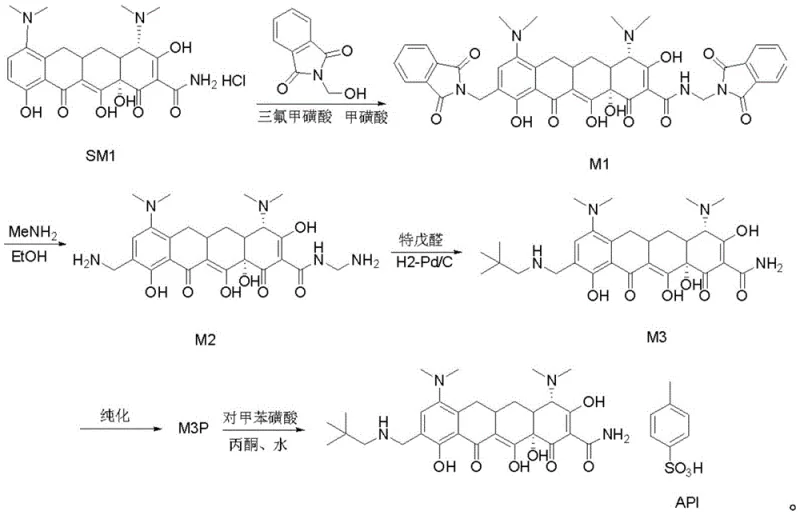
Advanced Copper-Catalyzed Synthesis of Omacycline Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust pathways for the production of next-generation tetracycline antibiotics, and the recent disclosure in patent CN111484424B offers a transformative approach to synthesizing omacycline intermediates. This technical breakthrough addresses critical bottlenecks in the conversion of intermediate M2 to M3, specifically targeting the persistent issue of beta-isomer impurities that have plagued traditional manufacturing routes. By introducing a soluble copper salt into the hydrogenation reaction system, the process achieves a remarkable suppression of unwanted stereoisomers without compromising the overall reaction yield. This innovation represents a significant leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials at scale. The strategic integration of copper catalysis not only refines the chemical profile of the product but also streamlines the downstream purification processes, setting a new benchmark for efficiency in antibiotic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of the M3 intermediate has relied heavily on high-performance liquid chromatography (HPLC), a technique that, while effective for separation, imposes severe constraints on industrial scalability and cost efficiency. The conventional process necessitates the use of massive volumes of organic solvents, including acetonitrile, dichloromethane, and n-heptane, creating a substantial environmental burden and inflating raw material procurement costs significantly. Furthermore, the post-treatment time for HPLC purification is excessively long, often extending up to 72 hours, which creates a bottleneck in the production schedule and limits the throughput of manufacturing facilities. The persistence of beta-isomer impurities at levels between 3.0% and 4.0% in the final product further complicates the quality control landscape, requiring additional remediation steps that erode profit margins. These inefficiencies highlight the urgent need for a more sustainable and economically viable alternative for cost reduction in API manufacturing.
The Novel Approach
The novel methodology described in the patent fundamentally reengineers the synthesis pathway by incorporating a copper salt additive directly into the catalytic hydrogenation step, thereby preventing the formation of impurities at the source rather than removing them later. This proactive approach allows for the replacement of complex chromatographic separation with a straightforward crystallization process using a dichloromethane and acetone mixed solvent system. The result is a drastic reduction in processing time from approximately 72 hours to merely 18 hours, enabling a much faster turnover rate for production batches. Additionally, the solvent consumption is minimized to just 5.5 liters of acetone and 5.5 liters of dichloromethane per kilogram of product, representing a monumental decrease in waste generation and handling costs. This streamlined workflow not only enhances the commercial scale-up of complex tetracyclines but also ensures a more consistent and high-quality output suitable for stringent regulatory requirements.
