Revolutionizing Opioid Analgesic Production via Advanced Total Synthesis of Morphine Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift in the production of opioid analgesics, driven by the urgent need to decouple supply chains from agricultural vulnerabilities. Patent CN114502560B introduces a groundbreaking total synthesis methodology for morphine derivatives, centering on a novel intermediate designated as Formula I. This intermediate serves as a pivotal precursor for essential medications such as codeine, oxycodone, naloxone, and naltrexone, which are critical for pain management and addiction treatment globally. Unlike traditional methods that rely on the extraction of morphine and thebaine from poppy plants—a process fraught with geopolitical risks and yield fluctuations—this chemical synthesis route offers a robust, reproducible alternative. The structural complexity of the morphine nucleus, characterized by five contiguous chiral centers and a compact five-ring fused system, has historically posed significant synthetic challenges. However, the innovative approach detailed in this patent successfully navigates these complexities through a series of highly selective transformations.
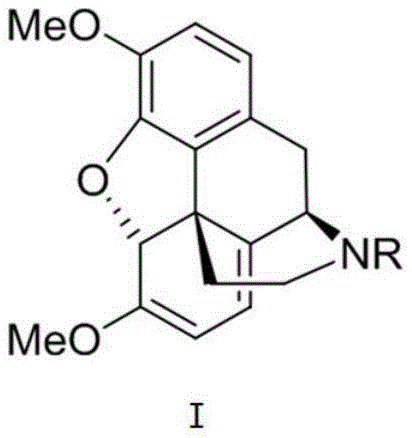
The strategic design of Intermediate I allows for the efficient construction of the morphine skeleton, bypassing the need for agricultural raw materials entirely. By establishing a reliable chemical pathway, manufacturers can mitigate the supply disruptions often caused by crop failures or regulatory restrictions on poppy cultivation. This technological advancement represents a significant leap forward for reliable pharmaceutical intermediates supplier networks, ensuring that the global demand for essential pain relief medications can be met with greater stability and predictability. The ability to synthesize these complex molecules from simple starting materials not only enhances supply security but also opens new avenues for the development of novel analogs with improved therapeutic profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of morphine-like drugs has been inextricably linked to the cultivation of opium poppies, a practice that consumes vast amounts of arable land and is highly susceptible to external variables. The reliance on biological sources introduces inherent instability into the supply chain, as yields can be drastically affected by pests, diseases, and climatic conditions. Furthermore, the semi-synthetic derivation from extracted thebaine involves complex purification processes to remove plant-based impurities, which adds significant cost and time to the manufacturing workflow. From a regulatory perspective, the control of poppy cultivation is stringent and varies by region, creating bottlenecks that can lead to shortages of critical medications. The existing total synthesis methods reported in prior art have also fallen short of industrial viability, often requiring an excessive number of steps—up to 17 for codeine—with low overall yields and poor stereocontrol, making them economically unfeasible for large-scale production.
The Novel Approach
The methodology disclosed in patent CN114502560B addresses these deficiencies by streamlining the synthetic route to just 12 steps for codeine, achieving a total yield of 29% compared to the 14% typical of previous methods. This significant improvement is anchored by the use of Intermediate I, which is constructed via a highly efficient intramolecular oxidative dearomatization Heck reaction. This key transformation allows for the rapid assembly of the complex polycyclic core with high regioselectivity and yield, overcoming the limitations of earlier biomimetic approaches that suffered from poor efficiency. By utilizing common chemical reagents and avoiding harsh conditions, the new process simplifies post-reaction processing and enables continuous operation without the need for extensive intermediate purification. This reduction in operational complexity translates directly into cost reduction in pharmaceutical intermediates manufacturing, making the total synthesis route commercially competitive with traditional extraction methods while offering superior consistency and control over the final product quality.
Mechanistic Insights into Intramolecular Oxidative Dearomatization Heck Reaction
The cornerstone of this synthetic breakthrough is the intramolecular oxidative dearomatization Heck reaction, which facilitates the construction of the quaternary carbon center essential for the morphine architecture. This reaction involves the coupling of a phenolic derivative with a halogenated side chain under palladium catalysis, leading to the formation of the characteristic spiro-dienone system found in Intermediate I. The patent details extensive optimization of this step, identifying specific ligand and catalyst combinations that maximize yield and minimize byproduct formation. For instance, the use of bulky phosphine ligands in conjunction with palladium chloride has been shown to enhance the reactivity of the substrate, allowing the reaction to proceed efficiently at moderate temperatures. The mechanistic pathway likely involves the oxidative addition of the palladium catalyst to the aryl halide, followed by migratory insertion into the alkene and subsequent beta-hydride elimination or oxidation to restore aromaticity or form the dienone.
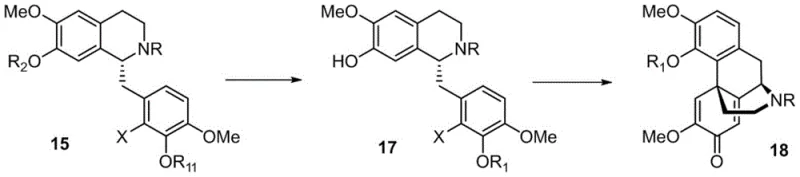
Impurity control is rigorously managed through the precise selection of reaction conditions and protecting groups. The patent emphasizes the importance of protecting group compatibility, particularly for the secondary amine and hydroxyl functionalities, to prevent side reactions during the oxidative coupling. By employing protecting groups such as tosyl or benzyl, the synthesis ensures that reactive sites are masked until the appropriate stage, thereby maintaining high chemical fidelity throughout the sequence. Furthermore, the asymmetric transfer hydrogenation step utilized earlier in the sequence establishes the initial chirality with high enantiomeric excess, which is preserved through the subsequent transformations. This stereochemical integrity is crucial for the biological activity of the final opioid products, ensuring that the synthesized drugs meet the stringent purity specifications required for clinical use. The ability to achieve 99.9% ee after recrystallization demonstrates the robustness of this chiral control strategy.
