Advanced Cefdinir Manufacturing: Overcoming Anhydrous Limitations for Commercial Scale-Up
Introduction to Next-Generation Cefdinir Production
The pharmaceutical landscape for third-generation cephalosporins is constantly evolving, driven by the need for more efficient and environmentally sustainable manufacturing processes. Patent CN101817835B introduces a transformative approach to the synthesis of Cefdinir, a broad-spectrum antibiotic critical for treating resistant bacterial infections. This technology pivots away from traditional, harsh activation methods, utilizing pentafluorophenol as a superior activating group for the key side chain acid. By integrating this advanced chemistry, manufacturers can achieve exceptional purity levels exceeding 99.9% while drastically simplifying the operational workflow. For global supply chains, this represents a pivotal shift towards more reliable cefdinir intermediate supplier capabilities, ensuring consistent quality and availability for downstream API production.
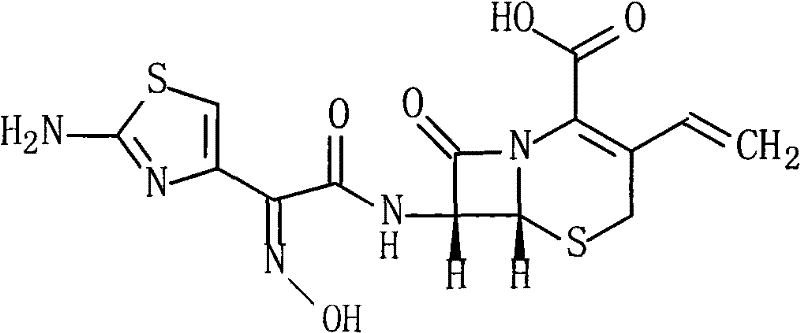
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefdinir has been plagued by significant technical bottlenecks associated with traditional activation reagents. Prior art, such as the methods disclosed in CN101182327A, relied heavily on phosphorus pentachloride (PCl5) to convert the side chain acid into an acyl chloride. This approach imposes draconian requirements for strictly anhydrous conditions, necessitating expensive solvent drying protocols and specialized equipment to exclude moisture. Furthermore, the generation of phosphorus-containing waste streams creates severe environmental compliance challenges and complicates the purification of the final product. The presence of these difficult-to-remove impurities often results in lower overall yields and necessitates complex, multi-step recrystallization procedures that erode profit margins and extend production cycles.
The Novel Approach
In stark contrast, the methodology outlined in CN101817835B leverages pentafluorophenol to form a highly reactive yet stable active ester in situ. This strategic substitution fundamentally alters the reaction landscape by relaxing the stringent anhydrous constraints, thereby allowing for more robust and forgiving process parameters. The reaction proceeds efficiently in DMF at mild temperatures ranging from 5°C to 10°C, coupling the activated ester directly with 7-AVCA. This pathway not only circumvents the generation of hazardous phosphorus byproducts but also streamlines the isolation of the intermediate through simple pH adjustment and filtration. The result is a process that is inherently safer, cleaner, and far more amenable to large-scale commercialization without compromising the structural integrity of the sensitive beta-lactam ring.
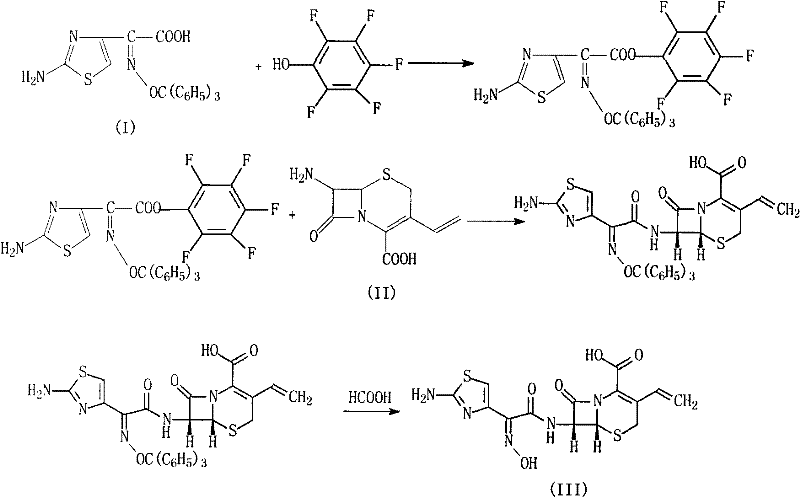
Mechanistic Insights into Pentafluorophenol-Mediated Activation
The core innovation of this synthesis lies in the electronic properties of the pentafluorophenol moiety, which serves as an exceptional leaving group during the nucleophilic attack by the 7-AVCA amine. Unlike acyl chlorides, which are hypersensitive to hydrolysis, the pentafluorophenyl ester maintains sufficient stability to allow for controlled addition of the beta-lactam nucleus. The electron-withdrawing fluorine atoms on the phenol ring significantly increase the electrophilicity of the carbonyl carbon, facilitating rapid amide bond formation even at low temperatures. This kinetic advantage ensures that the coupling reaction reaches completion quickly, minimizing the exposure time of the fragile 7-AVCA substrate to potentially degradative conditions. Consequently, the formation of unwanted open-ring impurities or epimerization at the chiral centers is effectively suppressed, preserving the potent biological activity of the final antibiotic.
