Advanced D-Ribose Based Manufacturing Strategy for High-Purity Sofosbuvir Intermediates
Advanced D-Ribose Based Manufacturing Strategy for High-Purity Sofosbuvir Intermediates
The global pharmaceutical landscape for Hepatitis C treatment has been fundamentally transformed by the introduction of direct-acting antivirals, with Sofosbuvir standing as a cornerstone molecule in this therapeutic class. As demand for high-quality generic versions and novel derivatives continues to surge, the efficiency of the underlying synthetic routes becomes a paramount concern for industry stakeholders. Patent CN106565805B, published in April 2020, introduces a significant technological advancement by detailing a preparation method that utilizes D-ribose as the primary starting material. This approach represents a strategic departure from traditional pathways that often rely on more expensive and less accessible uridine derivatives. By leveraging the natural abundance and low cost of D-ribose, this methodology addresses critical bottlenecks in the upstream supply chain, offering a viable solution for manufacturers seeking to optimize their production economics without compromising on the stringent purity standards required for antiviral APIs.
For R&D directors and process chemists, the significance of this patent lies in its elegant simplification of the molecular construction. The disclosed method navigates the complex stereochemical requirements of the Sofosbuvir molecule through a series of well-defined transformations, including esterification, hydroxyl protection, selective oxidation, and fluorination. The use of D-ribose allows for a more linear progression towards the target 2'-deoxy-2'-fluoro-2'-C-methyluridine scaffold. This linearity is crucial for minimizing the accumulation of impurities that often plague convergent syntheses. Furthermore, the patent outlines specific reaction conditions, such as the use of TEMPO for oxidation and DAST for fluorination, which are known for their reliability in industrial settings. The integration of these established reagents within a novel sequence demonstrates a pragmatic approach to process development, prioritizing robustness and reproducibility alongside chemical innovation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sofosbuvir and its key intermediates has been fraught with challenges related to raw material availability and process complexity. Traditional routes frequently commence with uridine or its derivatives, which are inherently more costly and subject to greater supply chain volatility compared to simple sugars. These conventional pathways often necessitate a cumbersome series of protection and deprotection steps to differentiate between the multiple hydroxyl groups present on the ribose ring. Each additional step not only increases the overall processing time and labor costs but also inevitably leads to a decrement in overall yield due to material losses during isolation and purification. Moreover, the harsh conditions sometimes required for modifying the nucleobase or the sugar moiety in these older methods can lead to the formation of difficult-to-remove impurities, complicating the downstream purification process and potentially impacting the safety profile of the final drug product.
The Novel Approach
In stark contrast, the methodology described in CN106565805B capitalizes on the structural simplicity of D-ribose to streamline the entire synthetic sequence. By starting with this inexpensive pentose sugar, the process effectively bypasses the need for expensive nucleoside starting materials, immediately establishing a significant cost advantage at the very beginning of the manufacturing chain. The strategy employs a logical sequence of functional group manipulations, beginning with the formation of a methyl glycoside to lock the anomeric configuration, followed by the installation of a robust silyl protecting group that shields the 3 and 5 positions. This protection strategy is particularly advantageous as it allows for highly selective reactions at the 2-position, facilitating the subsequent oxidation and methylation steps with minimal side reactions. The result is a cleaner reaction profile and a more efficient workflow that is inherently more suitable for large-scale industrial application.
