Advanced Base-Catalyzed Synthesis of 2H-Pyran-2-One Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access biologically active scaffolds, particularly the versatile 2H-pyran-2-one core found in numerous antiviral and anticancer agents. Patent CN111646964B introduces a groundbreaking base-catalyzed methodology that addresses long-standing challenges in constructing this privileged structure. This innovation utilizes sulfur ylides as nucleophiles reacting with cyclopropenones to form carbon-carbon bonds efficiently, followed by a seamless cyclization step. Unlike conventional approaches that often suffer from low yields and cumbersome purification, this novel route offers a direct, one-pot solution that significantly streamlines the synthetic workflow. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2H-pyran-2-one derivatives has been plagued by significant technical hurdles that impact both cost and timeline. Traditional methods frequently rely on multi-step sequences involving sensitive reagents, strict anhydrous conditions, or expensive transition metal catalysts that are difficult to remove to ppm levels required for API production. These legacy processes often exhibit poor atom economy, generating substantial chemical waste that complicates environmental compliance and disposal logistics. Furthermore, the harsh reaction conditions typically employed can lead to the degradation of sensitive functional groups, limiting the scope of substrates that can be utilized and restricting the chemical diversity of the final library. Such inefficiencies create bottlenecks in the supply chain, resulting in extended lead times and inflated production costs that erode profit margins for downstream manufacturers.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by leveraging the unique reactivity of strained small rings, specifically cyclopropenones, in conjunction with stable sulfur ylides. This approach bypasses the need for precious metal catalysts, relying instead on inexpensive and readily available inorganic bases such as sodium acetate or cesium carbonate to drive the reaction forward. The process operates under relatively mild thermal conditions, typically around 100°C, which reduces energy consumption and minimizes the risk of thermal decomposition of the product. By consolidating bond formation and ring closure into a single operational step, this strategy drastically reduces the number of unit operations required, thereby enhancing overall throughput. This simplification not only accelerates the development timeline but also provides a robust platform for the rapid generation of diverse analogues for structure-activity relationship studies.
Mechanistic Insights into Base-Catalyzed Cyclization
The core of this transformation lies in the nucleophilic attack of the sulfur ylide on the electrophilic carbonyl carbon of the cyclopropenone, initiating a cascade that ultimately yields the six-membered lactone ring. The base plays a critical role in generating the active ylide species in situ or facilitating the deprotonation steps necessary for the cyclization to proceed. This mechanism ensures high regioselectivity and minimizes the formation of unwanted byproducts, which is crucial for maintaining high purity standards in pharmaceutical manufacturing. The reaction pathway is designed to maximize atom utilization, ensuring that the majority of the starting material mass is incorporated into the final product structure rather than being lost as waste.
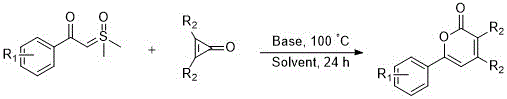
From an impurity control perspective, the use of simple inorganic bases and common organic solvents like 1,2-dichloroethane allows for straightforward workup procedures. The absence of heavy metals eliminates the need for complex scavenging steps, which are often sources of yield loss and contamination. The reaction conditions are tolerant to various electronic environments on the aromatic rings, as evidenced by the successful synthesis of derivatives bearing electron-donating methyl groups and electron-withdrawing chloro substituents. This robustness suggests that the mechanism is resilient against minor fluctuations in reaction parameters, making it highly suitable for transfer from laboratory scale to commercial production environments where consistency is paramount.
How to Synthesize 2H-Pyran-2-One Derivatives Efficiently
To implement this synthesis effectively, operators should follow a standardized protocol that ensures reproducibility and safety. The process begins with the precise weighing of the sulfur ylide and cyclopropenone precursors, followed by the addition of the chosen base and solvent into a clean, dry reactor. The mixture is then subjected to heating and stirring for a defined period to allow the reaction to reach completion. Detailed standard operating procedures regarding temperature ramping, stirring speeds, and quenching methods are essential for maintaining batch-to-batch consistency. The standardized synthesis steps outlined below provide a foundational guide for process engineers to adapt this chemistry for their specific facility capabilities.
- Combine sulfur ylide, cyclopropenone, base (e.g., sodium acetate), and solvent (e.g., 1,2-dichloroethane) in a reactor.
- Heat the mixture to 100°C and stir continuously for 24 hours to facilitate the insertion reaction and cyclization.
- Upon completion, concentrate the reaction mixture and purify the residue via silica gel column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this base-catalyzed route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio; sulfur ylides and cyclopropenones are commercially accessible or easily synthesized from commodity chemicals, reducing dependency on exotic or single-source suppliers. This diversification of the supply base mitigates the risk of shortages and price volatility, ensuring a more stable flow of materials for continuous manufacturing operations. Additionally, the elimination of expensive catalysts and the reduction in processing steps directly translate to lower variable costs per kilogram of product produced.
- Cost Reduction in Manufacturing: The economic profile of this process is significantly improved by removing the need for costly transition metal catalysts and the associated purification technologies required to meet residual metal specifications. By utilizing inexpensive inorganic bases and common solvents, the direct material costs are minimized, while the simplified workup reduces labor and utility expenses. This lean manufacturing approach allows for substantial cost savings that can be passed down the value chain or reinvested into further R&D initiatives, enhancing the overall competitiveness of the final pharmaceutical product in the marketplace.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher reliability in supply delivery. Since the reagents are stable and the reaction tolerates a range of conditions, the risk of batch failures due to minor deviations is reduced. This reliability ensures that production schedules can be met consistently, preventing delays in the downstream formulation of finished drugs. Furthermore, the use of standard solvents like 1,2-dichloroethane means that solvent recovery and recycling systems already present in most chemical plants can be utilized, further securing the supply chain against external market fluctuations.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the absence of hazardous reagents and the use of standard thermal conditions. The reaction does not generate excessive gaseous byproducts or toxic waste streams, simplifying the environmental permitting process and reducing the burden on waste treatment facilities. This alignment with green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major global pharmaceutical companies focused on reducing their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the fit of this technology within existing manufacturing portfolios and for planning future capacity expansions effectively.
Q: What are the primary advantages of this base-catalyzed method over traditional synthesis routes?
A: This method eliminates the need for complex multi-step sequences and harsh reaction conditions often associated with traditional pyranone synthesis. By utilizing readily available sulfur ylides and cyclopropenones with simple inorganic bases, it achieves high atom economy and simplifies the purification process, leading to significant operational efficiencies.
Q: Is this synthetic route suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process is highly scalable due to the use of common solvents like 1,2-dichloroethane and stable reagents that do not require specialized handling equipment. The reaction operates at a moderate temperature of 100°C, which is easily manageable in standard industrial reactors, ensuring robust supply chain continuity.
Q: What types of substituents are tolerated in this cyclization reaction?
A: The methodology demonstrates excellent functional group tolerance, accommodating various substituents on both the sulfur ylide and the cyclopropenone rings. Examples include methyl and chloro groups on the phenyl rings, indicating broad substrate compatibility for generating diverse libraries of bioactive intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2H-Pyran-2-One Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this base-catalyzed synthesis for the production of high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent and thermal requirements of this process, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for clinical and commercial applications. We are committed to delivering excellence in every kilogram we produce.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can optimize your supply chain for 2H-pyran-2-one derivatives.
