Advanced Manufacturing of Beta-Nitrostyrene Compounds for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high efficiency with uncompromising safety standards. Patent CN101765579A introduces a transformative methodology for the production of beta-nitrostyrene compounds, which serve as critical intermediates in the synthesis of therapeutic agents such as baclofen and various antispasmodics. This technology addresses a long-standing industrial challenge by shifting the reaction paradigm from hazardous high-temperature reflux conditions to a controlled, industrially safe temperature range of 70-80°C. By utilizing a primary amine catalyst in an acetic acid solvent system, this process not only mitigates the risk of thermal runaway—a critical concern when handling nitro compounds—but also delivers exceptional yields ranging from 80% to 99%. For R&D directors and process chemists, this represents a significant leap forward in process safety and impurity control, while supply chain managers will recognize the inherent value in a scalable, reliable pharmaceutical intermediate supplier capable of delivering consistent quality without the bottlenecks associated with dangerous exothermic reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-nitrostyrene compounds has relied heavily on methods utilizing ammonium acetate as a base, often necessitating heating under reflux conditions. As detailed in prior art such as JP 9-194457 and JP 11-71348, these conventional approaches require reaction temperatures approaching the boiling point of the solvent, typically around 95°C. This high-temperature operation presents severe safety liabilities; Accelerating Rate Calorimeter (ARC) data indicates that at 95°C, the time to maximum self-reaction rate (runaway) is merely 7.9 hours, which is dangerously close to typical residence times of 12 hours. Furthermore, alternative methods using nitroalkanes as solvents have demonstrated limited efficacy, often capping yields at approximately 67% even under rigorous reflux. These inefficiencies translate directly into higher raw material consumption, increased waste generation, and significant operational risks that complicate the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN101765579A leverages a primary amine, preferably benzylamine, within an acetic acid solvent matrix to drive the condensation of benzaldehyde derivatives and nitromethane. This strategic shift allows the reaction to proceed efficiently at a much lower temperature window of 70-80°C. By operating below the Adiabatic Decomposition Temperature for 24 hours (ADT24) of 78°C, the process establishes a robust safety margin that effectively prevents thermal runaway even in large-scale adiabatic systems. The result is a dramatic enhancement in process reliability and output, with yields consistently achieving between 80% and 99%. This approach not only resolves the safety deficits of the past but also streamlines the workflow, making it an ideal solution for cost reduction in API manufacturing where safety and yield are paramount.
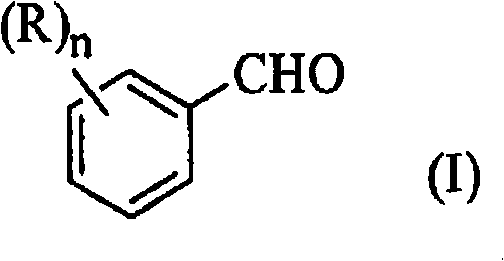
Mechanistic Insights into Primary Amine-Catalyzed Condensation
The core of this technological advancement lies in the specific interaction between the primary amine catalyst and the acetic acid solvent. Unlike ammonium acetate, which acts as a weaker base requiring higher thermal energy to drive the Henry reaction (nitroaldol condensation) followed by dehydration, primary amines such as benzylamine facilitate a more rapid and selective formation of the nitrostyrene double bond. The acetic acid solvent plays a dual role: it acts as a polar medium that stabilizes the transition states involved in the condensation and, crucially, enables a simplified isolation protocol. Upon completion of the reaction, the addition of water to the acetic acid mixture induces immediate crystallization of the beta-nitrostyrene product. This phenomenon is attributed to the reduced solubility of the nitro-compound in the aqueous-acetic acid mixture, allowing for direct solid-liquid separation without the need for energy-intensive distillation or complex organic extractions typically required with non-polar solvents like toluene.
From an impurity control perspective, the lower operating temperature of 70-80°C is instrumental in minimizing side reactions. High-temperature conditions often promote polymerization of the nitroalkene or degradation of sensitive functional groups on the aromatic ring. By maintaining the reaction within this narrow, safe thermal window, the process ensures a cleaner crude profile, which significantly reduces the burden on downstream purification steps. For a reliable agrochemical intermediate supplier or pharma partner, this means the final product meets stringent purity specifications with less rework. The ability to tune the amine loading between 0.2 to 1.5 molar equivalents further allows process engineers to optimize the balance between reaction rate and economic efficiency, ensuring that the catalytic cycle proceeds to completion without excessive reagent waste.
