Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Commercial Scale Production
Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Commercial Scale Production
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl substituted 1,2,4-triazine compounds, addressing critical bottlenecks in traditional heterocyclic synthesis. This innovation leverages a synergistic [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, facilitated by inexpensive inorganic bases. The significance of this technology extends beyond academic interest; it provides a robust platform for generating high-purity pharmaceutical intermediates that are essential for next-generation anticancer and antifungal agents. By eliminating the need for transition metal catalysts and harsh reaction conditions, this methodology aligns perfectly with the industry's shift towards greener, more sustainable manufacturing processes.
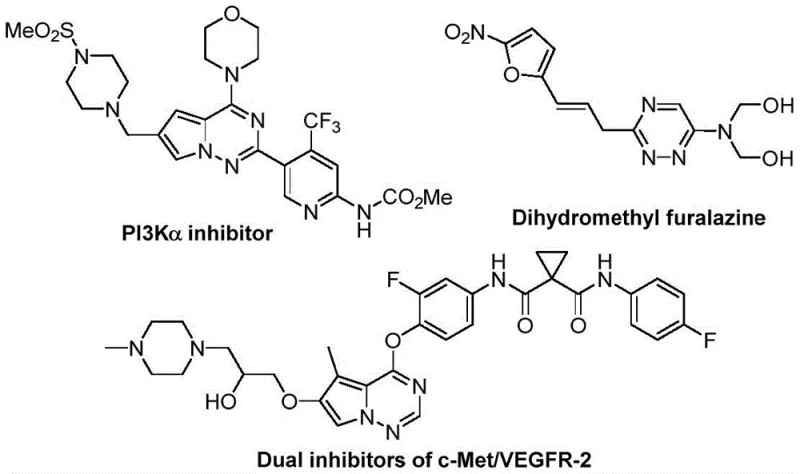
The strategic incorporation of trifluoromethyl groups into heterocyclic frameworks is a well-established tactic for enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As illustrated in the structural diversity of known bioactive molecules, the 1,2,4-triazine core serves as a privileged scaffold in drug discovery. However, accessing these fluorinated derivatives has historically been challenging due to the complexity of introducing the CF3 group late in the synthesis or the requirement for specialized fluorinating reagents. The disclosed method overcomes these hurdles by utilizing a trifluoroacetyl sulfur ylide precursor, which inherently carries the necessary fluorine content. This approach not only streamlines the synthetic sequence but also ensures that the resulting compounds possess the desired physicochemical properties required for rigorous biological evaluation and subsequent clinical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazine rings has relied heavily on condensation reactions between amidrazones and 1,2-diketones or alkynes, often necessitating high temperatures and prolonged reaction times. These traditional pathways frequently suffer from poor atom economy and limited substrate scope, restricting the ability of chemists to explore diverse chemical space efficiently. Furthermore, many classical methods require the use of expensive or toxic heavy metal catalysts to drive the cyclization, which introduces significant downstream purification challenges. The removal of trace metal residues to meet stringent pharmaceutical standards often requires additional processing steps, such as scavenger treatments or repeated recrystallizations, thereby increasing both the cost of goods and the overall lead time for project milestones. Additionally, the sensitivity of some intermediates to moisture or oxygen often mandates the use of inert atmospheres, adding operational complexity to the manufacturing process.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a mild, base-promoted cyclization that proceeds efficiently at room temperature under ambient air conditions. By employing potassium carbonate as a benign accelerator, the reaction avoids the pitfalls associated with strong bases or sensitive organometallic reagents. The mechanism involves the generation of a reactive nitrile imine intermediate from the chlorohydrazone, which then undergoes a concerted cycloaddition with the sulfur ylide. This pathway is remarkably tolerant of various functional groups, allowing for the introduction of electron-donating or electron-withdrawing substituents on the aromatic rings without compromising yield. The simplicity of the workup procedure, which typically involves filtration and standard column chromatography, further underscores the practical utility of this method for both laboratory-scale optimization and industrial production.
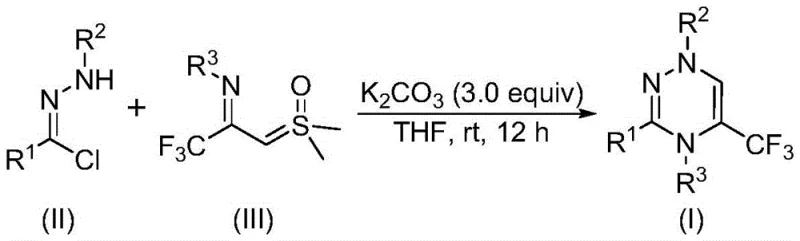
Mechanistic Insights into Base-Promoted [3+3] Cycloaddition
The mechanistic elegance of this transformation lies in the dual role of the base and the unique reactivity of the sulfur ylide species. Upon treatment with potassium carbonate, the chlorohydrazone undergoes dehydrohalogenation to generate a highly reactive nitrile imine dipole in situ. This transient species is immediately trapped by the trifluoroacetyl sulfur ylide, which acts as a dipolarophile in a formal [3+3] cycloaddition event. The reaction is driven forward by the elimination of dimethyl sulfoxide (DMSO), a thermodynamically favorable step that locks the six-membered triazine ring into place. Alternatively, the pathway may proceed through a stepwise sequence involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution, ultimately converging on the same stable heterocyclic product. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction kinetics and minimize the formation of potential byproducts.
