Advanced Manufacturing of Dihydro-1,3,5-Triazine Derivatives for Diabetes Treatment
Advanced Manufacturing of Dihydro-1,3,5-Triazine Derivatives for Diabetes Treatment
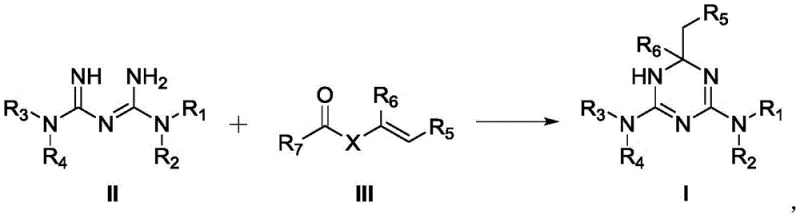
The pharmaceutical industry continuously seeks robust synthetic routes for active pharmaceutical ingredients (APIs) and their intermediates, particularly for treating metabolic disorders like Type II diabetes. Patent CN114763339A discloses a groundbreaking process for the preparation of dihydro-1,3,5-triazine derivatives, which have demonstrated significant potential in managing insulin resistance. This technology represents a paradigm shift from traditional aldehyde-based condensation methods to a more efficient vinyl ester or vinylamide amidation strategy. By leveraging the unique reactivity of vinyl groups with biguanide substrates, this method achieves exceptional conversion rates and simplifies downstream purification. For R&D directors and procurement specialists, understanding this novel pathway is critical for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials consistently. The following analysis details the mechanistic advantages and commercial viability of this synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydro-1,3,5-triazine derivatives has relied heavily on the condensation of biguanides with aldehydes, acetals, or hemiacetals under acidic or basic conditions, as documented in prior art such as WO2001055122 and WO2009095159. These conventional pathways suffer from inherent thermodynamic limitations, primarily due to the reversible nature of the imine formation step, which often necessitates the continuous removal of water to drive the equilibrium toward the product. Furthermore, the use of aldehydes can introduce stability issues, as they are prone to oxidation and polymerization, leading to complex impurity profiles that are difficult to remove during purification. The requirement for harsh acidic catalysts or strict anhydrous conditions also increases operational costs and safety risks, making these methods less attractive for cost reduction in pharmaceutical intermediates manufacturing on a commercial scale.
The Novel Approach
In contrast, the process disclosed in CN114763339A utilizes vinyl esters or vinylamides as electrophilic partners, which react with biguanide substrates to form the triazine ring through an irreversible addition-elimination mechanism. This approach effectively bypasses the equilibrium constraints of aldehyde condensations, as the resulting enol or amide tautomers stabilize the final product, driving the reaction to near-quantitative completion without the need for water scavengers. The use of readily available starting materials like vinyl acetate and metformin salts significantly lowers raw material costs while simplifying the reaction setup to ambient pressure and moderate temperatures. This innovation not only enhances the overall yield, reported up to 100% in specific embodiments, but also drastically reduces the generation of hazardous waste, aligning with modern green chemistry principles essential for sustainable commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Vinyl Ester Amidation and Cyclization
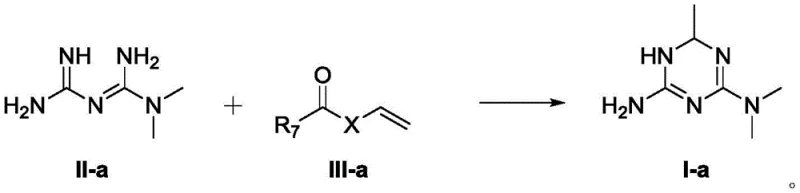
The core of this synthetic breakthrough lies in the nucleophilic attack of the biguanide nitrogen on the electron-deficient beta-carbon of the vinyl ester or vinylamide. Unlike traditional acylation, this Michael-type addition is followed by an intramolecular cyclization that is thermodynamically driven by the subsequent tautomerization of the intermediate. The patent explicitly illustrates that the final triazine product can exist in multiple tautomeric forms (A, B, and C), which contributes to the stability of the heterocyclic ring and prevents retro-reaction. This mechanistic feature ensures that once the ring is formed, it remains intact under the reaction conditions, thereby minimizing the formation of open-chain byproducts that typically plague biguanide chemistry. For process chemists, this implies a wider operating window where slight variations in temperature or stoichiometry do not critically compromise the integrity of the final API intermediate.
Furthermore, the control of stereochemistry is addressed through a sophisticated chiral resolution strategy that exploits the acidity of the triazine ring protons. The process allows for the formation of diastereomeric salts using optically pure acids, such as di-O,O'-p-toluoyl-D-tartaric acid, which exhibit distinct solubility profiles in alcoholic solvents. This differential solubility enables the selective crystallization of one enantiomer over the other, effectively separating the biologically active isomer from its less potent counterpart. The ability to achieve high enantiomeric excess (ee) values, exceeding 99% after recrystallization, is paramount for regulatory compliance and patient safety, ensuring that the final drug substance meets stringent purity specifications required by global health authorities for antidiabetic medications.
