Advanced Synthesis of 2-Deoxy-D-Glucose: Scalable Routes for Pharmaceutical Applications
Advanced Synthesis of 2-Deoxy-D-Glucose: Scalable Routes for Pharmaceutical Applications
The pharmaceutical landscape is constantly evolving, demanding more efficient and cost-effective pathways for critical active pharmaceutical ingredients (APIs) and intermediates. A pivotal advancement in this domain is detailed in patent CN1775791A, which outlines a robust novel method for preparing 2-deoxy-D-glucose (2-DG). This compound is increasingly recognized for its vital role in antiviral therapies, cancer research, and metabolic studies, acting as a glucose antimetabolite that interferes with glycoprotein synthesis in viruses and tumor cells. The disclosed technology addresses longstanding challenges in the industry, specifically the high production costs and complex purification steps associated with traditional extraction from natural sources or older synthetic routes. By leveraging glucosamine hydrochloride—a readily available derivative of chitin found in crustacean shells—this method offers a sustainable and economically superior alternative. For R&D directors and procurement specialists seeking a reliable 2-deoxy-D-glucose supplier, understanding the mechanistic elegance and commercial viability of this patent is essential for securing a stable supply chain for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-deoxy-D-glucose has been plagued by significant technical and economic hurdles that hindered its widespread application in bulk drug manufacturing. Early methods, such as those reported by Bergmann and Sowden in the early to mid-20th century, relied on the acid-catalyzed addition of water to glycals or the extension of carbon chains using nitromethane. These processes were characterized by extremely low yields, often necessitating complex chromatographic separations that are impractical for industrial scale-up. Furthermore, many conventional routes utilized toxic reagents and harsh conditions that led to unavoidable side reactions, resulting in poor product purity and significant racemization issues. The reliance on natural extraction from plant or microbial cell membranes also presented a bottleneck, as the content of the target lactone microbiotic in natural sources is inherently low, making it impossible to satisfy the day-by-day enlarging market requirement for antiviral and anticancer agents. These inefficiencies translated directly into prohibitive costs and supply chain volatility, limiting the clinical potential of 2-DG based medicines.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN1775791A introduces a rational, step-wise synthetic strategy that maximizes atom economy and operational simplicity. The core innovation lies in the utilization of glucosamine hydrochloride as the chiral starting material, which is not only inexpensive but also derived from renewable biomass waste, aligning with modern green chemistry principles. The process involves a clever sequence of selective protection, functional group transformation, and deprotection. Initially, the amino group is masked as a Schiff base (imine) with benzaldehyde, allowing for the subsequent per-acetylation of all hydroxyl groups without interference. This strategic protection ensures that subsequent reactions occur selectively at the C2 position. The transformation of the C2-amino group into a chlorine atom via diazotization, followed by reductive dechlorination, provides a clean and high-yielding route to the deoxy sugar skeleton. This approach effectively eliminates the side reactions and racemization risks associated with older methods, delivering a high-purity product suitable for stringent pharmaceutical applications.
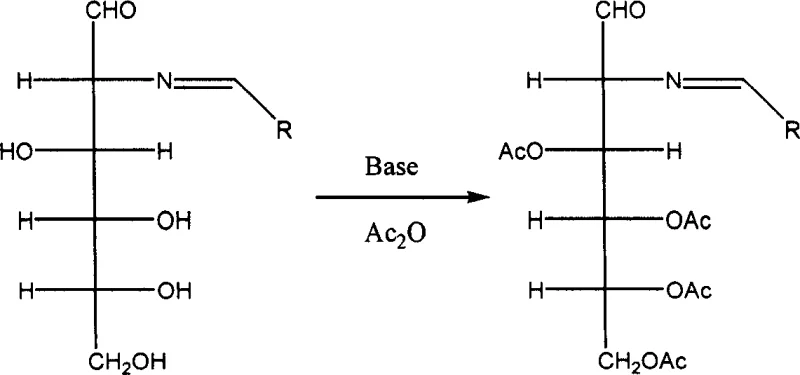
The initial stages of the synthesis demonstrate the power of orthogonal protection strategies in carbohydrate chemistry. As illustrated in the reaction scheme, the formation of the Schiff base protects the amine, while the subsequent treatment with acetic anhydride and a base catalyst efficiently acetylates the hydroxyl groups at positions 3, 4, 5, and 6. This step is critical because it solubilizes the intermediate and prevents unwanted side reactions at the hydroxyl sites during the subsequent harsh diazotization conditions. The use of mild bases like sodium acetate or triethylamine ensures that the acid-sensitive glycosidic bonds remain intact while driving the acetylation to completion. This level of control is paramount for achieving the high purity specifications required for a reliable pharmaceutical intermediates supplier, as it minimizes the formation of difficult-to-remove impurities that often plague sugar chemistry.