From an impurity control perspective, the absence of transition metals significantly simplifies the impurity profile of the final API intermediate. Traditional metal-catalyzed routes often generate complex mixtures of metal-ligand complexes or homocoupling byproducts that are difficult to separate. In this metal-free protocol, the primary impurities are likely to be unreacted starting materials or hydrolysis products, which are generally more polar and easier to remove via standard silica gel chromatography. The use of tetrahydrofuran (THF) as the preferred solvent ensures excellent solubility for both organic substrates and the inorganic base, facilitating homogeneous reaction conditions that promote consistent conversion rates. This level of control over the reaction environment is crucial for maintaining batch-to-batch reproducibility, a key metric for any reliable pharmaceutical intermediate supplier aiming to support global clinical trials.
How to Synthesize Trifluoromethyl Substituted 1,2,4-Triazine Efficiently
To implement this synthesis effectively, operators should adhere to the optimized molar ratios and solvent volumes specified in the patent data to ensure maximum yield and purity. The process begins with the precise weighing of chlorohydrazone and trifluoroacetyl sulfur ylide, followed by their suspension in dry tetrahydrofuran. Potassium carbonate is then added in a slight excess to drive the equilibrium towards product formation while neutralizing the generated hydrogen chloride. The reaction mixture is stirred at ambient temperature for a period ranging from 10 to 14 hours, allowing sufficient time for the cycloaddition to reach completion without the need for external heating sources. Detailed standardized synthesis steps for scaling this reaction are provided in the guide below.
- Prepare the reaction mixture by adding potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide into an organic solvent such as tetrahydrofuran.
- Stir the mixture at room temperature (20-40°C) in an air atmosphere for 10 to 14 hours to allow the cycloaddition reaction to proceed.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final trifluoromethyl substituted 1,2,4-triazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend far beyond the laboratory bench. The elimination of expensive noble metal catalysts represents a direct reduction in raw material costs, while the simplified purification process lowers the consumption of specialty scavengers and solvents. Moreover, the ability to run the reaction in air rather than under nitrogen removes the need for specialized equipment and reduces the risk of batch failures due to inert gas supply interruptions. These factors collectively contribute to a more resilient and cost-effective supply chain, ensuring that critical intermediates can be delivered reliably even in volatile market conditions.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with commodity chemicals like potassium carbonate drastically lowers the direct material cost per kilogram of product. Furthermore, the mild reaction conditions eliminate the energy costs associated with heating or cooling large reactors, leading to substantial operational expenditure savings. The streamlined workup procedure reduces labor hours and solvent usage, further enhancing the overall economic viability of the process for large-scale API manufacturing.
- Enhanced Supply Chain Reliability: Since all starting materials, including chlorohydrazones and sulfur ylides, are derived from commercially available precursors, the risk of supply chain disruption is minimized. The robustness of the reaction to atmospheric conditions means that production can continue uninterrupted regardless of minor fluctuations in facility infrastructure. This reliability is paramount for maintaining continuous supply to downstream partners and meeting tight delivery schedules for clinical trial materials.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies waste stream management, reducing the burden of hazardous waste disposal and ensuring compliance with increasingly strict environmental regulations. The process is inherently scalable from gram to multi-ton quantities without significant re-engineering, allowing manufacturers to respond quickly to increased demand. This flexibility supports a agile manufacturing model that can adapt to the changing needs of the pharmaceutical pipeline.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these trifluoromethyl triazine derivatives. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for potential partners and stakeholders.
Q: What are the key advantages of this trifluoromethyl triazine synthesis method?
A: The method described in patent CN116253692A operates at room temperature in air without heavy metal catalysts, using inexpensive potassium carbonate as a promoter, which significantly simplifies purification and reduces environmental impact.
Q: What starting materials are required for this reaction?
A: The synthesis utilizes readily available chlorohydrazones and trifluoroacetyl sulfur ylides as primary building blocks, allowing for broad structural diversity through simple substitution on the phenyl rings.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the protocol avoids sensitive conditions like nitrogen protection or cryogenic temperatures, making it highly amenable to commercial scale-up from gram levels to multi-ton production with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medicines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of trifluoromethyl 1,2,4-triazine meets the highest international standards. Our commitment to excellence makes us the preferred partner for companies seeking a reliable pharmaceutical intermediate supplier.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our advanced synthesis capabilities can accelerate your drug development timeline. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized processes can reduce your overall manufacturing expenses. We are ready to provide specific COA data and route feasibility assessments to support your next project milestone.
