Revolutionizing Pinoxaden Manufacturing: A Scalable One-Pot Catalytic Strategy for Global Agrochemical Supply Chains
The global demand for high-efficiency herbicides continues to drive innovation in process chemistry, particularly for key active ingredients like Pinoxaden. A significant technological breakthrough in this domain is documented in Chinese patent CN112028906A, which discloses a novel "one-pot cooking method" for the preparation of Pinoxaden. This patent represents a paradigm shift from traditional multi-step batch processes to a streamlined, catalytic one-pot synthesis. By utilizing specific nano-oxide catalysts, the inventors have successfully merged the critical coupling and acylation stages into a single operational sequence. This advancement is not merely a laboratory curiosity but a robust industrial solution designed to address the persistent challenges of yield optimization, waste reduction, and operational simplicity in agrochemical manufacturing. For R&D directors and process engineers, this technology offers a compelling pathway to enhance production efficiency while maintaining stringent quality standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pinoxaden has been plagued by operational inefficiencies inherent in stepwise construction. Conventional methodologies typically involve the separate execution of the coupling reaction between the malonic acid derivative and the diazepane ring, followed by a distinct acylation step with pivaloyl chloride. This segregation is necessitated by the kinetic instability of the intermediates; specifically, incomplete coupling in the first stage leaves residual 1-oxy-4,5-diazepane, which aggressively reacts with pivaloyl chloride in the subsequent step to generate difficult-to-remove impurities. Consequently, manufacturers are forced to implement rigorous isolation and purification protocols between steps, drastically increasing solvent consumption, energy usage, and overall processing time. These disjointed operations create significant bottlenecks in production throughput and elevate the environmental burden through increased generation of hazardous waste streams, posing a continuous challenge for supply chain sustainability and cost management.
The Novel Approach
In stark contrast to these fragmented protocols, the methodology outlined in patent CN112028906A introduces a cohesive "one-pot" strategy that fundamentally restructures the synthetic timeline. By employing nano-structured metal oxides as heterogeneous catalysts, the process achieves high conversion rates for the initial condensation without the need for intermediate isolation. This allows the subsequent addition of the acylating agent directly into the same reaction vessel, effectively capturing the intermediate in situ before it can degrade or form side products. The integration of these steps eliminates the tedious work-up procedures associated with traditional routes, thereby compressing the total online reaction time and simplifying the equipment footprint. This holistic approach not only enhances the atomic economy of the synthesis but also aligns perfectly with modern green chemistry principles, offering a scalable solution for the commercial production of high-purity agrochemical intermediates.
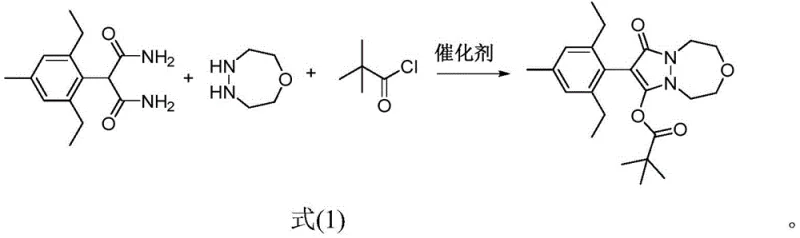
Mechanistic Insights into Nano-Oxide Catalyzed Cyclization
The cornerstone of this innovative process lies in the unique catalytic activity of nano-sized metal oxides, specifically nano-titanium oxide (TiO2), nano-magnesium oxide (MgO), or nano-zirconium oxide (ZrO2). Unlike traditional homogeneous bases that may lead to indiscriminate deprotonation and side reactions, these nano-oxides function as solid surface catalysts that promote the specific ammonolysis of the ester/amide functionalities. The high surface-area-to-volume ratio of the nano-particles provides abundant active sites that facilitate the nucleophilic attack of the diazepane nitrogen on the malonamide carbonyl group. This catalytic promotion ensures that the coupling reaction proceeds to near-completion before the acylation reagent is introduced, effectively suppressing the competitive reaction between the free amine and the acid chloride. This precise control over reaction kinetics is critical for minimizing the formation of N-acylated by-products, thereby securing a cleaner crude profile and reducing the load on downstream purification units.
Furthermore, the mechanistic pathway benefits from the mild basicity and thermal stability of these inorganic catalysts, which remain active throughout the temperature ramp from 100°C to 120°C required for cyclization. The catalyst acts as a proton scavenger during the condensation phase, driving the equilibrium towards the cyclic intermediate without decomposing the sensitive diazepane ring. Upon cooling and the addition of triethylamine and pivaloyl chloride, the system transitions smoothly to the acylation phase. The robustness of the nano-catalyst ensures that it does not interfere with the acylation step, nor does it require complex removal procedures prior to the second stage. This dual-functionality—promoting cyclization while tolerating acylation conditions—exemplifies a sophisticated understanding of heterogeneous catalysis in fine chemical synthesis, offering R&D teams a reliable mechanism to control impurity profiles and ensure batch-to-batch consistency in large-scale manufacturing environments.