How to Synthesize 4-Chloro-beta-nitrostyrene Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing multipurpose chemical plants. The process begins with the dissolution of the benzaldehyde derivative, such as 4-chlorobenzaldehyde, and the primary amine catalyst in acetic acid. Once the mixture is heated to the target range of 70-80°C, nitromethane is added dropwise over a controlled period, typically 1 to 3 hours, to manage the exotherm and maintain safety. Following the addition, a short stirring period ensures full conversion, after which water is introduced to precipitate the product. This straightforward sequence eliminates the need for specialized high-pressure equipment or cryogenic cooling, making it highly accessible for commercial production. For detailed operational parameters and specific stoichiometric ratios tailored to your facility, please refer to the standardized synthesis guide below.
- Dissolve the benzaldehyde derivative and a primary amine (such as benzylamine) in an acetic acid solvent within a reaction vessel.
- Heat the solution to a safe operating range of 70-80°C and slowly add nitromethane dropwise while maintaining temperature control.
- After reaction completion, induce crystallization of the beta-nitrostyrene product by adding water to the reaction mixture at 30-60°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic benefits that extend beyond simple yield improvements. The shift to a safer, lower-temperature process fundamentally alters the cost structure of manufacturing beta-nitrostyrene derivatives. By eliminating the need for prolonged reflux at near-boiling temperatures, the process drastically reduces energy consumption associated with heating and cooling cycles. Furthermore, the simplified workup procedure—crystallization via water addition rather than complex solvent swaps—minimizes solvent usage and waste disposal costs. These operational efficiencies translate into substantial cost savings in fine chemical intermediates manufacturing, allowing buyers to secure high-quality materials at a more competitive price point without compromising on safety or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of high-energy reflux conditions and the use of readily available, commodity-grade reagents like acetic acid and benzylamine significantly lowers the variable cost of production. Unlike methods requiring expensive transition metal catalysts or specialized solvents, this organic condensation relies on inexpensive inputs. The high yield of 80-99% ensures that raw material utilization is maximized, reducing the cost per kilogram of the final active pharmaceutical ingredient precursor. Additionally, the simplified isolation step reduces labor hours and equipment occupancy time, further driving down the overall manufacturing overhead.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply continuity. Processes that operate near thermal runaway limits are prone to unplanned shutdowns and regulatory scrutiny. By operating well within the ADT24 safety margin, this method ensures consistent batch-to-batch reliability and minimizes the risk of production halts due to safety incidents. The raw materials, including various substituted benzaldehydes and nitromethane, are globally sourced commodities, ensuring that supply chain disruptions are minimized. This stability makes the supplier a dependable partner for long-term contracts and Just-In-Time delivery models essential for modern pharmaceutical manufacturing.
- Scalability and Environmental Compliance: The inherent safety of the 70-80°C reaction window makes this process exceptionally scalable from pilot plant to multi-ton commercial production. The reduced risk profile simplifies the permitting process for new production lines and lowers insurance premiums. Environmentally, the use of acetic acid, which is biodegradable and easier to treat in wastewater systems compared to chlorinated solvents, aligns with green chemistry principles. The high atom economy and reduced solvent waste generation contribute to a lower environmental footprint, helping companies meet increasingly strict ESG (Environmental, Social, and Governance) targets and regulatory standards for industrial emissions.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is vital for stakeholders evaluating its integration into their supply chain. The following questions address common concerns regarding safety, scalability, and product quality, drawing directly from the experimental data and comparative examples provided in the patent literature. These insights are intended to clarify how this technology outperforms legacy methods and why it is becoming the preferred choice for high-volume production of nitrostyrene intermediates.
Q: Why is the primary amine method safer than conventional ammonium acetate methods?
A: Conventional methods often require reflux temperatures near the boiling point (approx. 95°C), where ARC data indicates a runaway reaction risk within 7.9 hours. The primary amine method operates safely at 70-80°C, well below the ADT24 safety threshold of 78°C, significantly reducing thermal runaway risks.
Q: What yields can be expected from this acetic acid condensation process?
A: This novel approach consistently achieves yields between 80% and 99%, with preferred embodiments reaching 85% to 98%. This is a substantial improvement over older methods which often struggled to exceed 67% yield under similar conditions.
Q: How does the choice of solvent impact downstream processing?
A: Using acetic acid instead of traditional solvents like toluene facilitates a unique workup procedure. The product can be easily crystallized simply by adding water to the reaction mixture, eliminating the need for complex extraction steps and reducing solvent waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nitrostyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the safety and yield benefits of this acetic acid-based method are fully realized at scale. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of beta-nitrostyrene meets the exacting standards required for downstream API synthesis. Our commitment to technical excellence ensures that you receive a high-purity pharmaceutical intermediate that facilitates smooth subsequent reactions in your own manufacturing processes.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to validate the superior quality and economic advantages of our manufacturing capabilities before committing to large-scale orders. Let us be your trusted partner in delivering safe, efficient, and high-quality chemical solutions.
