Advanced Copper-Catalyzed Synthesis of Omacycline Intermediates for Commercial Scale-up
Introduction to Advanced Omacycline Synthesis
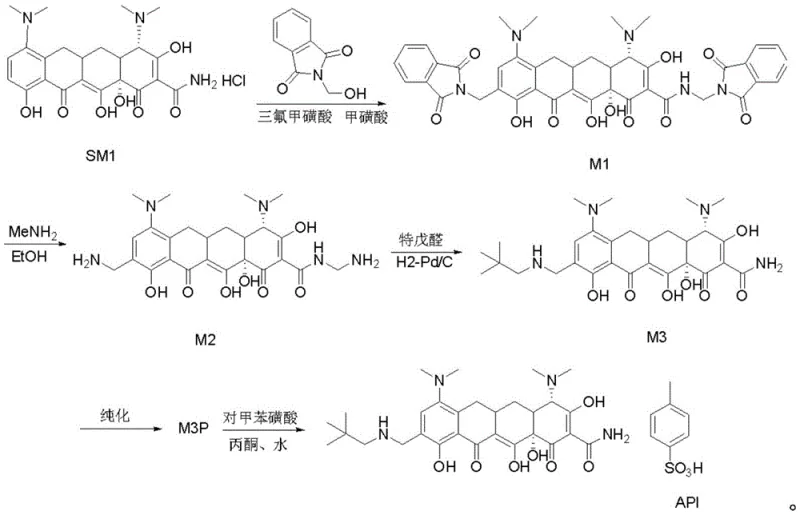
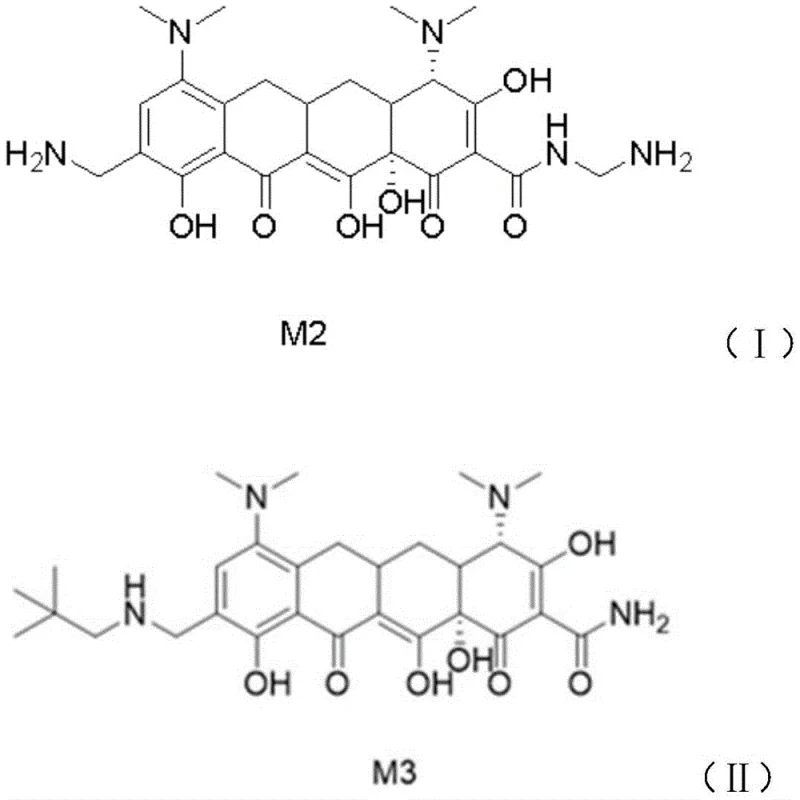
The pharmaceutical industry constantly seeks robust methodologies for synthesizing complex tetracycline derivatives, particularly novel agents like omacycline which target community-acquired pneumonia and acute skin infections. A pivotal advancement in this domain is detailed in patent CN111484424A, which discloses a highly efficient method for synthesizing omacycline intermediates. This technology addresses critical bottlenecks in traditional manufacturing, specifically focusing on the conversion of intermediate M2 to M3. By integrating a specialized copper salt additive into the hydrogenation system, the process achieves superior stereochemical control, effectively suppressing the formation of detrimental beta-isomers. For global supply chain leaders and R&D directors, this represents a significant leap forward in process chemistry, offering a pathway to higher purity active pharmaceutical ingredients (APIs) while simultaneously addressing the escalating costs associated with solvent waste and prolonged processing times in large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis and purification of omacycline intermediates have been plagued by inefficiencies that severely impact commercial viability. The prior art typically relies on preparative high-performance liquid chromatography (HPLC) for the purification of the M3 intermediate. This approach is inherently resource-intensive, requiring vast quantities of organic solvents such as acetonitrile, dichloromethane, and n-heptane to achieve acceptable separation. Furthermore, the conventional synthesis of M3 from M2 often suffers from poor stereocontrol, leading to high levels of beta-isomer impurities ranging from 3.0% to 4.0%. These impurities are notoriously difficult to remove in downstream processing, necessitating repeated purification cycles that extend the total production time to approximately 72 hours. From a supply chain perspective, this reliance on chromatographic separation creates a bottleneck, limiting throughput and generating substantial hazardous waste that complicates environmental compliance and increases disposal costs for manufacturing sites.