Mechanistic Insights into Copper-Salt Mediated Isomer Suppression
The core scientific advancement of this process lies in the unique interaction between the soluble copper salt and the nitrogen atoms within the omacycline molecular framework during the hydrogenation phase. Mechanistically, the copper ions act as a transient coordinating agent, forming a stable complex with the spatially accessible nitrogen centers on the tetracycline scaffold. This coordination effectively locks the molecule into a specific conformation that is energetically unfavorable for the epimerization or rearrangement reactions that typically lead to the formation of the beta-isomer. By stabilizing the desired alpha-configuration through this metal-ligand interaction, the reaction pathway is selectively guided towards the target product, ensuring that the stereochemical integrity of the molecule is preserved throughout the reduction process. This level of control is essential for maintaining the biological activity and safety profile of the final antibiotic drug substance.
From an impurity control perspective, the reduction of the beta-isomer content from a problematic 3.0-4.0% down to a highly acceptable 1.0-2.0% simplifies the entire downstream purification train. The lower impurity load means that the subsequent crystallization steps are far more efficient, as the solubility differences between the product and the impurities are easier to exploit without the need for repetitive recrystallizations. This mechanistic intervention essentially decouples the yield from the purity trade-off that often plagues organic synthesis, allowing manufacturers to achieve both high recovery rates and exceptional chemical purity simultaneously. The ability to fine-tune the reaction environment with simple inorganic salts like CuCl2 demonstrates a sophisticated understanding of coordination chemistry applied to practical industrial problems, offering a robust solution for reducing lead time for high-purity pharmaceutical intermediates.
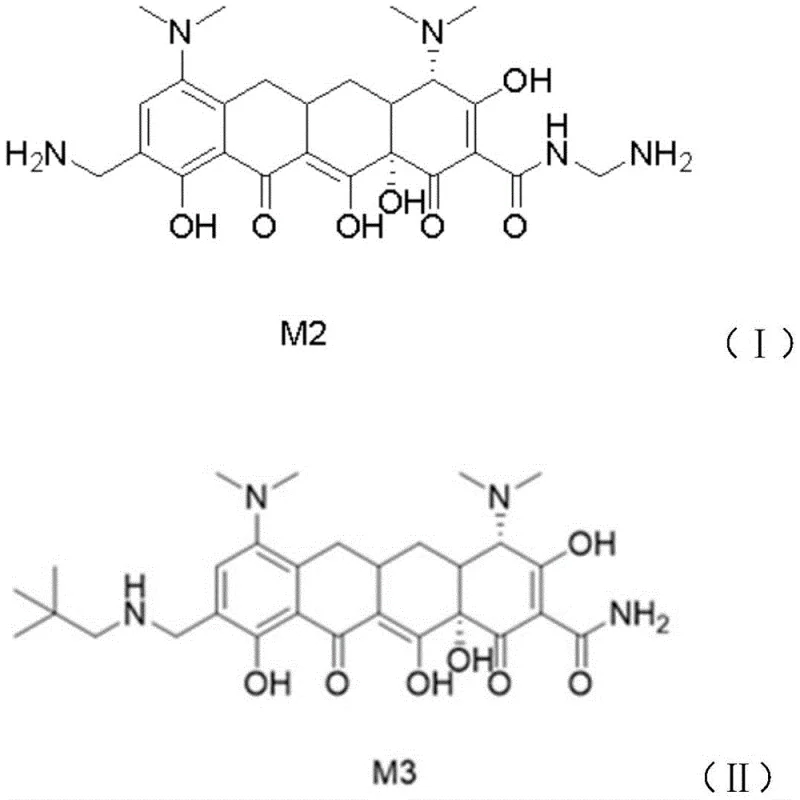
How to Synthesize Omacycline Intermediate M3 Efficiently
The synthesis of the M3 intermediate via this copper-enhanced route involves a carefully controlled sequence of mixing, hydrogenation, and extraction steps designed to maximize the benefits of the catalytic system. Operators must first prepare a homogeneous solution of the starting material M2 in a specific ratio of ethanol and methanol, ensuring that the temperature is rigorously maintained between 0-10°C to prevent premature side reactions. The addition of the copper salt and palladium catalyst must be performed under an inert atmosphere to protect the active species from oxidation, followed by a controlled pressurization with hydrogen gas to drive the reduction to completion. While the general principles are straightforward, precise adherence to the stoichiometric ratios and temperature profiles is critical to replicating the low isomer levels reported in the patent data. The detailed standardized synthesis steps are provided in the guide below.
