Advanced Chiral Bidentate Phosphite Ligands for High-Purity Pharmaceutical Intermediate Production
Introduction to Novel Chiral Ligand Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct chiral centers with high fidelity, a challenge directly addressed by the technology disclosed in patent CN102464681A. This patent introduces a novel class of chiral bidentate phosphite ligands, designated as L (specifically La and Lb), which are derived from a unique hybridization of carbohydrate chemistry and binaphthyl scaffolds. These ligands are specifically engineered to facilitate the asymmetric 1,4-conjugate addition of organozinc reagents to cyclic enones, a transformation of paramount importance for generating optically active β-substituted cyclic ketones. Such ketones serve as indispensable building blocks for high-value active pharmaceutical ingredients (APIs) including muscone, anti-mycobacterial agents, and non-steroidal anti-inflammatory drugs. The structural integrity and electronic properties of these ligands allow them to form highly active complexes with copper salts, providing a reliable pathway for industrial-scale enantioselective synthesis.
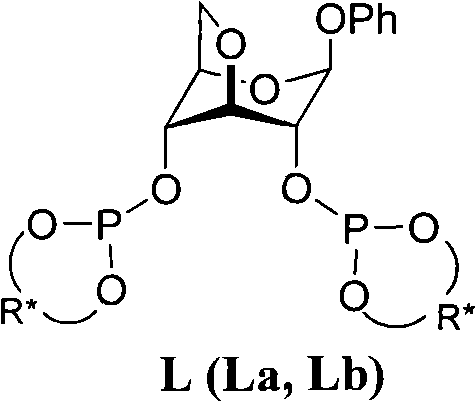
From a supply chain perspective, the physical properties of these ligands offer significant advantages over traditional liquid phosphoramidites. The patent describes the final product as a white solid that remains stable under a nitrogen atmosphere, simplifying logistics and storage requirements for procurement managers. This stability is crucial for maintaining consistent quality across large batches, ensuring that the reliable pharmaceutical intermediate supplier can deliver materials that meet stringent purity specifications without degradation during transit. Furthermore, the versatility of the ligand design, accommodating both H8-(R)-binaphthol and H8-(S)-binaphthol moieties, allows manufacturers to access either enantiomer of the target product simply by switching the starting chirality of the binaphthol component, thereby enhancing the flexibility of the production line.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric 1,4-conjugate addition reaction has relied heavily on a variety of chiral phosphorus ligands, including phosphoramidites and P,N-ligands, which often present significant operational challenges in a commercial setting. Many conventional ligands suffer from hydrolytic instability, requiring rigorous anhydrous conditions that increase the complexity and cost of reactor setup and maintenance. Additionally, traditional catalytic systems frequently exhibit narrow substrate scopes, meaning a ligand optimized for cyclohexenone might fail completely when applied to cyclopentenone or cycloheptenone derivatives, necessitating extensive and costly re-optimization campaigns for each new target molecule. The kinetic equilibrium between different organocopper species in these older systems can also lead to product racemization if the active complex is not sufficiently stable, resulting in lower enantiomeric excess (ee) values that require expensive downstream purification steps like chiral HPLC to rectify.
The Novel Approach
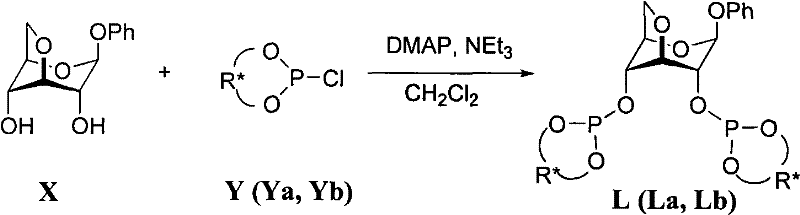
The approach detailed in CN102464681A overcomes these hurdles by introducing a rigid bicyclic carbohydrate framework linked to bulky H8-binaphthol groups, creating a sterically demanding environment that stabilizes the active copper catalyst species. This novel architecture prevents the formation of inactive or racemization-prone oligomeric copper species, thereby maintaining high catalytic activity and selectivity throughout the reaction duration. The synthesis of the ligand itself is remarkably straightforward, utilizing readily available starting materials like 3,6-anhydro-phenyl-β-D-glucoside and chiral phosphorous oxychlorides in a one-pot procedure. This simplicity translates directly into cost reduction in API manufacturing, as it eliminates the need for multi-step ligand synthesis protocols that typically plague the production of specialized chiral auxiliaries. The resulting catalyst system operates effectively under mild conditions, further reducing energy consumption and safety risks associated with extreme temperatures or pressures.
Mechanistic Insights into Cu-Catalyzed Asymmetric Conjugate Addition
The efficacy of the ligand/Cu catalyst system stems from the precise spatial arrangement of the phosphite oxygen atoms and the bulky binaphthyl substituents around the central copper ion. When the ligand coordinates with copper salts such as Cu(OTf)2 or (CuOTf)2·C6H6, it forms a well-defined chiral pocket that dictates the trajectory of the incoming organozinc nucleophile. The H8-binaphthol groups provide substantial steric bulk that blocks one face of the coordinated cyclic enone substrate, forcing the nucleophilic attack to occur exclusively from the less hindered face. This steric differentiation is the fundamental driver of the high enantioselectivity observed, with experimental data showing ee values reaching up to 96% for specific substrates like 2-cyclohexenone. The rigidity of the 3,6-anhydro sugar backbone prevents conformational flexibility that could otherwise blur the chiral environment, ensuring that the catalyst maintains its stereochemical integrity throughout the catalytic cycle.
