside the epoxide often possesses a boiling point extremely close to that of the target product, rendering standard distillation techniques ineffective for separation. This inability to cleanly separate the product from the sulfur byproduct results in low purity, poor yields, and the generation of substantial chemical waste. Consequently, the economic value of these conventional routes is diminished by high energy consumption and the inability to recycle the expensive sulfur-containing reagents.
The Novel Approach
The methodology described in CN111116441B fundamentally alters this landscape by introducing a sulfonate-based sulfur ylide system. Instead of volatile liquids, the process utilizes a solid sulfonate salt, specifically sodium 4-(4-(methylthio)phenoxy)butane-1-sulfonate, which exhibits excellent water solubility and low irritation. This physical property difference is the key to the process's success; after the epoxidation reaction, the mixture can be treated with water to dissolve the spent sulfonate salt while the organic epoxide remains in the organic phase. This allows for a simple liquid-liquid extraction or solid-liquid separation, completely bypassing the difficult distillation steps required by older methods. Furthermore, the aqueous layer containing the recovered sulfonate can be concentrated and reused to generate fresh ylide, creating a closed-loop system. This approach not only enhances the purity of the final epoxide but also significantly lowers the cost of goods sold by minimizing raw material loss.
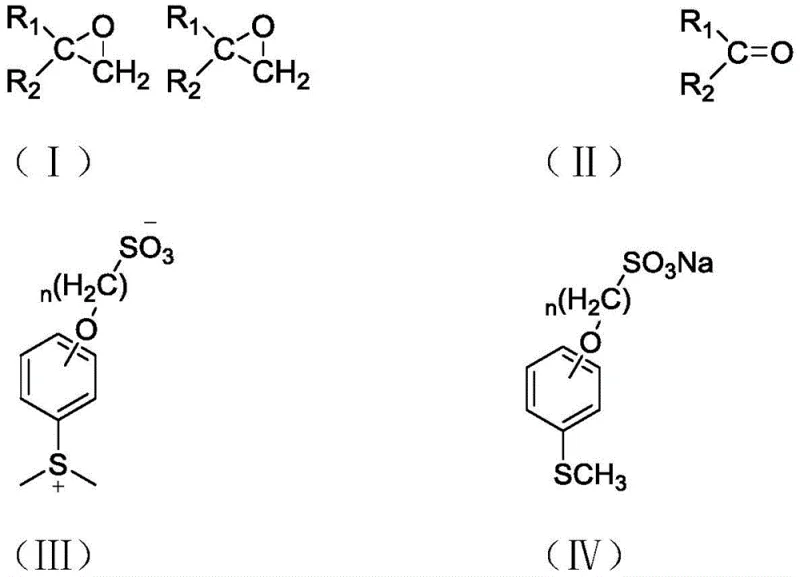
Mechanistic Insights into Sulfonate-Based Ylide Formation and Epoxidation
The core of this technology lies in the precise manipulation of sulfur oxidation states and nucleophilic properties. The synthesis begins with a nucleophilic substitution reaction where anhydrous sodium sulfite attacks 1-(4-bromobutoxy)-4-(methylthio)benzene. This step, typically conducted at 80°C in a mixed solvent system of water and ethanol, installs the critical sulfonic acid group onto the aromatic scaffold. The resulting sulfonate salt serves as a stable precursor that can be stored and handled safely. In the subsequent methylation step, this salt is heated to temperatures between 220°C and 260°C in the presence of a methylating agent such as dimethyl sulfate. Under these thermal conditions, the sulfur atom undergoes quaternization to form the reactive sulfonium salt, which acts as the sulfur ylide precursor. The high temperature ensures complete conversion while the nitrogen atmosphere prevents oxidative degradation of the sensitive sulfur species.
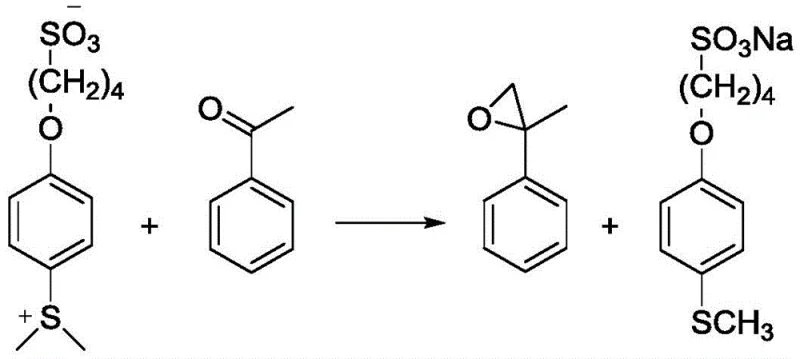
Once the ylide is generated, it reacts with carbonyl compounds, such as acetophenone, in the presence of a strong base like sodium hydroxide or potassium carbonate. The base deprotonates the alpha-carbon adjacent to the sulfonium center, generating the active ylide species in situ. This nucleophilic carbon then attacks the carbonyl carbon of the ketone, forming a betaine intermediate that cyclizes to release the epoxide and regenerate the sulfide. Crucially, because the sulfide moiety is tethered to a sulfonate group, the byproduct immediately reverts to the water-soluble sulfonate salt upon workup. This mechanistic feature ensures that the sulfur byproduct does not contaminate the organic product stream. The reaction conditions are mild, typically proceeding at 40-60°C, which preserves the integrity of sensitive functional groups on the carbonyl substrate. This controlled reactivity profile makes the method highly suitable for synthesizing complex pharmaceutical intermediates where functional group tolerance is paramount.