How to Synthesize Pinoxaden Efficiently
Implementing this one-pot protocol requires careful attention to stoichiometry and thermal management to maximize the benefits of the nano-catalytic system. The process begins with the charging of the malonamide precursor and the diazepane heterocycle into a solvent such as toluene or dioxane, alongside a catalytic loading of the selected nano-oxide. The mixture is then subjected to elevated temperatures to drive the cyclization to completion, a critical step that determines the success of the subsequent one-pot acylation. Once the coupling is verified, the reaction is cooled, and the acylating agents are introduced under controlled conditions to finalize the molecular architecture of Pinoxaden. For detailed operational parameters, stoichiometric ratios, and specific work-up procedures validated by experimental data, please refer to the standardized synthesis guide below.
- Charge 2-(2,6-diethyl-4-methylphenyl)malonamide, 1-oxy-4,5-diazepane, and a nano-oxide catalyst (such as nano-MgO or nano-TiO2) into a suitable solvent like toluene.
- Heat the reaction mixture to 100-120°C and maintain for 8-12 hours to facilitate the initial condensation and cyclization.
- Cool the mixture to room temperature, add triethylamine and pivaloyl chloride, and stir for 5-6 hours to complete the acylation and isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot catalytic technology translates directly into tangible operational efficiencies and risk mitigation. The consolidation of two major synthetic steps into a single vessel operation drastically reduces the requirement for intermediate storage, handling, and quality control testing, which are often hidden cost drivers in complex chemical supply chains. By simplifying the process flow, manufacturers can achieve faster turnaround times from raw material intake to finished goods, enhancing the agility of the supply network to respond to market fluctuations. Moreover, the use of commercially available nano-oxides and common solvents like toluene ensures that the raw material basket remains stable and cost-effective, avoiding reliance on exotic or volatile reagents that could jeopardize supply continuity. This process intensification strategy is a key enabler for cost reduction in agrochemical intermediate manufacturing, delivering value through both direct expense savings and indirect operational improvements.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for extensive solvent exchanges, filtration, and drying operations, which are traditionally resource-intensive. By avoiding these unit operations, the process significantly lowers energy consumption and solvent procurement costs. Additionally, the high conversion rates achieved by the nano-catalysts minimize the loss of valuable starting materials to waste streams, improving the overall mass balance and yield efficiency. This lean manufacturing approach allows for substantial cost savings per kilogram of produced Pinoxaden, making the final product more competitive in the global marketplace without compromising on quality specifications.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route reduces the number of potential failure points in the production line, thereby increasing the reliability of supply. Fewer processing steps mean less exposure to equipment downtime, operator error, or cross-contamination risks that can delay shipments. The robustness of the nano-oxide catalysts, which are stable and easy to handle, further contributes to process stability. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery expectations of downstream formulators and distributors in the agricultural sector.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis is inherently scalable, as it reduces the equipment footprint and complexity required for commercial production. Facilities can produce larger batches in existing reactors without the need for additional isolation tanks or dryers. Furthermore, the reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations regarding industrial emissions and effluent treatment. This proactive approach to environmental compliance mitigates regulatory risk and supports corporate sustainability goals, ensuring long-term viability for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Pinoxaden synthesis route. These insights are derived directly from the experimental data and technical disclosures within patent CN112028906A, providing a factual basis for evaluating the technology's fit within your existing production capabilities. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What is the primary advantage of using nano-oxide catalysts in Pinoxaden synthesis?
A: Nano-oxides like nano-MgO and nano-TiO2 significantly promote the ammonolysis conversion rate, allowing the coupling and acylation steps to proceed in a single pot without the formation of excessive by-products typically caused by unreacted intermediates.
Q: Which solvents are compatible with this one-pot catalytic method?
A: The patent specifies that common organic solvents such as toluene, nitrobenzene, and dioxane are effective for this reaction, providing flexibility for process optimization based on boiling point and solubility requirements.
Q: How does this method impact environmental compliance compared to traditional routes?
A: By eliminating the need for intermediate isolation and reducing the number of reaction vessels and work-up procedures, this one-pot method effectively reduces the volume of three wastes (waste water, waste gas, and solid waste) generated during production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pinoxaden Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112028906A for the production of high-value agrochemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial realities. Our facilities are equipped with state-of-the-art reactors capable of handling high-temperature heterogeneous catalysis, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Pinoxaden meets the exacting standards required for global registration and formulation.
We invite you to leverage our technical expertise to optimize your supply chain for this critical herbicide intermediate. Whether you are looking to validate this one-pot route or require a Customized Cost-Saving Analysis for your current procurement strategy, our team is ready to assist. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and comprehensive quotations that reflect the efficiencies of our advanced manufacturing capabilities.