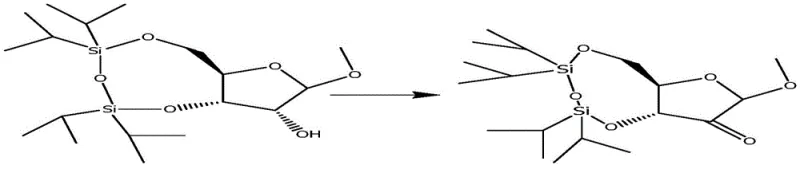
The core of this novel approach is exemplified by the selective oxidation step, where the secondary hydroxyl group is converted into a ketone functionality. As illustrated in the reaction scheme, the use of TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) in conjunction with sodium hypochlorite provides a mild yet effective means of achieving this transformation. This catalytic oxidation is highly desirable in a commercial setting because it avoids the use of toxic heavy metal oxidants like chromium or manganese, aligning with modern green chemistry principles. The resulting ketone intermediate serves as a pivotal junction in the synthesis, enabling the subsequent introduction of the methyl group and the fluorine atom with precise stereochemical control. This level of control is essential for ensuring that the final Sofosbuvir product possesses the correct biological activity, as the stereochemistry at the 2' position is critical for its interaction with the viral polymerase.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Fluorination
The mechanistic elegance of this synthesis is particularly evident in the oxidation and fluorination stages, which dictate the stereochemical outcome of the molecule. The TEMPO-mediated oxidation proceeds through a radical mechanism where the oxoammonium cation acts as the active oxidant, selectively abstracting a hydride from the secondary alcohol. This process is highly chemoselective, leaving the acid-labile glycosidic bond and the silyl ether protections intact, which is a testament to the mildness of the reaction conditions. Following the formation of the ketone, the addition of methyl magnesium chloride introduces the 2'-C-methyl group. This Grignard addition creates a tertiary alcohol, setting the stage for the crucial fluorination step. The subsequent reaction with DAST (Diethylaminosulfur trifluoride) involves the activation of the hydroxyl group followed by nucleophilic attack by fluoride, proceeding with inversion of configuration or retention depending on the specific mechanistic pathway, ultimately securing the vital 2'-fluoro-2'-methyl motif.
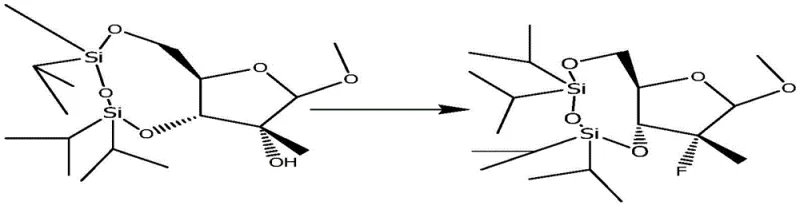
Impurity control is a major focus in this patented process, particularly regarding the removal of residual metals and side products from the coupling reactions. The use of tin tetrachloride in the coupling of the sugar moiety with the cytosine base requires careful workup procedures to ensure that tin residues are reduced to acceptable levels for pharmaceutical use. The patent details specific extraction and washing protocols, utilizing aqueous bicarbonate and brine washes to effectively partition organic products from inorganic salts and metal complexes. Furthermore, the final recrystallization steps are optimized to exclude diastereomeric impurities that may arise from the non-stereospecific nature of certain steps, such as the Grignard addition. By rigorously controlling these parameters, the process ensures that the intermediate 2-methyl-2-fluoro-beta-D-cytidine is obtained with high purity, which is a prerequisite for the success of the final phosphorylation step.
How to Synthesize Sofosbuvir Efficiently
The synthesis of Sofosbuvir via this D-ribose route offers a clear roadmap for process chemists aiming to establish a robust manufacturing line. The procedure begins with the conversion of D-ribose to methyl-beta-D-nucleoside using anhydrous methanol and acetyl chloride, a straightforward esterification that sets the anomeric center. Following this, the diol protection with TIPDSCl creates a rigid scaffold that directs subsequent reactivity. The detailed experimental section provides precise molar ratios and temperature controls, such as maintaining the oxidation reaction between -5°C and 0°C to prevent over-oxidation or degradation. These operational details are critical for scaling the process from the laboratory bench to pilot plant reactors, ensuring that heat transfer and mixing efficiencies do not compromise the reaction selectivity.
- Esterify D-ribose with methanol and acetyl chloride to form methyl-beta-D-nucleoside, followed by silyl protection using TIPDSCl.