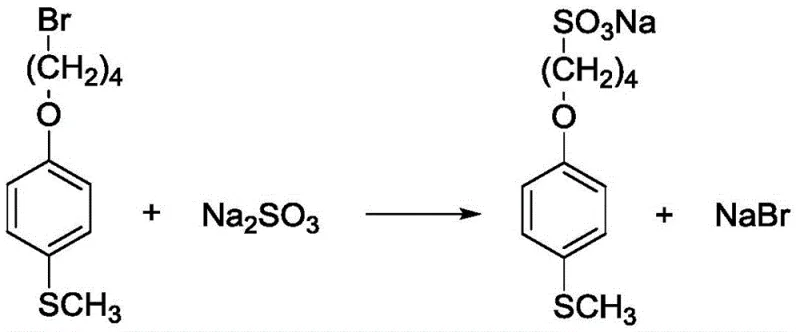
How to Synthesize Sodium 4-(4-(methylthio)phenoxy)butane-1-sulfonate Efficiently
The preparation of the key sulfonate precursor is the foundational step for the entire process, requiring careful control of stoichiometry and temperature to maximize yield. The patent outlines a robust protocol involving the reflux of bromo-precursors with sulfite salts, followed by purification via recrystallization from ethanol. This ensures that the starting material for the ylide generation is of high purity, which is critical for preventing side reactions during the high-temperature methylation step. Operators must adhere strictly to the specified molar ratios, typically maintaining a slight excess of sulfite to drive the substitution to completion.
- Synthesize the sulfonate precursor (Formula IV) by reacting 1-(4-bromobutoxy)-4-(methylthio)benzene with anhydrous sodium sulfite at 80°C.
- Convert the sulfonate salt into the sulfur ylide (Formula III) via methylation using dimethyl sulfate at 220-260°C under nitrogen protection.
- Perform the epoxidation reaction by mixing the ylide with a carbonyl compound and base in a solvent at 40-60°C, followed by aqueous workup to recover the sulfonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sulfonate-based ylide technology offers tangible strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the purification workflow, which directly translates to reduced operational expenditures. By eliminating the need for high-vacuum fractional distillation to separate close-boiling sulfur byproducts, manufacturers can significantly lower energy consumption and equipment maintenance costs. Additionally, the ability to recycle the sulfonate backbone means that the effective consumption of sulfur-containing raw materials is minimized, insulating the production process from volatility in sulfur commodity prices. This creates a more predictable cost structure for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of complex separation units and the recycling of the sulfonate salt lead to substantial cost savings in fine chemical manufacturing. Traditional processes often lose expensive sulfur reagents in the waste stream or require energy-intensive purification to remove them; this new method retains the sulfur value within the process loop. By recovering the solid sulfonate from the aqueous layer via simple distillation, the facility reduces its net raw material intake. This circular economy approach lowers the variable cost per kilogram of the final epoxide, enhancing margin potential without compromising quality standards.
- Enhanced Supply Chain Reliability: Replacing toxic, volatile dimethyl sulfide with a stable, solid sulfonate salt significantly improves supply chain security and safety compliance. Hazardous reagents often face strict transportation regulations and storage limitations that can disrupt logistics; solid sulfonates are easier to ship and store in bulk quantities. Furthermore, the reduced toxicity profile lowers the barrier for regulatory approval in various jurisdictions, ensuring smoother market access. This stability allows for larger batch sizes and longer campaign runs, reducing the frequency of changeovers and improving overall equipment effectiveness.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations like filtration and extraction rather than specialized distillation columns. The aqueous waste stream is primarily composed of inorganic salts and recovered sulfonate, which is far easier to treat than organic sulfur waste. This aligns with increasingly stringent environmental regulations regarding sulfur emissions and wastewater treatment. The ability to operate at moderate temperatures during the epoxidation step also reduces the thermal load on plant utilities, facilitating easier scale-up from pilot to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfur ylide technology. These insights are derived directly from the experimental data and claims within the patent documentation to ensure accuracy. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their specific intermediate portfolios.
Q: How does this new sulfur ylide method improve upon traditional dimethyl sulfide processes?
A: Traditional methods using dimethyl sulfide involve toxic, volatile, and foul-smelling reagents that are difficult to handle safely. Furthermore, alternative aromatic sulfides often produce byproducts with boiling points too close to the desired epoxide, making separation economically unviable. This new method utilizes a water-soluble sulfonate backbone, allowing for easy solid-liquid separation and recycling of the starting material, thereby eliminating complex distillation steps.
Q: What are the optimal reaction conditions for the methylation step?
A: According to the patent data, the methylation of the sulfonate salt to form the sulfur ylide is best conducted at temperatures between 220°C and 260°C. The reaction typically proceeds for 1 to 6 hours under a nitrogen atmosphere to prevent oxidation, utilizing methylating agents such as dimethyl sulfate or methyl iodide.
Q: Can the sulfonate byproduct be reused in subsequent batches?
A: Yes, a key advantage of this technology is the recyclability of the sulfonate. After the epoxidation reaction, the aqueous layer containing the regenerated sulfonate salt can be separated, concentrated via reduced pressure distillation, and directly reused for the methylation step, significantly reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur Ylide Supplier
As the demand for high-purity epoxide intermediates grows in the pharmaceutical sector, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless. Our rigorous QC labs and stringent purity specifications guarantee that every batch of sulfur ylide or epoxide derivative meets the exacting standards required for API synthesis. We understand the critical nature of supply continuity and have optimized our processes to deliver consistent quality regardless of batch size.
We invite you to discuss how this innovative sulfonate-based technology can be integrated into your manufacturing strategy to achieve superior cost efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate the viability of this advanced synthetic method for your supply chain.
