Advanced Enzymatic Route for Cefdinir Production: Enhancing Purity and Scalability for Global Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with operational efficiency, particularly for critical antibiotics like Cefdinir. Patent CN102153566A introduces a transformative method for preparing Cefdinir that fundamentally shifts the paradigm from harsh chemical hydrolysis to a sophisticated biocatalytic approach. This innovation centers on the use of immobilized carboxylic ester hydrolase to convert a Cefdinir ester intermediate into the final active pharmaceutical ingredient under remarkably mild conditions. By avoiding the ultra-low temperatures and aggressive alkaline environments typical of legacy processes, this technology offers a compelling value proposition for manufacturers aiming to reduce environmental impact while enhancing product quality. The method achieves a final purity exceeding 99% via HPLC analysis, demonstrating that biological catalysis can meet the rigorous standards required for third-generation cephalosporin production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefdinir has been plagued by complex multi-step sequences that impose heavy burdens on production facilities and supply chains. One established route, described in U.S. Patent 4,559,334, involves the reaction of a benzhydryl ester derivative with 4-acetobromoacetyl bromide, followed by nitroso-group oxidation and cyclization. As illustrated in the reaction scheme below, this pathway necessitates the introduction and subsequent removal of protecting groups, creating significant inefficiencies.
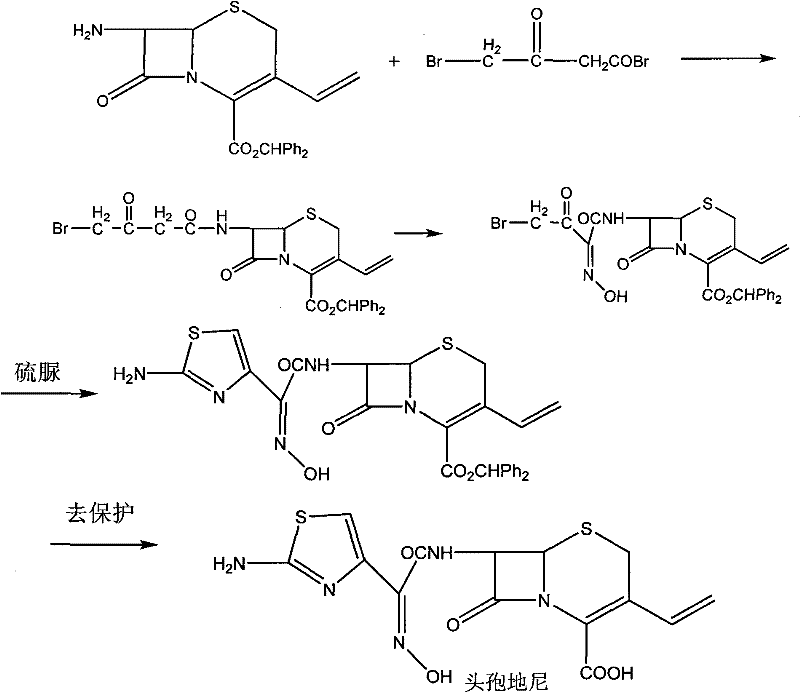
The drawbacks of such traditional chemical routes are manifold; they often require difficult-to-obtain raw materials and suffer from low overall yields due to the cumulative losses across multiple purification stages. Furthermore, these methods frequently rely on chromatographic separation and freeze-drying, which are not only capital-intensive but also difficult to scale for industrial production. Another common approach involves the use of strong inorganic bases for hydrolysis at low temperatures, which creates a highly alkaline system prone to causing product degradation and the generation of unwanted impurities. These harsh conditions demand specialized cryogenic equipment and rigorous safety protocols, driving up both operational costs and the risk of batch failure.
The Novel Approach
In stark contrast, the methodology disclosed in CN102153566A streamlines the synthesis by leveraging the specificity of enzymatic catalysis. Instead of isolating the Cefdinir ester intermediate, the process utilizes the aqueous solution directly for the hydrolysis step, effectively merging two major unit operations into a seamless flow. This eliminates the need for drying and redissolving the intermediate, which traditionally consumes vast amounts of organic solvents and time. The reaction scheme for the conventional strong base hydrolysis method highlights the complexity that the new enzymatic route successfully bypasses.
