Advanced Mannitol-Derived Chiral Ligands for Scalable Asymmetric Synthesis and Commercial Production
In the rapidly evolving landscape of asymmetric synthesis, the demand for robust, cost-effective chiral ligands has never been more critical for the production of high-value pharmaceutical intermediates. Patent CN102464672A introduces a groundbreaking class of mannitol-derived chiral bidentate phosphite ligands that address many of the historical limitations associated with traditional chiral catalysts. This technology leverages the inherent chirality and rigidity of the mannitol backbone to create a stable, white solid ligand capable of forming highly active complexes with copper salts. Unlike many legacy systems that rely on expensive, multi-step syntheses or unstable precursors, this innovation utilizes readily available raw materials to achieve exceptional performance in asymmetric 1,4-conjugate addition reactions. The resulting catalysts facilitate the conversion of cyclic enones into optically active beta-ethyl cyclic ketones with remarkable efficiency, achieving yields of up to 99% and enantioselectivity reaching 93%. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes for complex drug candidates, particularly those requiring precise stereochemical control at the beta-position of ketone functionalities.
The structural versatility of this ligand system is a key factor in its broad applicability across different substrate classes. The core architecture allows for modular variation at two distinct positions, denoted as R1 and R2, enabling fine-tuning of the steric and electronic environment around the metal center. As illustrated in the structural diagrams below, the R1 group can be derived from protected mannitol variants (structures a or b), while the R2 group incorporates diverse aromatic or cycloaliphatic moieties (structures a', b', c', or d'). This modularity is crucial for optimizing catalyst performance for specific substrates, ensuring that the ligand can be adapted to meet the rigorous purity and selectivity standards required in modern API manufacturing. The ability to systematically vary these substituents provides a powerful toolkit for overcoming selectivity issues that often plague conventional catalytic systems.
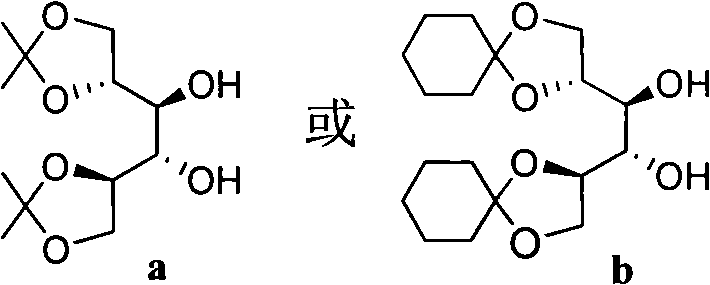
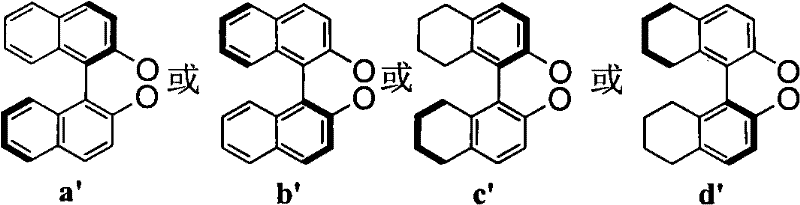
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric 1,4-conjugate addition of organometallic reagents to alpha,beta-unsaturated carbonyl compounds has relied heavily on chiral phosphoramidites or complex P,N-ligands derived from binaphthyl scaffolds. While effective in many cases, these conventional methods often suffer from significant drawbacks that hinder their widespread adoption in large-scale commercial manufacturing. A primary concern is the sensitivity of many phosphoramidite ligands to moisture and air, necessitating stringent handling conditions such as glovebox operations or extensive degassing protocols, which drastically increase operational costs and complexity. Furthermore, the synthesis of these traditional ligands frequently involves multiple steps, hazardous reagents, and difficult purification processes, leading to low overall yields and high material costs. In terms of catalytic performance, many existing systems exhibit substrate specificity, where a ligand optimized for one type of enone fails to deliver high enantioselectivity for another, forcing process chemists to engage in time-consuming trial-and-error screening campaigns. Additionally, some copper complexes formed with older ligand generations are prone to causing product racemization under certain reaction conditions, compromising the optical purity of the final pharmaceutical intermediate.
