Revolutionizing Beta-Nitrostyrene Production with Green Ethanolamine-Based Ionic Liquid Catalysts
Introduction to Advanced Catalytic Methodologies in Nitroolefin Synthesis
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the production of high-value pharmaceutical intermediates like beta-nitrostyrene. Patent CN103497082A introduces a groundbreaking approach that utilizes novel ethanolamine-based multiple acidic functional ionic liquids as highly efficient catalysts for the Henry condensation reaction. This technology represents a significant departure from conventional protocols that rely heavily on toxic organic solvents and less efficient heterogeneous catalysts. By leveraging the unique physicochemical properties of these task-specific ionic liquids, the process achieves remarkable reaction rates and selectivity under solvent-free conditions. For R&D directors and process chemists, this innovation offers a compelling solution to the persistent challenges of waste generation and catalyst deactivation. The ability to operate under normal pressure heating conditions while maintaining high yields underscores the robustness of this catalytic system. Furthermore, the structural tunability of the ionic liquid allows for precise optimization of acidity, ensuring compatibility with a diverse array of aromatic aldehydes and nitroalkanes. This patent not only addresses immediate synthetic efficiency but also aligns with long-term sustainability goals essential for modern chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for synthesizing beta-nitrostyrene and its derivatives have long been plagued by significant operational and environmental drawbacks that hinder large-scale adoption. Historically, these reactions have relied on heterogeneous catalysts such as polyamine-functionalized zirconia or silica-alumina-supported amines, which often suffer from relatively low catalytic activity and limited substrate scope. A critical issue with these legacy methods is the extensive requirement for toxic and harmful organic solvents to facilitate mass transfer and heat dissipation during the reaction. The reliance on volatile organic compounds (VOCs) not only escalates the cost of raw material procurement but also imposes a heavy burden on waste treatment facilities and regulatory compliance teams. Additionally, many conventional catalysts exhibit poor reusability, leading to frequent replacement cycles that disrupt production continuity and inflate operational expenditures. The separation of products from these solid catalysts can also be cumbersome, often requiring complex filtration and washing steps that reduce overall process efficiency. Consequently, there is an urgent industrial demand for a catalytic system that can eliminate solvent usage while providing superior stability and ease of separation.
The Novel Approach
The methodology disclosed in the patent presents a transformative solution by employing a series of multiple acidic functional ionic liquids derived from inexpensive ethanolamine precursors. Unlike traditional solid supports, these ionic liquids function as both the catalyst and the reaction medium, effectively creating a homogeneous yet separable system that operates without any additional organic solvent. The core of this innovation lies in the unique structure of the catalyst, which integrates multiple acidic sites capable of activating both the nitroalkane and the aromatic aldehyde simultaneously. ![General chemical structure of the ethanolamine-based multiple acidic ionic liquid catalyst [SFHEA]X showing the cationic backbone and variable anionic species](/insights/img/beta-nitrostyrene-ionic-liquid-catalyst-pharma-supplier-20260307105956-01.png) As illustrated in the structural representation, the cationic framework provides a stable scaffold, while the anionic component (X-) can be tuned to modulate acidity and solubility properties. This dual functionality enables the Henry condensation and subsequent dehydration to proceed smoothly at temperatures ranging from 100°C to 130°C. The absence of solvent not only simplifies the reaction setup but also dramatically increases the effective concentration of reactants, driving the equilibrium towards the desired nitroolefin product. This approach effectively resolves the trade-off between catalytic activity and environmental impact, offering a streamlined pathway for the synthesis of complex nitroolefins.
As illustrated in the structural representation, the cationic framework provides a stable scaffold, while the anionic component (X-) can be tuned to modulate acidity and solubility properties. This dual functionality enables the Henry condensation and subsequent dehydration to proceed smoothly at temperatures ranging from 100°C to 130°C. The absence of solvent not only simplifies the reaction setup but also dramatically increases the effective concentration of reactants, driving the equilibrium towards the desired nitroolefin product. This approach effectively resolves the trade-off between catalytic activity and environmental impact, offering a streamlined pathway for the synthesis of complex nitroolefins.