Furthermore, the electronic nature of the phosphite linkages plays a critical role in modulating the Lewis acidity of the copper center, optimizing it for the activation of the α,β-unsaturated carbonyl system. Unlike phosphines which are strong sigma donors, phosphites are pi-acceptors, which helps stabilize the lower oxidation state of copper required for the transmetallation step with the organozinc reagent. This electronic tuning minimizes side reactions such as 1,2-addition, which is a common competing pathway in conjugate additions, thereby directing the reaction almost exclusively toward the desired 1,4-addition product. The mechanism also benefits from the absence of aggressive additives; the use of mild bases like triethylamine and nucleophilic catalysts like DMAP during ligand synthesis ensures that the sensitive P-O bonds are formed cleanly without decomposition, leading to a high-purity ligand that performs consistently in the subsequent catalytic application.
How to Synthesize Chiral Bidentate Phosphite Ligand Efficiently
The synthesis protocol outlined in the patent provides a scalable and reproducible route to obtaining high-purity ligands La and Lb, suitable for immediate use in catalytic applications or for stockpiling as stable intermediates. The process leverages standard organic synthesis techniques that are easily transferable from laboratory scale to pilot plant operations, requiring only common solvents like dichloromethane and toluene which are readily available in most chemical manufacturing facilities. The reaction conditions are mild, operating between -15°C and 30°C, which avoids the need for specialized cryogenic equipment often required for sensitive organometallic syntheses. Following the reaction, the workup involves a simple filtration step to remove amine salts, followed by concentration and purification via flash column chromatography, yielding the final product as a analytically pure white solid ready for characterization and use.
- Prepare the reaction vessel under a nitrogen atmosphere and dissolve 3,6-anhydro-phenyl-β-D-glucoside and chiral phosphorous oxychloride in dichloromethane solvent with DMAP catalyst.
- Cool the solution to -15°C and slowly add triethylamine dropwise, maintaining the temperature before allowing the mixture to warm to room temperature for completion.
- Remove the solvent under reduced pressure, triturate the residue with toluene, filter off solids, and purify the filtrate via silica gel flash column chromatography to isolate the white solid ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ligand technology offers tangible benefits related to operational efficiency and risk mitigation. The stability of the ligand as a solid material significantly reduces the risk of degradation during storage and transportation, ensuring that the material received at the manufacturing site performs identically to the material shipped. This reliability is essential for maintaining continuous production schedules and avoiding costly downtime caused by reagent failure or variability. Moreover, the synthetic route utilizes commodity chemicals and avoids the use of exotic or controlled precursors, which simplifies the sourcing process and mitigates supply chain disruptions associated with specialized raw materials. The ability to produce the ligand in-house or source it from a reliable pharmaceutical intermediate supplier with a robust manufacturing process ensures a steady flow of critical catalytic materials.
- Cost Reduction in Manufacturing: The streamlined synthesis of the ligand eliminates multiple purification steps and reduces solvent consumption compared to complex multidentate ligand systems. By avoiding the use of expensive transition metals in the ligand backbone itself and relying on abundant copper salts for catalysis, the overall cost of goods sold (COGS) for the final chiral intermediate is significantly lowered. The high turnover number of the catalyst system means that only small quantities of the ligand are required to produce large amounts of product, further amplifying the economic advantage and driving down the unit cost of the final API intermediate.
- Enhanced Supply Chain Reliability: The use of stable, solid ligands simplifies inventory management and reduces the need for specialized cold-chain logistics. Since the ligand can be stored under nitrogen for extended periods without loss of activity, manufacturers can maintain strategic stockpiles to buffer against market fluctuations or raw material shortages. This stability also facilitates global distribution, allowing the technology to be deployed across multiple manufacturing sites worldwide without concern for thermal degradation, thereby strengthening the resilience of the global supply network for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The reaction conditions described are inherently safer and more environmentally friendly, operating at near-ambient temperatures and utilizing solvents that can be easily recovered and recycled. The absence of hazardous reagents and the generation of minimal waste streams align with green chemistry principles, reducing the burden on waste treatment facilities and lowering compliance costs associated with environmental regulations. The scalability of the process is demonstrated by the straightforward workup procedure, which relies on filtration and crystallization rather than complex distillations, making it ideal for commercial scale-up of complex organic catalysts in large-scale reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology in industrial processes. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is crucial for evaluating the feasibility of integrating this catalytic system into existing production lines for chiral ketone synthesis.
Q: What are the stability characteristics of the chiral bidentate phosphite ligand described in CN102464681A?
A: The ligand is characterized as a stable white solid that can exist stably under a nitrogen atmosphere, facilitating easier storage and handling compared to moisture-sensitive liquid ligands often found in asymmetric catalysis.
Q: What represents the primary industrial application of this ligand system?
A: The primary application is catalyzing the asymmetric 1,4-conjugate addition of organozinc reagents to cyclic enones, enabling the efficient synthesis of optically active β-substituted cyclic ketones which are critical intermediates for drugs like Muscone and Ibuprofen.
Q: How does the ligand structure influence enantioselectivity in copper-catalyzed reactions?
A: The ligand combines a rigid carbohydrate backbone with bulky H8-binaphthol groups, creating a defined chiral pocket around the copper center that effectively discriminates between enantiotopic faces of the substrate during the conjugate addition process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bidentate Phosphite Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug development and optimizing manufacturing costs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity chiral ligands and intermediates that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex organometallic chemistries under strict inert atmospheres guarantees the integrity of sensitive materials like the phosphite ligands described in CN102464681A.
We invite you to collaborate with us to leverage this innovative ligand technology for your specific synthetic challenges. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volume and specific target molecules. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our supply solutions can enhance your operational efficiency and reduce your time to market for high-value pharmaceutical products.