The Novel Approach
The innovative methodology presented in the patent data fundamentally reengineers the synthesis landscape by replacing chromatographic purification with a streamlined crystallization protocol. Central to this breakthrough is the introduction of a soluble copper salt, such as copper chloride, directly into the hydrogenation reaction system. This modification not only catalyzes the desired transformation but also acts as a stereochemical director, locking the molecular conformation to prevent isomerization. Consequently, the beta-isomer content is drastically reduced to between 1.0% and 2.0%, simplifying the subsequent purification burden. Instead of complex HPLC separation, the crude product is purified through a controlled crystallization process using a binary solvent system of acetone and dichloromethane. This shift reduces the overall processing time to roughly 18 hours and minimizes solvent consumption to merely 5.5 liters of acetone and 5.5 liters of dichloromethane per kilogram of product, representing a monumental improvement in operational efficiency and environmental sustainability.
Mechanistic Insights into Copper-Salt Mediated Stereocontrol
The core scientific innovation driving this process improvement lies in the specific interaction between the copper salt and the omacycline molecular framework. During the hydrogenation of M2 to M3, the nitrogen atoms within the tetracycline scaffold possess lone pair electrons that are highly susceptible to coordination with transition metals. The added copper salt, preferably copper(II) chloride, forms a transient coordination complex with these nitrogen centers. This complexation alters the electronic environment and steric hindrance around the reactive sites, effectively stabilizing the desired alpha-configuration and raising the energy barrier for the formation of the beta-isomer. This mechanism ensures that the reaction proceeds with high diastereoselectivity, yielding a crude product that is already enriched with the target isomer before any purification steps begin. For R&D teams, understanding this coordination chemistry is vital, as it suggests that precise control over copper salt concentration (optimally 5-15% relative to M2 weight) is critical for maximizing yield and minimizing impurity profiles in the final API.
Furthermore, the purification mechanism leverages the differential solubility properties of the target compound versus its impurities in a mixed solvent system. By dissolving the crude M3 in dichloromethane and slowly introducing acetone, the process induces supersaturation under controlled thermal conditions (10-15°C). The addition of seed crystals initiates nucleation, promoting the growth of pure M3 crystals while leaving soluble impurities, including residual isomers and catalyst traces, in the mother liquor. This crystallization-driven purification is far more scalable than chromatography, as it relies on thermodynamic equilibrium rather than kinetic separation on a stationary phase. The result is a high-purity intermediate suitable for final salt formation, achieved with significantly reduced equipment complexity and operator intervention, thereby enhancing the overall robustness of the manufacturing process for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Omacycline Intermediate M3 Efficiently
The synthesis of M3 from M2 involves a carefully orchestrated sequence of reductive amination and hydrogenation steps, optimized for both safety and yield. The process begins with the preparation of a reaction mixture containing M2, triethylamine, and pivalaldehyde in a methanol-ethanol solvent system, maintained at low temperatures to prevent side reactions. Following the initial mixing, the solution is transferred to a high-pressure reactor where the critical copper-mediated hydrogenation takes place using a palladium on carbon catalyst. Post-reaction workup involves pH adjustments and liquid-liquid extraction to isolate the crude product, which is then subjected to the proprietary crystallization protocol. For detailed operational parameters, stoichiometry, and safety precautions required for laboratory or pilot plant execution, please refer to the standardized synthesis guide below.
