Advanced Manganese-Promoted Synthesis of Nitro-Tetrahydrobenzofuran Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant advancement in this domain is detailed in Chinese Patent CN101628904B, which discloses a highly efficient method for synthesizing 2-nitro-3-aryl-2,3,5,7-tetrahydrobenzofuran-4-one derivatives. These compounds are not merely academic curiosities; they represent versatile intermediates capable of further transformation into physiologically active agents, including potential drugs and natural product analogs. The core innovation lies in the strategic replacement of harsh, halogenated starting materials with readily available beta-nitroalkenes, facilitated by a manganese(III) acetate promoter. This shift addresses long-standing challenges in process chemistry, offering a route that is both economically viable and environmentally superior. By leveraging ethanol as a green solvent and operating under mild thermal conditions, this technology provides a reliable pharmaceutical intermediate supplier with a distinct competitive edge in manufacturing high-purity scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of dihydrofuran derivatives relied heavily on methodologies that were fraught with operational and economic inefficiencies. As illustrated in the historical context of the field, earlier reports, such as those by Daniel Dauzonne, utilized 2-chloro-2-nitrostyrene reacting with 5,5-dimethyl-1,3-cyclohexanedione.

This legacy approach necessitated stringent anhydrous and oxygen-free conditions, requiring the use of expensive solvents like ethylene glycol dimethyl ether and prolonged heating periods of up to 24 hours under reflux. Furthermore, the reliance on halogenated substrates introduces significant atom economy penalties; the chlorine atom serves only as a leaving group, generating stoichiometric amounts of hydrogen chloride waste which poses environmental hazards and requires costly neutralization and disposal protocols. The multi-step preparation of the halogenated starting materials themselves further inflates the cost of goods and complicates the supply chain, making large-scale production of these intermediates prohibitively expensive for many applications.
The Novel Approach
In stark contrast, the methodology described in CN101628904B revolutionizes the synthetic landscape by employing beta-nitrostyrenes and their derivatives as the primary electrophilic partners. This novel approach utilizes manganese(III) acetate as a single-electron oxidant promoter to drive the cyclization with 1,3-cyclic diketones. The reaction proceeds smoothly in 95% aqueous ethanol, a solvent that is not only inexpensive and widely available but also aligns with green chemistry principles by reducing toxicity and flammability risks associated with ethers. The process operates at mild temperatures ranging from 30°C to 60°C, with optimal results observed at 50°C, completing the transformation in as little as 0.5 hours. This dramatic reduction in reaction time, coupled with the elimination of halogenated waste and the use of commodity chemicals, represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, enabling producers to achieve high yields of up to 86% with minimal downstream processing.
Mechanistic Insights into Mn(OAc)3-Promoted Cyclization
The success of this synthetic route hinges on the unique reactivity profile of manganese(III) acetate, which acts as a potent one-electron oxidant to initiate a radical cascade. The mechanism likely involves the generation of a carbon-centered radical from the enol form of the 1,3-dicarbonyl compound, which then undergoes conjugate addition to the electron-deficient beta-carbon of the nitroalkene. This initial addition creates a new carbon-carbon bond and a stabilized radical intermediate adjacent to the nitro group. Subsequent intramolecular cyclization occurs when the oxygen of the nitro group or the enol oxygen attacks the radical center, followed by oxidation and deprotonation steps to restore aromaticity or establish the final double bond within the furan ring system. This radical pathway bypasses the need for strong bases or nucleophilic substitution mechanisms that typically require activated leaving groups like halides.
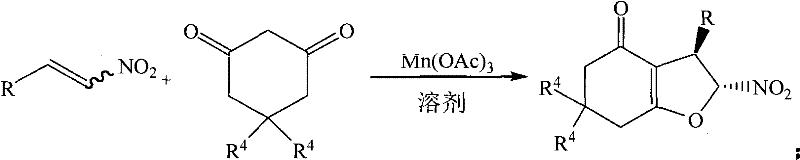
From an impurity control perspective, this mechanism offers superior regioselectivity and stereoselectivity compared to ionic alternatives. The radical nature of the reaction ensures that the addition occurs specifically at the beta-position of the nitroalkene, minimizing the formation of regioisomers that could complicate purification. Furthermore, because the reaction does not generate acidic byproducts like HCl, there is a reduced risk of acid-catalyzed decomposition of the sensitive nitro or carbonyl functionalities during the reaction course. The use of ethanol as a protic solvent may also assist in stabilizing transition states through hydrogen bonding, further enhancing the fidelity of the cyclization. This precise control over the reaction trajectory ensures that the resulting 2-nitro-3-aryl-2,3,5,7-tetrahydrobenzofuran-4-one derivatives are obtained with high chemical purity, reducing the burden on downstream crystallization or chromatography steps and ensuring consistent quality for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Nitro-3-Aryl-Tetrahydrobenzofuran-4-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the manganese-promoted pathway. The protocol is designed to be operationally simple, avoiding the need for specialized equipment like gloveboxes or rigorous drying trains. Typically, the beta-nitrostyrene derivative and the 1,3-cyclic diketone are combined in a molar ratio of approximately 1:2, with two equivalents of manganese(III) acetate added to drive the oxidative cyclization to completion. The choice of solvent is critical; while various alcohols and acids were screened, 95% ethanol was identified as the optimal medium, balancing solubility and reaction rate. The mixture is heated gently, often in a water bath to prevent localized overheating, and monitored via thin-layer chromatography (TLC) to determine the endpoint. Once the starting material is consumed, the workup is straightforward, involving standard extraction or direct purification techniques.