Mechanistic Insights into Diazotization and Reductive Dechlorination
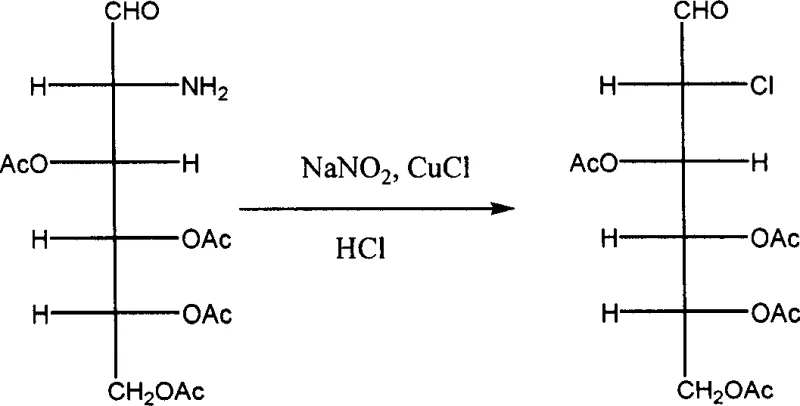
The heart of this synthetic route lies in the precise manipulation of the C2 functionality, transforming an amino group into a hydrogen atom through a chloro-intermediate. This is achieved via a Sandmeyer-type reaction sequence that is both elegant and highly effective. The process begins with the hydrolysis of the Schiff base to reveal the free amine, which is then subjected to diazotization using sodium nitrite and hydrochloric acid at low temperatures (typically -5 to 5°C). The resulting diazonium salt is unstable and is immediately treated with cuprous chloride (CuCl). This step replaces the diazonium group with a chlorine atom, yielding 2-deoxy-2-chloro-3,4,5,6-tetra-O-acetyl-D-glucose. The stereochemical outcome of this substitution is carefully controlled by the reaction conditions, ensuring the retention of the desired configuration necessary for the biological activity of the final 2-DG product.
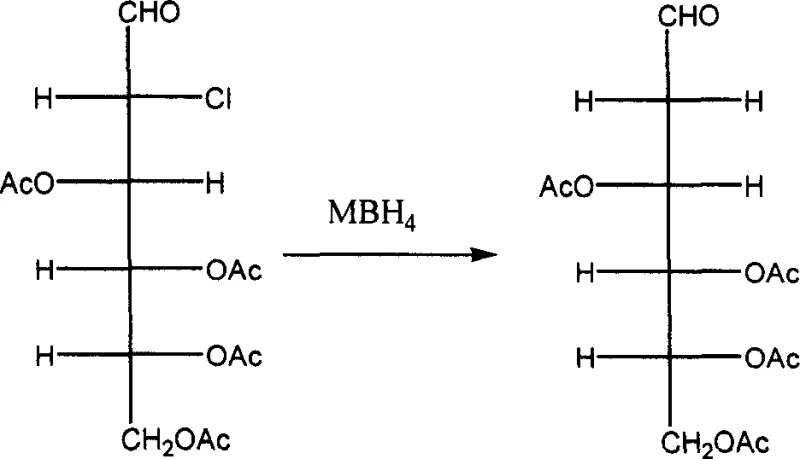
Following the introduction of the chlorine atom, the final deoxygenation is accomplished through a reductive dechlorination step. The chloro-intermediate is treated with a borohydride reducing agent, such as sodium borohydride or potassium borohydride, in a basic solvent system. This reduction cleaves the carbon-chlorine bond and replaces it with a hydrogen atom, effectively completing the "deoxy" transformation. The mechanism likely involves a radical or hydride transfer process that is highly selective for the C2-chlorine bond over the acetate esters, thanks to the carefully tuned pH and temperature conditions (0 to 10°C). This selectivity is crucial; it prevents the premature removal of the acetyl protecting groups, which are only removed in the final step via alkaline hydrolysis. By segregating the deprotection steps, the process ensures that the sugar ring remains stable throughout the synthesis, minimizing degradation and maximizing overall yield. This mechanistic precision is what allows for the commercial scale-up of complex sugar derivatives with consistent quality.
How to Synthesize 2-Deoxy-D-Glucose Efficiently
The synthesis of 2-deoxy-D-glucose via this patented route requires strict adherence to reaction parameters to ensure optimal yield and purity. The process is designed to be operationally simple, utilizing standard unit operations common in fine chemical manufacturing, such as filtration, extraction, and recrystallization. The detailed standardized synthesis steps involve precise stoichiometry, particularly in the diazotization step where the ratio of nitrite to amine must be carefully controlled to prevent the formation of byproducts. The subsequent reduction and deprotection steps also require careful monitoring of pH and temperature to avoid epimerization or degradation of the sensitive carbohydrate backbone. For a comprehensive guide on the exact molar ratios, solvent choices, and workup procedures, please refer to the technical protocol below.
