Advanced Synthesis of 2-Deoxy-D-Glucose for Commercial Scale-Up and API Production
Advanced Synthesis of 2-Deoxy-D-Glucose for Commercial Scale-Up and API Production
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for critical active pharmaceutical ingredients (APIs) and their precursors. Patent CN102180914A introduces a transformative preparation method for 2-Deoxy-D-Glucose, a vital metabolite and building block for antiviral and anticancer therapeutics. This technology addresses the longstanding bottlenecks of traditional extraction methods, which suffer from low yields and complex purification requirements. By leveraging a selective addition reaction followed by efficient hydrolysis, this patent outlines a streamlined route that promises to enhance the reliability of the pharmaceutical intermediates supply chain. For R&D directors and procurement strategists, understanding this synthetic breakthrough is essential for securing long-term access to high-quality glycomimetics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-Deoxy-D-Glucose has been fraught with significant technical and economic challenges. Traditional approaches often rely on the extraction and separation of lactone antibiotics from plant and microbial cell membranes, a process that is inherently inefficient and resource-intensive. As illustrated in prior art such as the route described in IN187908, older chemical methods frequently involve cumbersome multi-step sequences, including transesterification and hydrolysis, which degrade overall yield and increase waste generation. 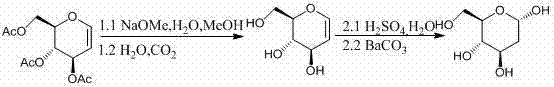 Furthermore, biological extraction methods are susceptible to seasonal variations and low natural abundance of precursors, leading to inconsistent supply and inflated costs. These conventional pathways often struggle with stereochemical control, risking racemization that compromises the biological activity of the final drug product, thereby necessitating expensive downstream purification efforts that erode profit margins.
Furthermore, biological extraction methods are susceptible to seasonal variations and low natural abundance of precursors, leading to inconsistent supply and inflated costs. These conventional pathways often struggle with stereochemical control, risking racemization that compromises the biological activity of the final drug product, thereby necessitating expensive downstream purification efforts that erode profit margins.
The Novel Approach
In stark contrast, the methodology disclosed in CN102180914A offers a streamlined, two-step chemical synthesis that bypasses the inefficiencies of extraction and complex multi-step derivatization. The core innovation lies in the direct selective addition reaction of acetylglucosene (3,4,6-tri-O-acetyl-D-glucal) to generate the key intermediate, 2-deoxy-1,3,4,6-tetra-O-acetyl-D-glucose. 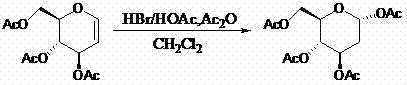 This approach utilizes readily available starting materials and common reagents like hydrogen bromide and acetic anhydride, drastically simplifying the operational workflow. By eliminating the need for rare natural extracts and reducing the number of unit operations, this novel approach not only accelerates production timelines but also significantly lowers the barrier to entry for commercial scale-up of complex sugar derivatives, making it an attractive option for cost reduction in API manufacturing.
This approach utilizes readily available starting materials and common reagents like hydrogen bromide and acetic anhydride, drastically simplifying the operational workflow. By eliminating the need for rare natural extracts and reducing the number of unit operations, this novel approach not only accelerates production timelines but also significantly lowers the barrier to entry for commercial scale-up of complex sugar derivatives, making it an attractive option for cost reduction in API manufacturing.
Mechanistic Insights into Selective Addition and Hydrolysis
The chemical elegance of this process is rooted in the precise control of regioselectivity during the addition phase. The reaction initiates with the electrophilic addition of hydrogen bromide across the double bond of the glucal derivative in the presence of acetic anhydride. This specific condition favors the formation of the alpha-anomer of the glycosyl bromide intermediate, which is subsequently trapped by acetate to form the stable tetra-acetylated product. The use of solvents such as dichloromethane or tetrahydrofuran at controlled temperatures ranging from 0°C to 40°C ensures that side reactions are minimized, preserving the integrity of the carbohydrate scaffold. This mechanistic precision is critical for R&D teams focused on impurity profiling, as it inherently limits the formation of undesired isomers that are difficult to separate later in the process.
Following the formation of the protected intermediate, the process employs a robust acid-catalyzed hydrolysis strategy to unveil the final free sugar. By treating the tetra-acetyl intermediate with mineral acids like hydrochloric acid or sulfuric acid at moderate temperatures between 20°C and 100°C, the ester bonds are cleaved efficiently without degrading the sensitive glycosidic linkage. 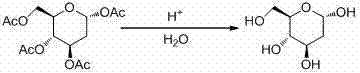 The subsequent neutralization and recrystallization steps are designed to maximize recovery while ensuring the removal of residual salts and solvents. This hydrolysis mechanism is particularly advantageous because it avoids harsh conditions that could lead to decomposition, thereby supporting the production of high-purity 2-Deoxy-D-Glucose that meets stringent pharmacopeial standards for clinical applications.