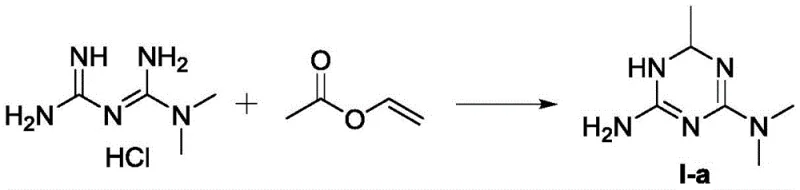
How to Synthesize 1,6-Dihydro-2-amino-4-dimethylamino-6-methyl-1,3,5-triazine Efficiently
The practical implementation of this technology involves a straightforward one-pot reaction followed by a crystallization-induced resolution step, making it highly accessible for manufacturing teams. The process begins with the dissolution of a biguanide salt, such as metformin hydrochloride or acetate, in a polar solvent system like methanol or water, where it is treated with a base to generate the free base in situ. Subsequent addition of vinyl acetate initiates the cyclization, which proceeds rapidly at room temperature or with mild heating to 60°C, eliminating the need for energy-intensive reflux conditions. Detailed standardized synthesis steps for replicating this high-yield protocol are provided in the guide below, ensuring reproducibility across different production scales.
- React metformin hydrochloride or acetate with vinyl acetate in methanol/water with a base like KOH or ammonia at room temperature to 60°C.
- Concentrate the reaction mixture and purify the crude triazine derivative via recrystallization from ethanol or isobutanol.
- Resolve the racemic mixture using optically pure di-O,O'-p-toluoyl-D-tartaric acid in methanol to isolate the desired enantiomer with high ee value.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented process offers transformative benefits by replacing specialized, often expensive aldehyde reagents with commodity chemicals like vinyl acetate and ammonia. This substitution significantly de-risks the supply chain, as vinyl acetate is produced on a massive global scale for the polymer industry, ensuring consistent availability and price stability even during market fluctuations. The elimination of complex water-removal equipment and harsh acidic catalysts further reduces capital expenditure (CAPEX) for reactor modifications, allowing existing multipurpose facilities to adopt this technology with minimal downtime. For procurement managers, this translates to a more resilient sourcing strategy where the dependency on niche chemical suppliers is drastically reduced, fostering a more competitive bidding environment for raw materials.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing low-cost feedstocks and simplifying the workup procedure to mere concentration and filtration, removing the need for chromatographic purification in many cases. The high atom economy of the vinyl addition reaction minimizes waste disposal costs, while the ability to recover and recycle solvents like methanol and ethanol further enhances the overall economic efficiency of the production line. Additionally, the high yield reduces the cost per kilogram of the active intermediate, directly improving the margin profile for the final drug product without compromising quality standards.
- Enhanced Supply Chain Reliability: By relying on stable, non-hazardous reagents that are liquid at room temperature, the logistics of raw material transport and storage are significantly simplified, reducing the risk of supply disruptions due to regulatory restrictions on hazardous goods. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent mixtures, ensures that production schedules are not easily derailed by minor environmental variances or equipment limitations. This reliability is crucial for maintaining continuous supply to downstream formulation partners, preventing stockouts that could impact patient access to critical diabetes medications.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts and the use of aqueous or alcoholic solvent systems align perfectly with increasingly strict environmental regulations regarding volatile organic compounds (VOCs) and heavy metal residues in APIs. Scaling this process from pilot plant to multi-ton production is straightforward, as the exotherm is manageable and the viscosity of the reaction mixture remains low, facilitating efficient heat transfer and mixing in large reactors. This scalability ensures that the technology can meet growing global demand for antidiabetic therapies while maintaining a low environmental footprint, a key metric for modern corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazine synthesis technology, based on the detailed experimental data provided in the patent documentation. These insights are designed to clarify the operational feasibility and quality advantages of the new route for stakeholders evaluating its adoption. Understanding these nuances is essential for making informed decisions about process validation and regulatory filing strategies.
Q: What are the advantages of using vinyl esters over aldehydes for triazine synthesis?
A: Using vinyl esters avoids equilibrium issues common with aldehydes, driving the reaction to completion through irreversible tautomerization, resulting in higher yields up to 100% without needing water removal agents.
Q: How is high optical purity achieved in this process?
A: High optical purity is achieved through diastereomeric salt formation using chiral acids like di-O,O'-p-toluoyl-D-tartaric acid, allowing for efficient crystallization and separation of enantiomers with ee values exceeding 99%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commodity chemicals like vinyl acetate and metformin salts in common solvents like methanol and water, operating at ambient pressure and moderate temperatures, making it highly scalable and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative synthetic methodologies to deliver superior pharmaceutical intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of triazine derivatives meets the highest international standards for potency and enantiomeric purity.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain and reduce overall production costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective chemical solution available.
