Advanced Nickel-Catalyzed Synthesis of Heterocyclic Compounds for Commercial Pharmaceutical Production
Advanced Nickel-Catalyzed Synthesis of Heterocyclic Compounds for Commercial Pharmaceutical Production
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more sustainable and cost-effective methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN113387876A, which details a novel nickel-catalyzed direct reduction cross-coupling method. This technology specifically targets the construction of pyridine and diazine moieties, which are ubiquitous structural motifs in modern drug discovery and agrochemical development. By leveraging heterocyclic phosphonium salts as electrophiles and coupling them directly with aryl bromides, this invention circumvents the traditional reliance on pre-formed organometallic nucleophiles. For R&D directors and procurement specialists alike, this represents a paradigm shift towards simpler, greener, and economically superior synthetic routes that can be seamlessly integrated into existing manufacturing pipelines for high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of biaryl heterocycles has relied heavily on palladium-catalyzed cross-coupling reactions such as the Suzuki-Miyaura or Negishi couplings. While effective, these methods impose significant logistical and financial burdens on the supply chain. The Suzuki reaction, for instance, necessitates the use of arylboronic acids, which can be unstable, prone to protodeboronation, and often require multi-step synthesis themselves. Furthermore, the Negishi coupling involves organozinc reagents that are highly sensitive to moisture and air, demanding rigorous anhydrous conditions and specialized handling equipment that increase operational expenditures. Additionally, these conventional pathways frequently employ expensive palladium catalysts and, in some advanced variations, costly silver additives, which complicates the downstream removal of heavy metal residues to meet stringent pharmaceutical purity standards.
The Novel Approach
The methodology disclosed in the patent offers a transformative alternative by utilizing readily available aryl bromides instead of iodides or boronic acids, coupled with stable heterocyclic phosphonium salts. This approach not only broadens the substrate scope but also dramatically simplifies the reaction setup by eliminating the need for separate organometallic preparation steps. The use of a nickel catalyst system, activated by magnesium and lithium chloride, provides a robust platform for carbon-carbon bond formation under mild conditions. As illustrated below, this direct reductive coupling allows for the efficient synthesis of complex structures like 4-phenylpyridine with high selectivity.
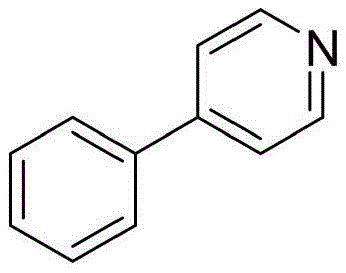
This strategic shift from palladium to nickel chemistry, combined with the use of stable phosphonium salts, results in a process that is inherently safer and more scalable. The ability to use aryl bromides, which are generally cheaper and more abundant than their iodide counterparts, further enhances the economic viability of this route for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nickel-Catalyzed Reductive Cross-Coupling
The core of this innovation lies in the unique catalytic cycle facilitated by the nickel species in the presence of a magnesium-lithium chloride activating system. The reaction initiates with the in situ generation of a low-valent nickel species, likely Ni(0), which undergoes oxidative addition with the aryl bromide. Simultaneously, the heterocyclic phosphonium salt acts as an activated electrophile. The magnesium serves as the terminal reductant, regenerating the active nickel catalyst and facilitating the reductive elimination step that forms the final C-C bond. This mechanism avoids the formation of free radical species that often lead to homocoupling byproducts, thereby ensuring a cleaner reaction profile. The general reaction scheme demonstrates the versatility of this transformation across various substituted pyridines and diazines.
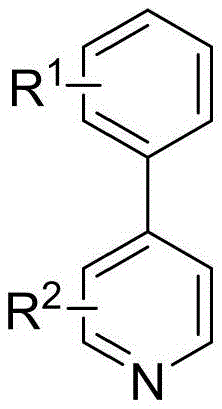
Impurity control is a critical aspect of this mechanism, particularly for pharmaceutical applications. The specific choice of ligands, such as 1,10-phenanthroline-5,6-dione, plays a pivotal role in stabilizing the nickel center and modulating its electronic properties to favor cross-coupling over side reactions. The patent data indicates that optimizing the ligand environment can push yields significantly higher, minimizing the formation of dehalogenated byproducts or homocoupled dimers. This level of control over the catalytic cycle ensures that the resulting crude product requires less intensive purification, directly translating to higher overall process efficiency and reduced solvent consumption during workup.