- Dissolve M2 in a mixed solvent of ethanol and methanol, then add triethylamine and pivalaldehyde under inert gas protection at low temperature.
- Transfer the mixture to a high-pressure reactor, add Pd/C catalyst and a specific amount of copper salt (e.g., CuCl2), and perform hydrogenation at controlled pressure and temperature.
- Filter the reaction mixture, adjust pH sequentially with acid/base, extract with dichloromethane, and purify the crude product via acetone/dichloromethane crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed synthesis route offers transformative economic and logistical benefits. The most immediate impact is observed in the drastic reduction of processing time, which shrinks the production cycle from three days to less than one day. This acceleration directly translates to increased asset utilization and higher throughput capacity without the need for capital expenditure on additional reactors or separation columns. Moreover, the elimination of preparative HPLC removes a major bottleneck in the production line, allowing for continuous or semi-continuous processing modes that are essential for meeting the demands of global pharmaceutical markets. The simplified workflow also reduces the dependency on highly skilled chromatography operators, lowering labor costs and minimizing the risk of human error during the purification stage.
- Cost Reduction in Manufacturing: The financial implications of switching from HPLC to crystallization are profound. Preparative chromatography is notoriously expensive due to the high cost of chromatography media, the energy required for pumping large solvent volumes, and the extensive waste treatment fees associated with mixed solvent streams. By replacing this with a simple crystallization step, the process eliminates the need for expensive resin columns and significantly lowers solvent procurement costs. The reduction in solvent volume—from hundreds of liters per kilogram to just over ten liters—means that waste disposal costs are slashed, and solvent recovery systems become much more efficient. Additionally, the removal of transition metal catalysts is streamlined, avoiding the need for costly scavengers or complex filtration setups often required to meet strict heavy metal limits in APIs.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step purifications that are prone to failure or variability. The new method enhances reliability by simplifying the unit operations involved. Crystallization is a well-understood, robust unit operation that scales linearly from the laboratory to the multi-ton scale, unlike chromatography which often faces scaling challenges. The reduced cycle time also means that inventory turnover is faster, allowing manufacturers to respond more agilely to fluctuations in market demand. Furthermore, the reagents used, such as copper salts and common organic solvents, are commodity chemicals with stable supply chains, reducing the risk of raw material shortages that can plague specialized chromatography consumables.
- Scalability and Environmental Compliance: As regulatory bodies impose stricter limits on solvent emissions and waste generation, the environmental profile of a manufacturing process becomes a key competitive advantage. This novel route significantly reduces the E-factor (mass of waste per mass of product) by minimizing solvent usage. The switch to a acetone/dichloromethane system, which is easier to recover and recycle compared to the complex mixtures used in HPLC, aligns with green chemistry principles. This not only facilitates easier permitting for new manufacturing sites but also future-proofs the supply chain against tightening environmental regulations. The ability to produce high-purity intermediates with a smaller environmental footprint positions the supplier as a preferred partner for multinational corporations committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of the copper-salt mediated approach. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: How does the addition of copper salt improve the synthesis of Omacycline intermediates?
A: The copper salt forms a coordination complex with the nitrogen atom in the omacycline structure. This unique spatial configuration significantly inhibits the conversion to the unwanted beta-isomer, reducing impurity levels from approximately 3.0%-4.0% down to 1.0%-2.0%.
Q: What are the solvent consumption advantages of this new purification method?
A: Unlike the prior art which relies on preparative HPLC consuming massive volumes of acetonitrile, dichloromethane, and n-heptane, the new method utilizes a direct crystallization process. This drastically reduces solvent usage to approximately 5.5L of acetone and 5.5L of dichloromethane per kilogram of product.
Q: Why is the crystallization method preferred over HPLC for M3 purification?
A: Preparative HPLC is time-consuming (requiring roughly 72 hours total process time) and generates significant waste. The crystallization method shortens the cycle time to about 18 hours, simplifies post-treatment, and effectively removes isomer impurities without the need for complex chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Omacycline Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process validation. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the copper-catalyzed omacycline synthesis are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and advanced crystallization units capable of handling the specific requirements of this protocol. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of M3 intermediate meets the exacting standards required for final API synthesis, providing our partners with absolute confidence in product quality and consistency.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their omacycline projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bill of materials. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. By partnering with us, you gain access to a supply chain that is not only cost-effective and reliable but also aligned with the highest standards of pharmaceutical innovation and environmental stewardship.