How to Synthesize Morphine Intermediate I Efficiently
The synthesis of Intermediate I is a multi-stage process that begins with the condensation of protected phenethylamine and phenylacetic acid derivatives to form an amide backbone. This precursor then undergoes a Bischler-Napieralski cyclization to generate a dihydroisoquinoline, which is subsequently subjected to asymmetric transfer hydrogenation to install the critical chiral center. The resulting tetrahydroisoquinoline derivative is then functionalized to introduce the necessary halogen and phenolic groups for the key Heck coupling. Detailed standardized synthesis steps for the preparation of Intermediate I and its conversion to various opioid analgesics are provided in the guide below.
- Perform amine-acid condensation between protected phenethylamine and phenylacetic acid derivatives to form the amide precursor.
- Execute Bischler-Napieralski cyclization followed by asymmetric transfer hydrogenation to establish the chiral tetrahydroisoquinoline core with high enantiomeric excess.
- Conduct the critical intramolecular oxidative dearomatization Heck reaction using palladium catalysis to construct the complex morphine skeleton.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this total synthesis technology offers transformative benefits that extend beyond mere chemical efficiency. The primary advantage lies in the decoupling of production from agricultural dependencies, which significantly enhances supply chain reliability. By sourcing chemical starting materials rather than plant extracts, manufacturers can secure a stable supply of raw materials that is immune to the vagaries of weather and crop cycles. This stability is critical for maintaining continuous production schedules and meeting the consistent demand for essential pain medications in the global market. Additionally, the streamlined nature of the synthesis, with fewer steps and higher yields, reduces the overall consumption of solvents and reagents, leading to substantial cost savings in raw material procurement and waste disposal.
- Cost Reduction in Manufacturing: The reduction of synthetic steps from 17 to 12 for codeine production fundamentally alters the cost structure of manufacturing. Fewer steps mean less energy consumption, reduced labor hours, and lower capital expenditure on equipment utilization. The elimination of expensive transition metal catalysts in certain steps, or the optimization of their loading as described in the patent, further drives down the cost of goods sold. Moreover, the high yield of the key Heck reaction minimizes the loss of valuable intermediates, ensuring that a greater proportion of input materials are converted into saleable product. These efficiencies collectively contribute to a more competitive pricing model without compromising on the quality or purity of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Dependence on poppy cultivation introduces significant geopolitical and logistical risks, including trade restrictions and transportation delays associated with controlled substances. A fully synthetic route mitigates these risks by allowing production to be localized in regions with robust chemical infrastructure, closer to end markets. This localization reduces lead times and enhances the responsiveness of the supply chain to sudden spikes in demand, such as those seen during public health crises. The use of common, commercially available reagents ensures that supply bottlenecks are unlikely, providing a resilient foundation for long-term production planning and inventory management.
- Scalability and Environmental Compliance: The mild reaction conditions and tolerance to water and oxygen described in the patent make this process highly amenable to scale-up. Large-scale reactors can be employed without the need for specialized inert atmosphere equipment, reducing capital investment. Furthermore, the reduction in step count and the ability to telescope reactions without intermediate isolation significantly decrease the volume of organic waste generated. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the burden of wastewater treatment and hazardous waste disposal. The process thus offers a greener alternative to traditional methods, enhancing the company's environmental profile while maintaining operational efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this total synthesis technology. The answers are derived directly from the experimental data and embodiments provided in patent CN114502560B, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: How does this total synthesis method improve upon traditional poppy extraction?
A: Traditional extraction relies on agricultural poppy cultivation, which is subject to climate variability, political instability, and strict regulatory controls. This total synthesis method provides a consistent, laboratory-controlled supply chain that eliminates agricultural risks and reduces the number of synthetic steps from 17 to 12 for codeine production.
Q: What is the enantiomeric purity achievable with this synthetic route?
A: The patent data indicates that through optimized asymmetric transfer hydrogenation and recrystallization processes, the intermediate and final products can achieve an enantiomeric excess (ee) of up to 99.9%, ensuring high pharmaceutical grade purity suitable for strict regulatory compliance.
Q: Is this synthesis method scalable for industrial manufacturing?
A: Yes, the method utilizes common chemical reagents and mild reaction conditions that are not highly sensitive to water or oxygen. The reduction in synthetic steps and the ability to perform continuous multi-step operations without intermediate purification significantly enhance scalability and reduce three-waste emissions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Morphine Intermediate Supplier
The technological potential of this total synthesis route is immense, offering a viable path to secure the global supply of essential opioid analgesics. As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses the expertise and infrastructure to translate this complex chemistry into commercial reality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of intermediate meets the highest industry standards, facilitating seamless regulatory approval for your final drug products.
We invite you to explore the possibilities of this advanced synthesis method for your portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how partnering with us can optimize your supply chain and reduce manufacturing costs effectively.
