Advanced Synthesis of 2-Deoxy-D-Glucose for Commercial API Manufacturing
Advanced Synthesis of 2-Deoxy-D-Glucose for Commercial API Manufacturing
The pharmaceutical and biotechnology sectors are increasingly demanding high-purity intermediates for the development of next-generation antiviral and anticancer therapeutics. Among these critical building blocks, 2-deoxy-D-glucose stands out as a vital metabolic inhibitor and viral envelope component disruptor. As detailed in Chinese Patent CN100572387C, a novel chemical synthesis pathway has been established that fundamentally shifts the production paradigm from inefficient natural extraction to a robust, scalable chemical process. This innovation addresses the longstanding bottlenecks of low yield and high cost associated with legacy methods, providing a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates. By leveraging abundant marine biomass derivatives, this technology offers a sustainable route that aligns with modern green chemistry principles while ensuring supply chain continuity for global drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of 2-deoxy-D-glucose has been plagued by significant technical and economic hurdles that hinder its widespread application in large-scale API manufacturing. Traditional methods often relied on the extraction and separation of lactone antibiotics from plant or microbial cell membranes, a process inherently limited by the low natural abundance of these precursors. Furthermore, early chemical syntheses, such as those reported by Bergmann or Sowden, utilized hazardous reagents like nitromethane or suffered from extremely low yields due to unavoidable side reactions and difficult purification steps. These legacy processes not only incurred substantial environmental costs through toxic waste generation but also failed to deliver the consistent stereochemical purity required for clinical-grade applications. The reliance on scarce natural sources or dangerous synthetic routes created a fragile supply chain, characterized by volatile pricing and unpredictable lead times, making it difficult for procurement managers to secure long-term contracts for essential drug ingredients.
The Novel Approach
The methodology outlined in the patent introduces a transformative strategy that utilizes glucosamine hydrochloride, readily derived from shrimp and crab shell waste, as the primary starting material. This approach bypasses the limitations of natural extraction by employing a rational, multi-step organic synthesis that ensures high atom economy and operational simplicity. The process involves a clever sequence of protection, functional group transformation, and deprotection that maintains the integrity of the sugar backbone while precisely modifying the 2-position. By shifting the feedstock to an abundant, low-cost marine byproduct, the novel method drastically simplifies the upstream supply chain and reduces the overall cost of goods sold. This transition from scarcity-driven sourcing to abundance-driven synthesis represents a critical advancement for cost reduction in pharmaceutical intermediate manufacturing, enabling producers to meet the growing global demand for antiviral and oncology drugs without compromising on quality or sustainability.
Mechanistic Insights into Diazotization and Reductive Dehalogenation
The core innovation of this synthesis lies in the precise manipulation of the amino group at the C-2 position, achieved through a sophisticated diazotization and reductive dechlorination sequence. Initially, the amino group of glucosamine is protected as a Schiff base using benzaldehyde, which prevents unwanted side reactions during the subsequent acetylation of the hydroxyl groups. Following selective hydrolysis to regenerate the free amine, the molecule undergoes a critical Sandmeyer-type reaction where the amino group is converted into a diazonium salt using sodium nitrite and hydrochloric acid at low temperatures. This unstable intermediate is immediately trapped by cuprous chloride to form a chloro-derivative, effectively replacing the nitrogen functionality with a chlorine atom while preserving the stereochemistry of the adjacent chiral centers.
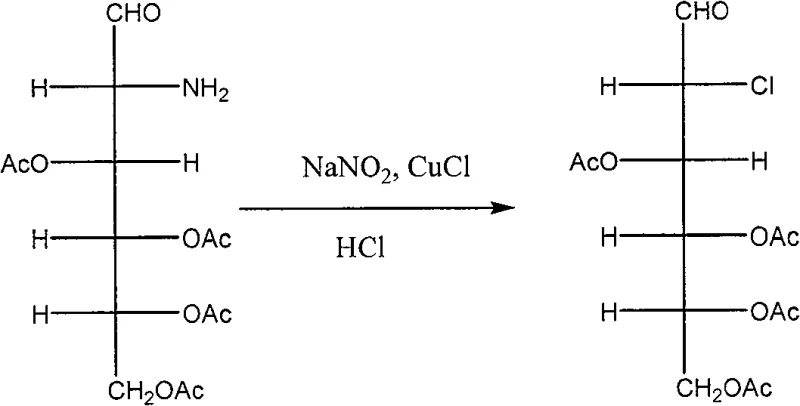
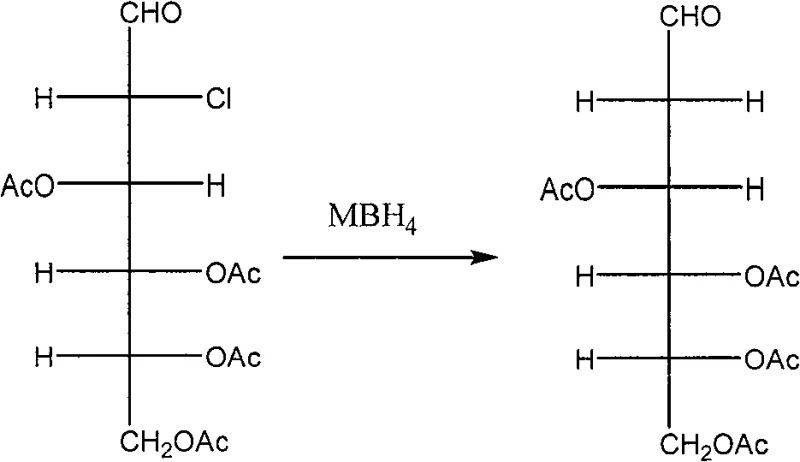
Subsequent to the chlorination step, the process employs a mild yet effective reductive dehalogenation using metal borohydrides, such as sodium borohydride, under controlled alkaline conditions. This step is pivotal as it removes the chlorine atom at the 2-position to generate the desired deoxy structure without affecting the acetyl protecting groups on the remaining hydroxyls. The use of borohydride ensures a clean reduction profile, minimizing the formation of elimination byproducts that could compromise the purity of the final API. Finally, a global deacetylation under basic conditions reveals the free hydroxyl groups, yielding the target 2-deoxy-D-glucose. This mechanistic pathway demonstrates exceptional control over regioselectivity and chemoselectivity, ensuring that the final product meets the rigorous impurity profiles demanded by regulatory agencies for human therapeutic use.