Mechanistic Insights into Ethanolamine-Based Ionic Liquid Catalysis
The exceptional performance of the [SFHEA]X ionic liquid catalyst can be attributed to its sophisticated molecular architecture which facilitates a synergistic activation mechanism during the Henry reaction. The preparation of this catalyst involves a straightforward two-step synthesis starting from 2-ethanolamine and chlorosulfonic acid, followed by an ion exchange process to introduce specific acidic anions. ![Synthesis route of the zwitterionic salt [SFHEA] and subsequent ion exchange to form the acidic functional ionic liquid](/insights/img/beta-nitrostyrene-ionic-liquid-catalyst-pharma-supplier-20260307105956-02.webp) This synthetic accessibility ensures that the catalyst can be produced reliably on a commercial scale using readily available feedstocks. Once formed, the ionic liquid possesses both Brønsted and Lewis acidic characteristics, allowing it to protonate the carbonyl oxygen of the aromatic aldehyde while simultaneously stabilizing the nitronate intermediate formed from the nitroalkane. This bifunctional activation lowers the energy barrier for the carbon-carbon bond formation step, which is typically the rate-determining step in Henry condensations. Moreover, the ionic nature of the catalyst creates a polar environment that favors the elimination of water, driving the dehydration step to completion without the need for external dehydrating agents. The structural integrity of the ionic liquid remains intact throughout the reaction cycle, preventing leaching of active species and ensuring consistent catalytic performance batch after batch.
This synthetic accessibility ensures that the catalyst can be produced reliably on a commercial scale using readily available feedstocks. Once formed, the ionic liquid possesses both Brønsted and Lewis acidic characteristics, allowing it to protonate the carbonyl oxygen of the aromatic aldehyde while simultaneously stabilizing the nitronate intermediate formed from the nitroalkane. This bifunctional activation lowers the energy barrier for the carbon-carbon bond formation step, which is typically the rate-determining step in Henry condensations. Moreover, the ionic nature of the catalyst creates a polar environment that favors the elimination of water, driving the dehydration step to completion without the need for external dehydrating agents. The structural integrity of the ionic liquid remains intact throughout the reaction cycle, preventing leaching of active species and ensuring consistent catalytic performance batch after batch.
A key feature of this catalytic system is the tunability of its acidic properties through the selection of different counter-anions, which directly influences reaction kinetics and selectivity. The patent highlights several viable anionic species, including bisulfate (HSO4-), nitrate (NO3-), trifluoroacetate (CF3COO-), and methanesulfonate (CH3SO3-), each offering distinct electronic environments. 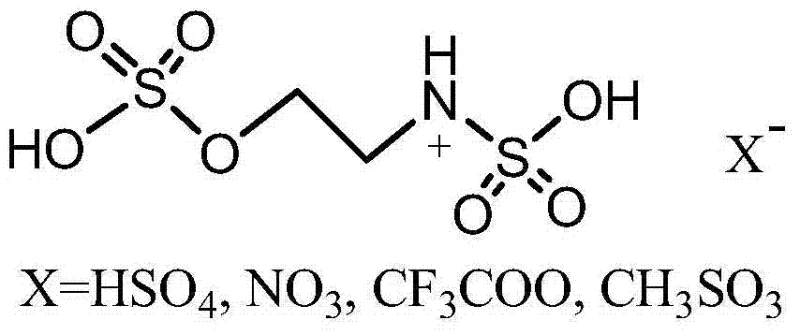 By varying the anion, chemists can fine-tune the acidity strength to match the electronic nature of the specific aromatic aldehyde substrate being used. For instance, electron-deficient aldehydes may require a milder acidic environment to prevent side reactions, while electron-rich substrates might benefit from stronger activation. This level of control is rarely achievable with fixed-structure heterogeneous catalysts. Furthermore, the ionic liquid matrix acts as a protective cage for the reactive intermediates, minimizing polymerization or decomposition pathways that often plague nitroolefin synthesis. The result is a highly selective process that consistently delivers products with purity levels exceeding 96%, as evidenced by the experimental data. This mechanistic robustness provides R&D teams with a versatile tool for optimizing synthetic routes for a wide variety of pharmaceutical building blocks.