The Novel Approach
The methodology disclosed in CN102464672A offers a transformative solution to these challenges by introducing a mannitol-based phosphite framework that combines stability with high catalytic activity. The novel approach utilizes a straightforward condensation reaction between a mannitol derivative and a chiral chlorophosphite, catalyzed by DMAP and triethylamine in dichloromethane. This synthesis is remarkably simple, operating under mild temperatures ranging from -15°C to 30°C, which eliminates the need for energy-intensive cryogenic cooling often required for ligand preparation. The resulting ligands are stable white solids that can be stored under a nitrogen atmosphere without rapid degradation, simplifying inventory management and logistics for supply chain teams. In application, these ligands form in situ catalysts with copper salts that demonstrate broad substrate scope and exceptional tolerance, delivering high yields and enantioselectivity without the risk of product racemization seen in other systems. The general reaction scheme below highlights the efficiency of this coupling process, showcasing how complex chiral architectures are assembled from simple precursors in a single pot.
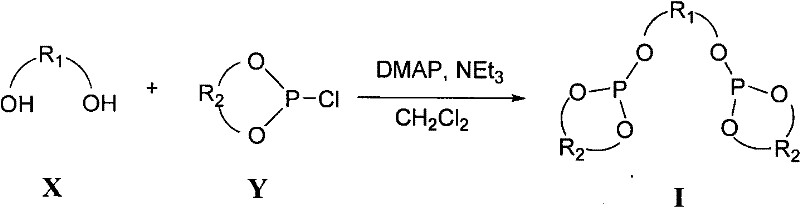
Mechanistic Insights into Mannitol-Derived Phosphite Coordination
The exceptional performance of these ligands stems from the unique coordination geometry imposed by the rigid mannitol backbone. When the bidentate phosphite ligand coordinates with a copper(I) or copper(II) species, it forms a well-defined chiral pocket that effectively discriminates between the enantiotopic faces of the incoming cyclic enone substrate. The phosphite moieties act as soft Lewis bases, stabilizing the copper center while allowing for the necessary lability to facilitate the transmetallation with diethylzinc. The steric bulk introduced by the R2 substituents (such as the bulky aromatic groups in structures a' or b') plays a critical role in shielding one face of the metal center, thereby directing the nucleophilic attack of the ethyl group to the opposite face of the enone. This precise spatial arrangement is essential for achieving the high enantiomeric excess values reported in the patent data. Furthermore, the electron-withdrawing nature of the phosphite oxygen atoms modulates the Lewis acidity of the copper, optimizing the activation of the carbonyl group for conjugate addition without promoting side reactions such as 1,2-addition.
Impurity control is another critical aspect where this mechanistic design excels. In many asymmetric catalytic processes, the formation of inactive catalyst aggregates or the decomposition of the ligand leads to the generation of difficult-to-remove impurities that can poison downstream reactions or fail regulatory specifications. The mannitol-derived phosphite ligands described here exhibit high stability under the reaction conditions, minimizing ligand degradation and the subsequent formation of phosphorus-containing byproducts. The use of a bidentate chelating system ensures that the copper remains tightly bound throughout the catalytic cycle, preventing the release of free copper ions that could catalyze non-selective background reactions. This stability translates directly into cleaner reaction profiles, as evidenced by the high GC assay results in the patent examples, which simplifies the downstream purification process. For manufacturing teams, this means fewer chromatography columns, less solvent consumption for recrystallization, and a more robust process that consistently meets stringent purity specifications required for GMP production.
How to Synthesize Mannitol-Derived Chiral Ligands Efficiently
The preparation of these high-performance ligands is designed for operational simplicity, making it accessible for both laboratory scale optimization and pilot plant production. The process avoids the use of exotic reagents or extreme conditions, relying instead on standard organic synthesis techniques that are familiar to most process chemistry teams. The key to success lies in maintaining an inert atmosphere to prevent hydrolysis of the chlorophosphite precursor and carefully controlling the addition rate of the base to manage the exotherm. Detailed standardized synthetic procedures, including specific molar ratios, solvent volumes, and workup protocols, are essential for reproducing the high yields and selectivity reported in the literature. The following section outlines the critical operational parameters required to execute this synthesis successfully.
- Prepare the reaction vessel under nitrogen atmosphere and dissolve the mannitol skeleton derivative (Component X) and chiral chlorophosphite (Component Y) in dichloromethane.