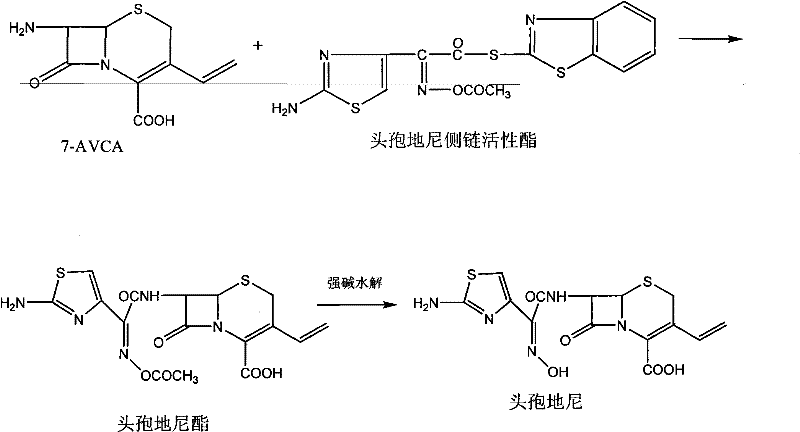
By employing an immobilized carboxylic ester hydrolase, the new method operates within a neutral to slightly alkaline pH range of 5.5 to 8.5, completely avoiding the corrosive nature of sodium hydroxide or potassium hydroxide systems. This shift not only preserves the integrity of the sensitive beta-lactam ring but also simplifies the downstream processing requirements. The result is a process that is inherently safer, more environmentally friendly, and capable of delivering a superior crystalline form of Cefdinir without the need for extensive purification interventions.
Mechanistic Insights into Enzymatic Hydrolysis of Cefdinir Ester
The core of this technological advancement lies in the precise mechanism of the immobilized carboxylic ester hydrolase, which acts as a highly selective biocatalyst. Unlike non-specific chemical bases that attack various electrophilic sites on the molecule, the enzyme targets the specific ester linkage on the side chain with exceptional fidelity. This selectivity is crucial because the Cefdinir molecule contains a reactive beta-lactam ring that is susceptible to ring-opening under harsh alkaline conditions. The enzymatic process ensures that hydrolysis occurs exclusively at the intended acetoxy imido group, thereby preventing the formation of ring-opened impurities that are difficult to remove and can compromise the safety profile of the antibiotic. The immobilization of the enzyme further enhances this mechanism by providing a stable microenvironment that maintains catalytic activity over extended periods.
From an impurity control perspective, the mild reaction conditions play a pivotal role in defining the quality of the final product. In conventional strong base hydrolysis, the high pH can lead to epimerization or degradation of the vinyl group on the cephem nucleus. However, by maintaining the reaction temperature between 0°C and 50°C, and specifically optimizing it around 25°C to 35°C, the enzymatic route suppresses these thermal degradation pathways. The patent data indicates that this controlled environment allows for the direct crystallization of Cefdinir with a content surpassing 97% and purity greater than 99%. This level of quality is achieved not through aggressive post-reaction purification but through the intrinsic cleanliness of the biocatalytic transformation itself, representing a significant leap forward in process chemistry design.
How to Synthesize Cefdinir Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this enzymatic strategy at scale. The process begins with the acylation of 7-amino-3-vinyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid (7-AVCA) using a Cefdinir active ester in the presence of an organic base. Following the formation of the ester intermediate, the mixture undergoes extraction and washing to remove organic impurities, after which the solvent is recovered via vacuum distillation. The resulting aqueous phase is then subjected to enzymatic hydrolysis, followed by pH adjustment and crystallization to yield the final product. For a detailed breakdown of the specific reagent quantities, timing, and temperature controls required for each stage, please refer to the standardized synthesis guide below.
- React 7-AVCA with Cefdinir active ester in an organic solvent/water mixture using an organic base at 0-35°C to form the Cefdinir ester intermediate.