By varying the anion, chemists can fine-tune the acidity strength to match the electronic nature of the specific aromatic aldehyde substrate being used. For instance, electron-deficient aldehydes may require a milder acidic environment to prevent side reactions, while electron-rich substrates might benefit from stronger activation. This level of control is rarely achievable with fixed-structure heterogeneous catalysts. Furthermore, the ionic liquid matrix acts as a protective cage for the reactive intermediates, minimizing polymerization or decomposition pathways that often plague nitroolefin synthesis. The result is a highly selective process that consistently delivers products with purity levels exceeding 96%, as evidenced by the experimental data. This mechanistic robustness provides R&D teams with a versatile tool for optimizing synthetic routes for a wide variety of pharmaceutical building blocks.
How to Synthesize Beta-Nitrostyrene Efficiently
Implementing this advanced catalytic protocol in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and catalyst longevity. The process begins with the precise preparation of the [SFHEA]X ionic liquid, ensuring that the ion exchange is complete to avoid contamination with residual salts that could inhibit catalysis. Once the catalyst is ready, the reaction is conducted by simply mixing the nitroalkane and aromatic aldehyde substrates with the ionic liquid in a standard reactor equipped with magnetic stirring. The beauty of this method lies in its simplicity; there is no need for inert gas protection or specialized high-pressure equipment, as the reaction proceeds efficiently under normal atmospheric pressure. Heating the mixture to the optimal temperature range of 100°C to 130°C initiates the condensation, which can be monitored conveniently using thin-layer chromatography (TLC). Upon completion, the product is easily separated by extraction with ethyl acetate, leaving the ionic liquid behind in the aqueous phase for recovery. Detailed standardized synthesis steps see the guide below.
- Prepare the acidic functional ionic liquid catalyst [SFHEA]X by reacting ethanolamine with chlorosulfonic acid followed by ion exchange with specific acids like H2SO4 or HNO3.
- Mix nitroalkane and aromatic aldehyde substrates with the ionic liquid catalyst in a reactor without adding any organic solvent.
- Heat the mixture to 100-130°C under normal pressure for 0.5 to 24 hours, then extract the product with ethyl acetate and recover the catalyst for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-catalyzed process offers substantial strategic benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic reduction of raw material costs associated with organic solvents. By eliminating the need for large volumes of dichloromethane, toluene, or other volatile solvents, manufacturers can significantly lower their expenditure on chemical inputs and reduce the logistical complexity of storing hazardous materials. This solvent-free approach also translates to a leaner waste management profile, as there is no contaminated solvent waste to treat or dispose of, thereby reducing environmental compliance costs. Furthermore, the high stability and reusability of the [SFHEA]X catalyst mean that the frequency of catalyst procurement is minimized. The patent data indicates that the ionic liquid can be recovered and reused multiple times without a noticeable decline in performance, which effectively amortizes the initial cost of the catalyst over many production batches. This reliability ensures a steady production flow and mitigates the risk of supply disruptions caused by catalyst degradation or availability issues.
- Cost Reduction in Manufacturing: The elimination of organic solvents removes a major cost center from the production budget, as solvents often account for a significant portion of the total mass input in traditional batch processes. Additionally, the energy consumption for solvent recovery and distillation is completely eradicated, leading to lower utility bills and a reduced carbon footprint for the manufacturing facility. The high atom economy of the solvent-free reaction further ensures that raw materials are converted into product rather than waste, maximizing the value derived from every kilogram of feedstock purchased. These cumulative efficiencies result in a more competitive cost structure for the final pharmaceutical intermediate, allowing companies to maintain healthy margins even in volatile market conditions.