- Dissolve beta-nitrostyrene derivative, manganese acetate (Mn(OAc)3), and 1,3-cyclic diketone (such as dimedone) in 95% ethanol solvent.
- Heat the reaction mixture in a water bath at 50°C and monitor progress via TLC until completion (approx. 0.5 hours).
- Upon completion, isolate the target product using flash column chromatography with a petroleum ether and acetone mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this manganese-promoted synthesis offers tangible strategic benefits that extend beyond simple yield metrics. The transition from specialized halogenated precursors to commodity beta-nitrostyrenes fundamentally alters the cost structure of the supply chain. Beta-nitrostyrenes are widely produced industrial chemicals with stable pricing and multiple global sources, mitigating the risk of supply disruption that often plagues custom-synthesized halogenated intermediates. Additionally, the elimination of anhydrous conditions and expensive ether solvents reduces the overhead costs associated with solvent recovery and hazardous waste disposal. The ability to run the reaction in aqueous ethanol simplifies the engineering controls required for the manufacturing facility, potentially lowering capital expenditure for new production lines dedicated to these intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the raw material portfolio and the reduction of processing time. By removing the requirement for 2-chloro-2-nitrostyrene, manufacturers avoid the multi-step synthesis and purification costs inherent to halogenated substrates. Furthermore, the reaction time is compressed from 24 hours to under one hour, which drastically increases reactor throughput and asset utilization rates. The use of ethanol, a low-cost and renewable solvent, replaces expensive glymes, and the absence of corrosive HCl byproducts eliminates the need for specialized corrosion-resistant reactors and extensive scrubbing systems, leading to substantial cost savings in both CAPEX and OPEX without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available starting materials. Unlike niche halogenated compounds that may have limited suppliers, beta-nitrostyrenes and dimedone are bulk chemicals with robust global supply networks. This diversity of supply sources reduces the risk of bottlenecks and allows for more flexible sourcing strategies. Moreover, the shortened reaction cycle time means that production batches can be turned over much more rapidly, allowing manufacturers to respond more agilely to fluctuations in demand. The simplified workup procedure also reduces the lead time for high-purity pharmaceutical intermediates, ensuring that downstream customers receive their materials faster and with greater consistency.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is facilitated by its inherent safety and environmental profile. The use of ethanol minimizes volatile organic compound (VOC) emissions compared to chlorinated or ether solvents, aiding in compliance with increasingly stringent environmental regulations. The atom economy of the reaction is superior because all atoms from the starting materials are incorporated into the product or benign byproducts, aligning with green chemistry mandates. The absence of heavy metal catalysts (using manganese which is less toxic than many transition metals) and halogenated waste streams simplifies wastewater treatment and solid waste disposal. This environmental compatibility not only reduces regulatory burdens but also enhances the sustainability credentials of the final product, a factor of growing importance to end-users in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios and for procurement teams assessing the long-term viability of the supply source.
Q: What are the key advantages of this Mn(OAc)3 method over traditional halogenated routes?
A: This method eliminates the need for difficult-to-prepare 2-halo-2-nitrostyrenes, uses inexpensive ethanol instead of toxic glymes, and avoids generating corrosive HCl byproducts, significantly improving atom economy and environmental safety.
Q: What is the optimal reaction temperature and time for this synthesis?
A: According to patent optimization data, the reaction performs best at 50°C in 95% ethanol, achieving maximum yields of up to 86% within just 0.5 hours, which is drastically faster than the 24-hour reflux required by older methods.
Q: Can this process accommodate diverse aromatic substituents?
A: Yes, the protocol demonstrates excellent substrate scope, successfully tolerating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as nitro, fluoro, chloro, and bromo on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-3-Aryl-Tetrahydrobenzofuran-4-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the manganese-promoted cyclization route described in CN101628904B and possesses the technical expertise to optimize it for your specific needs. We offer comprehensive CDMO services that leverage this advanced chemistry to deliver high-quality intermediates with rigorous QC labs ensuring stringent purity specifications. Our facilities are equipped to handle the commercial scale-up of complex pharmaceutical intermediates, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to providing a seamless transition from gram-scale research to multi-ton manufacturing, ensuring that your supply chain remains robust and uninterrupted.
We invite you to collaborate with us to explore how this cost-effective and green synthesis can benefit your project pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us today to request specific COA data for our catalog of tetrahydrobenzofuran derivatives or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in turning innovative chemistry into commercial reality.