- Protect hydroxyl groups via acetylation and form a Schiff base with benzaldehyde to mask the amino group.
- Perform diazotization followed by chlorination using cuprous chloride to replace the amino group with chlorine.
- Execute reductive dechlorination using borohydride to remove the chlorine atom, followed by final deacetylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards this glucosamine-based synthesis represents a strategic opportunity to optimize costs and secure supply continuity. Traditional methods relying on fermentation or extraction are inherently limited by biological variability and low titers, leading to volatile pricing and potential shortages. In contrast, this chemical synthesis utilizes commodity chemicals and biomass-derived starting materials that are available in vast quantities globally. The decoupling of production from biological sources mitigates the risk of supply disruptions caused by seasonal variations or agricultural issues. Furthermore, the streamlined nature of the synthesis, which avoids exotic catalysts or extreme high-pressure conditions, translates directly into lower capital expenditure (CAPEX) and operating expenditure (OPEX) for manufacturing partners. This efficiency enables a more competitive pricing structure for the final API, facilitating cost reduction in pharmaceutical intermediates manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this route is the raw material selection. Glucosamine hydrochloride is a low-cost commodity derived from the processing of shrimp and crab shells, which are abundant waste products of the seafood industry. By valorizing this waste stream, the process achieves a significantly lower input cost compared to synthetic starting materials derived from petroleum or complex fermentation broths. Additionally, the high selectivity of the reaction sequence reduces the burden on downstream purification. Fewer side reactions mean less solvent consumption for chromatography and higher recovery rates during crystallization. The elimination of expensive transition metal catalysts (beyond the stoichiometric copper used in the Sandmeyer step, which is recoverable) further lowers the material bill. These factors combine to create a robust economic model that supports substantial cost savings over the product lifecycle.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical for life-saving medications, and this synthetic route offers distinct advantages in terms of reliability. Because the starting materials are non-perishable chemicals and biomass derivatives with established global supply networks, manufacturers are not subject to the lead times associated with growing biological cultures. The synthetic steps are robust and can be performed in standard stainless steel reactors, allowing for flexible production scheduling. This flexibility means that production capacity can be ramped up quickly to meet surges in demand, such as during viral outbreaks where 2-DG based therapeutics might be critical. The ability to source raw materials from multiple geographic regions further diversifies risk, ensuring that the supply of high-purity 2-deoxy-D-glucose remains uninterrupted even if one region faces logistical challenges.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is well-suited for modern regulatory environments. The reactions proceed in common organic solvents like methanol, ethanol, and ethyl acetate, which are easier to recover and recycle compared to chlorinated solvents often used in older carbohydrate chemistry. The aqueous workups and crystallization steps minimize the generation of hazardous waste streams. Moreover, the process avoids the use of heavy metals that are difficult to purge from the final product, simplifying the compliance burden for residual solvent and metal impurity testing. The scalability is evidenced by the use of standard exothermic control measures (e.g., cooling for diazotization) that are easily managed in large-scale reactors. This makes the commercial scale-up of complex sugar derivatives feasible and environmentally sustainable, aligning with the increasing pressure on the pharmaceutical industry to adopt greener manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-deoxy-D-glucose using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of this route for potential partners. Understanding these details is crucial for evaluating the technical readiness and commercial potential of integrating this supply source into your portfolio.
Q: What is the primary advantage of using glucosamine hydrochloride as a starting material?
A: Glucosamine hydrochloride is derived from abundant crustacean waste like shrimp and crab shells, offering a significantly lower cost basis compared to traditional fermentation or extraction methods, while ensuring a consistent chiral pool source.
Q: How does this process control stereochemistry at the C2 position?
A: The process utilizes a diazotization-chlorination sequence followed by reductive dechlorination. This specific pathway allows for the clean removal of the C2-amino group without inducing racemization, preserving the D-configuration essential for biological activity.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the patent describes conditions using common industrial reagents like acetic anhydride and sodium borohydride, with straightforward workup procedures such as filtration and recrystallization, indicating high feasibility for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Deoxy-D-Glucose Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN1775791A for the global supply of 2-deoxy-D-glucose. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of this critical intermediate. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, including detailed impurity profiling to ensure the safety and efficacy of the final drug product. We understand that the transition from lab-scale patent examples to industrial reality requires deep process engineering expertise, which is exactly what our technical team brings to every project. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and compliant with international regulatory standards.
We invite you to collaborate with us to optimize your supply chain for 2-deoxy-D-glucose and related antiviral intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Whether you are in the early stages of drug development or looking to secure a long-term commercial supply, NINGBO INNO PHARMCHEM is committed to delivering excellence in every batch.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