How to Synthesize Arylated Pyridines Efficiently
Implementing this synthesis route requires careful attention to the activation of the magnesium source and the maintenance of an inert atmosphere. The process begins with the thermal activation of magnesium turnings mixed with lithium chloride, which is crucial for generating the active reducing surface. Following this activation, the reaction components are combined in an ultra-dry solvent such as tetrahydrofuran. The detailed standardized synthesis steps for replicating this high-yielding transformation are outlined in the guide below.
- Activate magnesium turnings with lithium chloride under heat and vacuum to prepare the reducing agent system.
- Combine the activated magnesium mixture with ultra-dry solvent, phosphonium salt, nickel catalyst, ligand, and aryl bromide under nitrogen.
- Stir the reaction at room temperature, then quench, extract, and purify via column chromatography to isolate the target heterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed protocol offers tangible strategic benefits beyond mere technical novelty. The primary advantage is the substantial reduction in raw material costs achieved by replacing precious metal catalysts with base metals and utilizing commodity aryl bromides. This shift mitigates the volatility associated with palladium pricing and reduces the dependency on specialized organometallic suppliers. Furthermore, the simplified workflow eliminates entire unit operations related to the preparation and handling of sensitive reagents, leading to a streamlined manufacturing process that is easier to validate and scale.
- Cost Reduction in Manufacturing: The transition from palladium to nickel catalysts represents a direct decrease in catalyst expenditure, while the use of aryl bromides instead of iodides or boronic acids lowers the cost of goods sold. By avoiding the synthesis of organometallic intermediates, the process reduces labor hours and energy consumption associated with cryogenic conditions or extended reaction times. These cumulative efficiencies result in a leaner cost structure that enhances competitiveness in the global API intermediate market without compromising on quality.
- Enhanced Supply Chain Reliability: Relying on stable phosphonium salts and common aryl bromides significantly de-risks the supply chain compared to methods requiring fresh preparation of organozinc or organoboron reagents. These stable starting materials have longer shelf lives and are less susceptible to degradation during transport and storage. This stability ensures consistent batch-to-batch quality and reduces the likelihood of production delays caused by reagent failure, thereby securing a more reliable supply of critical heterocyclic building blocks for downstream drug synthesis.
- Scalability and Environmental Compliance: The mild reaction conditions, often proceeding effectively at room temperature, reduce the energy footprint of the manufacturing process. Additionally, the avoidance of toxic tin or zinc waste streams simplifies effluent treatment and aligns with increasingly stringent environmental regulations. The straightforward workup procedure involving standard extraction and chromatography facilitates easier scale-up from kilogram to tonne quantities, making this technology highly attractive for contract development and manufacturing organizations aiming to expand their portfolio of green chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed cross-coupling technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: Why is this nickel-catalyzed method superior to traditional palladium couplings?
A: This method eliminates the need for pre-synthesized organometallic reagents like boronic acids or zinc species, which are often moisture-sensitive and expensive. By using stable phosphonium salts and cheaper aryl bromides with a nickel catalyst, it significantly reduces raw material costs and simplifies the operational workflow.
Q: What represents the primary cost-saving advantage in this process?
A: The primary advantage lies in substituting expensive palladium catalysts and silver additives with abundant nickel systems, and replacing costly aryl iodides with widely available aryl bromides. This shift drastically lowers the bill of materials for large-scale production.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction operates under mild conditions (room temperature to 60°C) and utilizes simple post-treatment steps like extraction and column chromatography. The avoidance of air-sensitive organometallic intermediates enhances safety and scalability for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed methodology for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that guarantee every batch meets the exacting standards required by the global pharmaceutical industry.
We invite you to explore how this advanced synthesis route can optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next breakthrough in drug development.