- Add catalytic amounts of DMAP and triethylamine as the base, maintaining the temperature between -15°C and 30°C to ensure controlled phosphorylation.
- Upon completion, remove the solvent under reduced pressure, triturate the residue with toluene to precipitate salts, filter, and purify the filtrate via flash chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this mannitol-derived ligand technology offers compelling economic and logistical benefits that extend beyond simple performance metrics. The primary driver for cost reduction lies in the raw material profile; mannitol is a commodity chemical produced on a massive scale for the food and pharmaceutical industries, ensuring a stable and low-cost supply chain compared to specialized chiral pool starting materials like amino acid derivatives or complex binaphthols. The synthetic route itself is concise, typically requiring only a single coupling step followed by a simple filtration and concentration, which drastically reduces labor hours and reactor occupancy time. By eliminating the need for complex multi-step ligand syntheses, manufacturers can significantly lower the cost of goods sold (COGS) for the final chiral intermediate. Furthermore, the high catalytic efficiency allows for lower catalyst loading in some embodiments, further reducing the consumption of precious metal salts and expensive ligand mass per kilogram of product.
- Cost Reduction in Manufacturing: The streamlined synthesis of the ligand eliminates the need for expensive transition metal catalysts often used in ligand construction, relying instead on organic bases like triethylamine and DMAP which are inexpensive and readily available. The reaction workup involves a simple precipitation of salts in toluene followed by filtration, avoiding costly and time-consuming aqueous extraction sequences or column chromatography on a large scale. This simplification of the unit operations directly translates to reduced utility consumption and waste disposal costs, providing a substantial margin improvement for high-volume production runs.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved because the key starting materials, mannitol derivatives and chlorophosphites, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The stability of the final ligand as a solid allows for long-term storage and batch consolidation, enabling procurement teams to purchase in larger quantities during favorable market conditions without fear of rapid degradation. This inventory flexibility ensures continuous production capability even during periods of raw material volatility, securing the supply chain against disruptions that commonly affect more sensitive chiral reagents.
- Scalability and Environmental Compliance: The process operates in common solvents like dichloromethane and toluene, which are well-understood in terms of recovery and recycling infrastructure within modern chemical plants, facilitating easier compliance with environmental regulations. The absence of heavy metal catalysts in the ligand synthesis step simplifies waste stream management, as the effluent does not require specialized treatment for metal removal prior to discharge or incineration. The mild reaction temperatures reduce the energy load on heating and cooling systems, contributing to a lower carbon footprint for the manufacturing process and aligning with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances is critical for evaluating the feasibility of integrating this catalytic system into existing manufacturing workflows.
Q: What are the primary advantages of using mannitol-derived ligands over traditional binaphthyl systems?
A: Mannitol-derived ligands offer a significant cost advantage due to the abundance of the starting material and often provide superior stability against hydrolysis compared to some phosphoramidites, while maintaining high enantioselectivity in copper-catalyzed conjugate additions.
Q: Can this ligand system be scaled for industrial production of chiral intermediates?
A: Yes, the synthesis utilizes common solvents like dichloromethane and toluene and operates at mild temperatures (-15°C to 30°C), making it highly amenable to scale-up without requiring specialized cryogenic equipment often needed for other asymmetric protocols.
Q: What level of enantioselectivity can be expected in asymmetric 1,4-addition reactions?
A: According to the patent data, specific embodiments of this ligand system have demonstrated enantioselectivity (ee) values reaching up to 93% with chemical yields as high as 99% in the synthesis of beta-ethyl cyclic ketones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bidentate Phosphite Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN102464672A can be reliably translated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR instrumentation to guarantee that every batch of ligand meets the exacting standards required for asymmetric catalysis. Our commitment to quality ensures that the enantioselectivity and yield performance you expect is delivered consistently, batch after batch.
We invite you to collaborate with us to evaluate the potential of this mannitol-derived ligand for your specific synthetic challenges. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current process, highlighting exactly where efficiencies can be gained. Please contact us to request specific COA data for our available ligand variants and to discuss route feasibility assessments for your target molecules. Let us help you optimize your chiral synthesis strategy with solutions that balance performance, cost, and reliability.