Furthermore, the workup procedure capitalizes on the solubility differences between the desired product and the pentafluorophenol byproduct. Following the condensation, the reaction mixture is treated with water and acidified to a specific pH range of 6 to 7. Under these conditions, the protected Cefdinir intermediate precipitates selectively as a solid, while the soluble pentafluorophenol and excess reagents remain in the aqueous-organic supernatant. This phase separation is critical for impurity control, as it physically removes the bulk of organic contaminants before the final deprotection step. The subsequent reflux with formic acid cleanly removes the trityl protecting group without attacking the vinyl side chain or the beta-lactam core, delivering a final product with a remarkably clean impurity profile that meets rigorous pharmacopeial standards.
How to Synthesize Cefdinir Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to maximize the economic and technical benefits described above. The process begins with the activation of the thiazole side chain acid, followed by a direct coupling with the cephalosporin nucleus, and concludes with a deprotection sequence. Operators must adhere to the specified molar ratios, typically maintaining a 1:1:1 ratio between the side chain acid, pentafluorophenol, and 7-AVCA to prevent excess reagent accumulation. The detailed standardized operating procedures for temperature ramping, pH adjustments, and crystallization kinetics are essential for reproducing the high yields reported in the patent data. For a comprehensive breakdown of the exact operational parameters and safety protocols required for implementation, please refer to the technical guide below.
- Activate (Z)-2-(2-aminothiazol-4-yl)-2-trityloxyiminoacetic acid with pentafluorophenol in DMF at 5-10°C.
- Condense the activated ester with 7-AVCA and triethylamine, adjusting pH to 6-7 to isolate the protected intermediate.
- Reflux the intermediate with formic acid to remove the trityl protecting group, followed by pH adjustment and crystallization to obtain high-purity Cefdinir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this pentafluorophenol-based route offers substantial strategic advantages beyond mere chemical elegance. The elimination of phosphorus pentachloride removes a major hazard from the facility, reducing the need for specialized corrosion-resistant infrastructure and lowering insurance and safety compliance costs. Additionally, the relaxation of anhydrous requirements means that solvent quality specifications can be slightly broadened without risking batch failure, leading to direct savings on raw material procurement. The simplified workup, which relies on precipitation rather than complex chromatographic separations or extensive washing, significantly reduces solvent consumption and waste disposal volumes. These factors combine to create a manufacturing process that is not only more cost-effective but also more resilient to supply chain disruptions affecting specialized reagents.
- Cost Reduction in Manufacturing: The transition to pentafluorophenol activation eliminates the need for expensive and hazardous phosphorus reagents, which often require costly neutralization and disposal procedures. By avoiding the generation of phosphorus waste, facilities can significantly reduce their environmental treatment overheads and avoid the capital expenditure associated with handling corrosive acyl chlorides. Furthermore, the high selectivity of the reaction minimizes the loss of valuable 7-AVCA starting material, ensuring that the theoretical yield is closely approached in practice, which directly improves the cost-per-kilogram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Traditional methods relying on strict anhydrous conditions are prone to batch failures due to minor fluctuations in humidity or solvent water content, leading to unpredictable production schedules. This new method's tolerance to moisture variations ensures consistent batch-to-batch reproducibility, allowing supply chain planners to forecast delivery dates with greater confidence. The use of commercially available and stable reagents like pentafluorophenol also mitigates the risk of sourcing bottlenecks, ensuring a continuous flow of materials for uninterrupted commercial scale-up of complex antibiotic intermediates.
- Scalability and Environmental Compliance: As regulatory pressure on pharmaceutical manufacturing intensifies, the ability to demonstrate a green chemistry profile is a competitive differentiator. This process generates significantly less hazardous waste compared to conventional routes, aligning with global sustainability goals and simplifying the permitting process for capacity expansion. The straightforward isolation steps are easily transferable from pilot plant to multi-ton reactors, enabling rapid response to market demand surges without the need for extensive process re-engineering or additional purification trains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these nuances is vital for R&D teams evaluating technology transfer and for quality assurance personnel establishing control strategies. The answers provided are derived directly from the experimental data and comparative analysis presented in the patent literature, ensuring accuracy and relevance to real-world manufacturing scenarios. For further clarification on specific validation parameters or regulatory filing support, our technical team is available to provide detailed documentation.
Q: What is the primary advantage of using pentafluorophenol over phosphorus pentachloride in Cefdinir synthesis?
A: The use of pentafluorophenol eliminates the need for strict anhydrous conditions required by phosphorus pentachloride (PCl5) methods. This significantly reduces operational complexity, minimizes environmental pollution from phosphorus byproducts, and simplifies the purification process, leading to higher final product purity.
Q: What are the typical reaction conditions for the condensation step in this novel route?
A: The condensation reaction is conducted under mild thermal conditions, specifically between 5°C and 10°C. The process utilizes DMF as a solvent and requires precise pH control (adjusted to 6-7 using hydrochloric acid) to ensure optimal precipitation of the intermediate while maintaining stereochemical integrity.
Q: How does this method impact the impurity profile of the final API?
A: By avoiding the formation of difficult-to-remove phosphorus-containing impurities and utilizing a highly selective activation group, this method achieves a final purity of up to 99.9%. The streamlined workup procedure effectively removes side products, ensuring a cleaner impurity profile suitable for stringent regulatory requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our facility is equipped to handle the specific requirements of beta-lactam chemistry, including dedicated containment systems for potent compounds and state-of-the-art analytical laboratories for impurity profiling. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which verify every batch against the highest international standards before release.
We invite you to leverage our technical proficiency to optimize your Cefdinir supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of this pentafluorophenol activation technology can drive efficiency and reliability in your antibiotic manufacturing operations.