- Dissolve M2 in ethanol/methanol mixture, cool to 0-10°C, and add triethylamine and pivalaldehyde.
- Transfer to a high-pressure reactor, add Pd/C catalyst and 5-15% copper salt (e.g., CuCl2), then pressurize with hydrogen.
- After reaction, extract with dichloromethane, concentrate, and crystallize using acetone/dichloromethane to obtain pure M3.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed synthesis route presents a compelling value proposition centered on operational efficiency and resource optimization. The elimination of the HPLC purification step removes a major capital-intensive bottleneck, allowing existing infrastructure to be utilized more effectively without the need for specialized chromatographic equipment. This shift translates directly into a more agile supply chain capable of responding rapidly to market demands for omacycline and related tetracycline derivatives. The reduction in solvent usage not only lowers direct material costs but also significantly decreases the logistical burden associated with the storage, handling, and disposal of hazardous chemical wastes. These factors combine to create a manufacturing process that is both economically superior and environmentally more sustainable.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the dramatic decrease in solvent consumption, which moves from hundreds of liters per kilogram in the old process to merely roughly 11 liters in the new process. This reduction eliminates the need for large-scale solvent recovery systems and reduces the purchase volume of expensive reagents like acetonitrile and n-heptane. Furthermore, the removal of the HPLC step saves on the operational costs associated with column maintenance, mobile phase preparation, and the energy required for prolonged pumping and separation. By simplifying the unit operations, the overall cost of goods sold (COGS) is significantly lowered, enhancing the competitiveness of the final API in the global market.
- Enhanced Supply Chain Reliability: The compression of the production cycle from 72 hours to approximately 18 hours means that manufacturing slots can be turned over four times faster than before. This increased throughput capacity provides a buffer against supply disruptions and allows for more flexible scheduling to meet urgent orders from downstream pharmaceutical partners. The reliance on common, commercially available reagents like copper chloride and standard solvents ensures that the supply chain is not vulnerable to shortages of exotic or highly specialized catalysts. Consequently, manufacturers can offer more reliable delivery timelines and maintain higher inventory levels of critical intermediates without tying up excessive working capital.
- Scalability and Environmental Compliance: Transitioning from a batch-wise chromatographic process to a continuous or semi-continuous crystallization process greatly facilitates scale-up from pilot plant to commercial tonnage production. The simplified waste stream, characterized by lower volumes of organic solvents, makes it easier to comply with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The ability to handle larger batch sizes without a proportional increase in processing time or waste generation supports the long-term growth strategies of pharmaceutical companies. This scalability ensures that the technology remains viable and cost-effective even as demand for the antibiotic grows, securing the supply chain for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method, derived directly from the experimental data and beneficial effects outlined in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines. The answers provide clarity on the mechanism of action, the quantitative improvements in efficiency, and the impact on product quality metrics. This information serves as a foundational resource for decision-makers assessing the strategic value of adopting this technology.
Q: How does the addition of copper salt improve the synthesis of Omacycline intermediates?
A: The copper salt forms a complex with the nitrogen atom in the omacycline structure, stabilizing the spatial configuration and significantly reducing the formation of the beta-isomer from 3.0-4.0% down to 1.0-2.0%.
Q: What are the solvent consumption advantages of this new method compared to prior art?
A: The new crystallization process consumes drastically less solvent, requiring only 5.5L of acetone and 5.5L of dichloromethane per kilogram of product, whereas the prior HPLC method required approximately 54L of acetonitrile, 290L of dichloromethane, and 178L of n-heptane.
Q: Does the copper-catalyzed method affect the overall yield of the M3 intermediate?
A: No, the method maintains a robust yield comparable to the prior art, achieving approximately 60.5% yield in Example 1 versus 61.1% in the comparative example, while significantly improving purity and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Omacycline Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the copper-catalyzed omacycline route can be seamlessly transferred from the laboratory to the factory floor. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in process optimization, we help our partners navigate the complexities of tetracycline synthesis with confidence and precision.
We invite you to collaborate with us to explore how this efficient synthesis method can transform your supply chain dynamics and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of partnering with a leader in fine chemical innovation. Let us work together to bring high-quality, cost-effective antibiotics to the market faster and more efficiently.