How to Synthesize 2-Deoxy-D-Glucose Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly temperature control during the diazotization phase and stoichiometry during the reduction step. The patent provides a comprehensive framework for operators to follow, emphasizing the importance of maintaining specific pH levels and molar ratios to maximize yield and minimize waste. For R&D teams looking to implement this technology, the process is designed to be modular, allowing for optimization at each stage from the initial Schiff base formation to the final crystallization. Detailed standardized operating procedures regarding solvent selection, reaction times, and workup protocols are essential to replicate the high success rates reported in the experimental examples. Please refer to the structured guide below for the specific operational parameters required for successful implementation.
- Protect the amino group of glucosamine hydrochloride using benzaldehyde under alkaline conditions to form the Schiff base.
- Perform complete acetylation of the hydroxyl groups using acetic anhydride and a base catalyst.
- Hydrolyze the Schiff base selectively under acidic conditions to regenerate the free amino group.
- Execute diazotization followed by chlorination using sodium nitrite and cuprous chloride to replace the amino group with chlorine.
- Reduce the chloro-derivative using a borohydride reagent to remove the chlorine atom at the 2-position.
- Final deacetylation via alkaline hydrolysis to yield pure 2-deoxy-D-glucose.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers profound strategic benefits that extend far beyond simple unit cost metrics. By decoupling production from the volatility of natural extraction and the hazards of obsolete chemical methods, organizations can secure a more resilient and predictable supply of this critical intermediate. The utilization of glucosamine hydrochloride, a commodity chemical derived from the massive global seafood industry, ensures that raw material availability is virtually unlimited and price-stable. This shift mitigates the risk of supply disruptions caused by seasonal variations in biological sources or geopolitical issues affecting niche chemical suppliers, thereby enhancing the overall reliability of the pharmaceutical manufacturing pipeline.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of expensive, scarce starting materials with low-cost, abundant marine byproducts. The elimination of complex extraction protocols and the reduction in hazardous waste disposal costs contribute to a significantly leaner cost structure. Furthermore, the high efficiency of the reaction sequence minimizes the loss of valuable intermediates, leading to substantial overall yield improvements that directly translate to lower production costs per kilogram. This economic efficiency allows manufacturers to offer competitive pricing without sacrificing margin, creating a strong value proposition for downstream API producers seeking to optimize their bill of materials.
- Enhanced Supply Chain Reliability: Sourcing raw materials from the established crustacean processing industry provides a robust buffer against market fluctuations that typically plague specialty chemical supply chains. The scalability of the synthesis is inherently supported by the vast availability of shrimp and crab shells, ensuring that production can be ramped up rapidly to meet surges in demand for antiviral or cancer therapies. This reliability is crucial for maintaining continuous manufacturing operations and avoiding costly downtime, giving supply chain heads the confidence to commit to long-term delivery schedules with their own customers.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common solvents and reagents that are easily handled in standard stainless steel reactors. The avoidance of highly toxic reagents like nitromethane and the implementation of solvent recovery loops align the process with strict environmental regulations, reducing the burden of compliance and waste management. This environmental stewardship not only lowers operational risks but also enhances the corporate sustainability profile of the manufacturer, a factor that is increasingly important for multinational corporations adhering to ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-deoxy-D-glucose synthesized via this patented method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance and feasibility. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their own drug development pipelines.
Q: What are the primary advantages of this synthesis route over traditional extraction methods?
A: Traditional extraction from natural sources suffers from low yields, complex purification, and potential racemization. This novel chemical synthesis utilizes abundant glucosamine hydrochloride derived from crustacean waste, offering a more consistent, scalable, and cost-effective supply chain solution with higher product purity.
Q: How does the process ensure high stereochemical purity?
A: The route employs specific protection and deprotection strategies, including benzylidene shielding and controlled acidic hydrolysis, which minimize side reactions and prevent racemization at the chiral centers, ensuring the final product meets stringent pharmaceutical specifications.
Q: Is the raw material supply stable for large-scale production?
A: Yes, the starting material, glucosamine hydrochloride, is sourced from shrimp and crab shells, which are abundant byproducts of the seafood industry. This ensures a robust, renewable, and economically stable raw material base compared to scarce natural extracts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Deoxy-D-Glucose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By partnering with us, you gain access to a supply chain that is both technically superior and commercially robust, designed to support your long-term growth in the competitive pharmaceutical landscape.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project needs. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to our optimized supply model. Our experts are ready to provide specific COA data and route feasibility assessments to help you make informed decisions that drive efficiency and innovation in your manufacturing operations.