The subsequent neutralization and recrystallization steps are designed to maximize recovery while ensuring the removal of residual salts and solvents. This hydrolysis mechanism is particularly advantageous because it avoids harsh conditions that could lead to decomposition, thereby supporting the production of high-purity 2-Deoxy-D-Glucose that meets stringent pharmacopeial standards for clinical applications.
How to Synthesize 2-Deoxy-D-Glucose Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and temperature control to replicate the high yields reported in the patent examples. The process begins with the dissolution of acetylglucosene in a suitable organic solvent, followed by the sequential addition of the brominating agent and acetylating agent under inert conditions. Detailed standard operating procedures regarding mixing times, quenching protocols, and crystallization solvent ratios are critical for reproducibility. For a comprehensive guide on executing this pathway with optimal safety and efficiency, please refer to the standardized synthesis steps outlined below.
- Perform selective addition reaction of acetylglucosene with HBr/AcOH and acetic anhydride at 0-40°C to form 2-deoxy-1,3,4,6-tetra-O-acetyl-D-glucose.
- Conduct acid-catalyzed hydrolysis of the intermediate using HCl or H2SO4 at 20-100°C to remove acetyl groups.
- Neutralize, extract, and recrystallize the final product to obtain high-purity 2-Deoxy-D-Glucose.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards this synthetic route represents a strategic opportunity to de-risk the supply of critical oncology and antiviral intermediates. The reliance on commodity chemicals rather than scarce biological extracts fundamentally alters the cost structure and availability profile of the material. By adopting this method, organizations can mitigate the volatility associated with agricultural sourcing and achieve a more predictable manufacturing cadence. The simplicity of the workup procedures further translates into reduced processing time and lower utility consumption, contributing to substantial cost savings in pharmaceutical intermediates manufacturing without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of expensive enzymatic steps or complex extraction protocols directly reduces the bill of materials. Since the reagents involved, such as acetic anhydride and hydrobromic acid, are bulk industrial chemicals, their procurement is straightforward and economically favorable. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting material to byproducts, effectively increasing the mass balance efficiency. This logical deduction of cost benefits suggests a significantly improved margin profile for producers who adopt this technology, allowing for more competitive pricing in the global market for sugar-based therapeutics.
- Enhanced Supply Chain Reliability: Dependence on plant-derived lactone antibiotics creates a fragile supply chain vulnerable to harvest failures and geopolitical instability. In contrast, acetylglucosene is a stable, synthetically accessible commodity that can be sourced from multiple established chemical suppliers globally. This diversification of raw material sources ensures continuous production capability even during external disruptions. Additionally, the robust nature of the chemical steps means that production can be easily shifted between different manufacturing sites with minimal requalification effort, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent delivery to downstream drug manufacturers.
- Scalability and Environmental Compliance: The reaction conditions described operate within standard temperature and pressure ranges, removing the need for specialized high-pressure reactors or cryogenic infrastructure. This accessibility facilitates seamless commercial scale-up of complex sugar derivatives from pilot plants to multi-ton production facilities. Moreover, the process generates less hazardous waste compared to heavy metal-catalyzed alternatives, aligning with modern green chemistry principles. The ability to recycle solvents and the use of aqueous workups simplify waste treatment protocols, ensuring that the manufacturing process remains environmentally compliant and sustainable over the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on feasibility and performance. Understanding these details is crucial for technical teams evaluating the integration of this process into existing production lines.
Q: How does this synthesis method improve upon traditional extraction processes?
A: Unlike traditional methods relying on low-yield extraction from natural lactone antibiotics, this chemical synthesis uses readily available acetylglucosene, offering significantly higher yields, better selectivity, and independence from biological source variability.
Q: What are the key purity advantages for pharmaceutical applications?
A: The process utilizes specific recrystallization steps and controlled hydrolysis conditions that minimize racemization and impurity formation, ensuring the high-purity 2-Deoxy-D-Glucose required for sensitive antiviral and anticancer drug formulations.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions (0-40°C for addition, 20-100°C for hydrolysis) are mild and utilize common industrial solvents and reagents, facilitating easy commercial scale-up of complex sugar derivatives without requiring exotic catalysts or extreme pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Deoxy-D-Glucose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis plays in the development of next-generation antiviral and anticancer therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory concept to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-Deoxy-D-Glucose meets the exacting standards required for clinical-grade applications. Our commitment to technical excellence ensures that we can handle the complexities of carbohydrate chemistry with precision and reliability.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug development needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for 2-Deoxy-D-Glucose and related pharmaceutical intermediates.