- Enhanced Supply Chain Reliability: The robustness of the ionic liquid catalyst contributes to a more resilient supply chain by reducing dependency on specialized catalyst suppliers who may have long lead times. Since the catalyst can be synthesized from common industrial chemicals like ethanolamine and chlorosulfonic acid, the risk of raw material shortages is minimized. The ability to recycle the catalyst in-house also decouples production capacity from external catalyst supply constraints, giving manufacturing teams greater autonomy and control over their schedules. This self-sufficiency is crucial for maintaining continuous operations and meeting tight delivery deadlines for downstream API manufacturers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling up a solvent-free process is inherently safer and more straightforward than scaling a solvent-dependent one, as issues related to heat transfer and solvent flammability are largely mitigated. The reduced volume of reaction mass allows for higher throughput in existing reactor vessels, effectively increasing production capacity without the need for capital-intensive infrastructure expansion. From a regulatory perspective, the absence of VOC emissions simplifies the permitting process and reduces the burden of environmental monitoring. This alignment with green chemistry principles enhances the corporate sustainability profile, which is increasingly becoming a key criterion for partnerships with major global pharmaceutical companies committed to responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this ionic liquid technology in industrial settings. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential adopters. Understanding these nuances is critical for process engineers and quality assurance teams who are evaluating the feasibility of integrating this method into their existing production lines. The answers reflect the proven capabilities of the [SFHEA]X system as demonstrated in the reported examples.
Q: What are the primary advantages of using [SFHEA]X ionic liquids over traditional catalysts for Henry reactions?
A: The [SFHEA]X ionic liquids offer a solvent-free reaction environment, eliminating the need for toxic organic solvents and simplifying downstream purification. Furthermore, the catalyst demonstrates exceptional thermal stability and can be recycled multiple times without significant loss in catalytic activity or yield, addressing both cost and environmental concerns inherent in traditional heterogeneous catalysis.
Q: Can this ionic liquid catalyst system accommodate diverse aromatic aldehyde substrates?
A: Yes, the patent data confirms high compatibility with a wide range of substrates including electron-rich and electron-deficient aromatic aldehydes such as 4-methoxybenzaldehyde, 4-nitrobenzaldehyde, and heterocyclic aldehydes like 2-thiophenealdehyde. The tunable acidity of the ionic liquid allows for effective activation of various aldehyde electrophiles, ensuring broad substrate scope for pharmaceutical intermediate synthesis.
Q: How does the solvent-free nature of this process impact industrial scalability?
A: Operating without organic solvents drastically reduces the reactor volume required per unit of product and eliminates the complex infrastructure needed for solvent recovery and distillation. This leads to a significantly smaller physical footprint for production facilities and reduces the safety risks associated with handling large volumes of flammable volatile organic compounds, making the scale-up process more straightforward and economically viable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nitrostyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies like the ethanolamine-based ionic liquid system for producing high-purity beta-nitrostyrene intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required by the global pharmaceutical industry. We understand that consistency and quality are paramount, and our team is dedicated to optimizing every step of the synthesis to minimize impurities and maximize yield. By leveraging our expertise in green chemistry and process intensification, we help our clients achieve their sustainability goals without compromising on product performance or supply security.
We invite you to collaborate with us to explore how this advanced solvent-free technology can enhance your supply chain efficiency and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out to us to request specific COA data and route feasibility assessments for your target molecules. Whether you are looking to secure a reliable source of beta-nitrostyrene derivatives or need assistance in developing a custom synthetic route, NINGBO INNO PHARMCHEM is your trusted partner for delivering excellence in fine chemical manufacturing. Let us help you navigate the complexities of modern chemical production with solutions that are both economically and environmentally superior.