- Perform selective TEMPO-mediated oxidation to generate the 2-oxo intermediate, followed by Grignard methylation and DAST fluorination.
- Couple the fluorinated sugar with protected cytosine, deprotect the silyl groups, and finalize with phosphoramidate coupling to yield Sofosbuvir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this D-ribose based synthesis route presents compelling economic and logistical benefits. The primary advantage stems from the substitution of expensive, specialized nucleoside starting materials with D-ribose, a commodity chemical produced on a massive scale for the food and beverage industry. This shift drastically reduces the raw material cost basis, insulating the manufacturing process from the price volatility often seen in the fine chemical sector. Additionally, the reduction in the total number of synthetic steps translates directly into lower operational expenditures, as fewer unit operations mean reduced consumption of solvents, reagents, and energy. The simplified workflow also shortens the overall production cycle time, allowing for faster turnaround from raw material intake to finished API, which is a critical metric for maintaining lean inventory levels and responding swiftly to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of costly uridine derivatives and the minimization of protection-deprotection cycles lead to substantial savings in direct material costs. By utilizing common reagents like TEMPO and DAST which are available from multiple global suppliers, the process avoids reliance on single-source proprietary catalysts, fostering a competitive bidding environment for inputs. The high yields reported in key steps, such as the 95% yield in the deprotection stage, further enhance the overall mass balance, reducing waste disposal costs and maximizing the output per batch. This economic efficiency is vital for maintaining profitability in the highly competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Sourcing D-ribose is significantly more secure than sourcing complex nucleoside intermediates, as it is derived from renewable biomass and produced by numerous manufacturers worldwide. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic high-pressure or cryogenic setups, ensures that the technology can be transferred easily between different manufacturing sites. This flexibility is invaluable for building a resilient supply chain capable of withstanding geopolitical disruptions or logistical bottlenecks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction types that are well-understood and easily managed in large-scale reactors. The avoidance of heavy metal oxidants in favor of catalytic TEMPO systems aligns with increasingly stringent environmental regulations regarding heavy metal discharge. This 'greener' profile simplifies the permitting process for new manufacturing facilities and reduces the long-term liability associated with hazardous waste management. The ability to scale this process from 100 kgs to multi-ton annual production volumes without significant re-engineering makes it an ideal candidate for meeting the growing global demand for Hepatitis C therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from a detailed analysis of the patent specifications and are intended to provide clarity on the feasibility and advantages of the D-ribose route. Understanding these nuances is essential for stakeholders evaluating the potential for technology transfer or licensing agreements.
Q: Why is the D-ribose route preferred over the uridine route for Sofosbuvir production?
A: The D-ribose route utilizes a cheaper, more abundant starting material compared to uridine derivatives. It simplifies the synthetic pathway by reducing the number of protection and deprotection steps required, thereby lowering overall production costs and improving scalability.
Q: What are the critical quality control points in this synthesis method?
A: Critical control points include the stereoselectivity during the fluorination step using DAST and the purity of the final phosphoramidate coupling. The patent emphasizes rigorous TLC monitoring and recrystallization steps to ensure high optical purity and remove residual metal catalysts.
Q: How does this method impact the supply chain stability for Hepatitis C treatments?
A: By relying on commodity chemicals like D-ribose rather than specialized nucleosides, manufacturers can mitigate supply chain risks associated with raw material scarcity. The robust nature of the reaction conditions allows for consistent large-scale production, ensuring steady availability of the active pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sofosbuvir Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the D-ribose synthesis route for Sofosbuvir and are fully equipped to leverage this technology for our global partners. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, large-scale operations. We maintain stringent purity specifications across all our manufacturing lines, supported by rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Our commitment to quality assurance means that we can consistently deliver high-purity Sofosbuvir intermediates and APIs that meet the exacting standards of international regulatory bodies.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains and reduce their cost of goods sold. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of antiviral manufacturing and secure a reliable, cost-effective supply of this critical medicine for patients worldwide.