- Extract and wash the reaction mixture, remove organic solvents via vacuum distillation to obtain an aqueous solution of the Cefdinir ester.
- Add immobilized carboxylic ester hydrolase to the aqueous solution at pH 5.5-8.5 and 0-50°C, followed by pH adjustment and crystallization to isolate Cefdinir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis route translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of harsh chemical reagents and the reduction in unit operations directly address the growing pressure to minimize manufacturing costs while ensuring supply continuity. By removing the need for cryogenic cooling systems and expensive corrosion-resistant reactors, capital expenditure requirements for new production lines are significantly lowered. Furthermore, the ability to recycle the immobilized enzyme adds a layer of economic efficiency that is absent in single-use chemical catalyst systems, driving down the variable cost per kilogram of the active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The transition to enzymatic hydrolysis removes the dependency on large quantities of inorganic strong alkalis and the associated neutralization acids, which substantially reduces the cost of raw materials and waste treatment. Because the process avoids the isolation of the intermediate ester, there is a drastic reduction in the volume of organic solvents required for dissolution and recrystallization steps. This solvent saving not only lowers procurement costs but also reduces the energy load associated with solvent recovery and distillation. Additionally, the milder conditions mean that standard stainless steel equipment can be utilized instead of specialized alloys, further optimizing the asset utilization rate and extending the lifecycle of manufacturing infrastructure.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route by combining the hydrolysis and deprotection steps inherently shortens the production cycle time, allowing for faster turnaround on customer orders. The robustness of the immobilized enzyme against varying reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. Moreover, the raw materials required, such as 7-AVCA and the active ester, are commercially available and do not rely on exotic or hard-to-source reagents that could bottleneck the supply chain. This reliability is critical for maintaining steady inventory levels of high-demand antibiotics in a volatile global market.
- Scalability and Environmental Compliance: The enzymatic process is inherently scalable, as the reaction kinetics are not limited by heat transfer issues associated with exothermic chemical hydrolysis. This makes the technology suitable for commercial scale-up of complex pharmaceutical intermediates from pilot plants to multi-ton production facilities without significant re-engineering. From an environmental standpoint, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on pharmaceutical effluent. The absence of heavy metal catalysts and the minimization of saline waste streams simplify wastewater treatment protocols, ensuring that the manufacturing facility remains compliant with environmental standards while operating at maximum capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this enzymatic Cefdinir synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology compares to established industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this greener and more efficient manufacturing route.
Q: What are the primary advantages of using immobilized enzymes over strong alkali for Cefdinir hydrolysis?
A: Using immobilized carboxylic ester hydrolase allows for reaction conditions that are significantly milder (pH 5.5-8.5, 0-50°C) compared to the harsh strong alkali systems required in conventional methods. This gentleness minimizes the degradation of the beta-lactam ring and reduces the formation of impurities, resulting in a final product with purity greater than 99% and improved crystal form.
Q: Does this process require the isolation of the Cefdinir ester intermediate?
A: No, one of the key innovations in this method is the elimination of the need to isolate and dry the Cefdinir ester intermediate before hydrolysis. The process proceeds directly from the aqueous solution of the ester to the enzymatic hydrolysis step, which shortens the production cycle, reduces solvent consumption, and improves the overall yield of the final Cefdinir product.
Q: What specific reaction conditions are optimized for the enzymatic hydrolysis step?
A: The patent specifies that the enzymatic hydrolysis is most effective when the pH of the system is maintained between 5.5 and 8.5, with a preferred temperature range of 25-35°C. The use of immobilized enzymes not only facilitates easy separation and reuse but also ensures that the hydrolysis of the acetoxy imido group is complete without the need for extreme temperatures or corrosive reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced biocatalytic processes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102153566A are fully realized in practical application. We operate stringent purity specifications and maintain rigorous QC labs equipped to handle the complex analytical requirements of beta-lactam antibiotics, guaranteeing that every batch of Cefdinir meets the highest international pharmacopoeia standards.
We invite global pharmaceutical partners to collaborate with us to leverage this cutting-edge synthesis technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and quality in your antibiotic manufacturing operations.
