Advanced Green Synthesis of Cefdinir Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the production of critical antibiotics, and the synthesis of cefdinir represents a pivotal area of innovation for third-generation cephalosporins. Patent CN102020664A introduces a groundbreaking methodology that leverages alkaline ionic liquids coupled with heterogeneous solid base catalysts to streamline the acylation process of the key cephem nucleus. This approach addresses the longstanding challenges associated with traditional synthesis routes, which often suffer from harsh reaction conditions, difficult product isolation, and significant environmental burdens due to solvent waste. By integrating green chemistry principles directly into the manufacturing workflow, this technology offers a robust framework for producing high-purity cefdinir hydrochloride, as depicted in the final product structure below.

For global procurement leaders and supply chain directors, the adoption of such advanced catalytic systems translates directly into enhanced supply security and reduced operational risks. The ability to operate under milder conditions while maintaining exceptional yield and purity profiles ensures that manufacturers can meet stringent regulatory standards without compromising on throughput. As a reliable cefdinir intermediate supplier, understanding the nuances of this patented technology is essential for evaluating long-term partnerships that prioritize both economic efficiency and ecological responsibility in the competitive landscape of beta-lactam antibiotic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for cefdinir have historically relied on homogeneous base catalysts or stoichiometric amounts of reactive reagents that generate substantial quantities of saline waste and require complex downstream processing. These conventional methods often necessitate strict temperature controls, frequently dipping into cryogenic ranges to prevent the degradation of the sensitive beta-lactam ring, which significantly increases energy consumption and capital expenditure on specialized refrigeration equipment. Furthermore, the separation of the product from the reaction matrix in older technologies is notoriously difficult, often requiring multiple crystallization steps or chromatographic purification to remove trace impurities and residual catalysts that could compromise the safety profile of the final API. The reliance on volatile organic solvents and non-recyclable catalysts also poses significant environmental compliance challenges, making it increasingly difficult for manufacturers to adhere to modern green chemistry mandates and sustainability goals.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a synergistic combination of alkaline ionic liquids and a specifically formulated solid base catalyst to create a highly efficient reaction medium that overcomes these historical bottlenecks. This system allows the acylation reaction to proceed smoothly at temperatures ranging from 10°C to 60°C, eliminating the need for energy-intensive cooling while preserving the structural integrity of the cephem core. The use of a solid base catalyst, composed of gamma-alumina and magnesium hydroxide, facilitates easy separation through simple filtration or extraction, allowing the ionic liquid medium to be recovered and reused multiple times without significant loss of activity. This shift not only simplifies the post-treatment workflow but also drastically reduces the volume of three wastes generated, positioning this method as a superior choice for cost reduction in cephalosporin manufacturing and aligning perfectly with the needs of environmentally conscious supply chains.
Mechanistic Insights into Ionic Liquid-Mediated Acylation
The core of this technological advancement lies in the unique interaction between the alkaline ionic liquid and the solid base catalyst, which creates a microenvironment conducive to high-selectivity nucleophilic substitution. The ionic liquid acts as both a solvent and a promoter, stabilizing the transition state of the reaction between 7-amino-3-vinyl-3-cephem-4-carboxylic acid and the activated side-chain acid chloride. This stabilization effect minimizes unwanted side reactions, such as the hydrolysis of the beta-lactam ring or the isomerization of the vinyl group, which are common pitfalls in less controlled systems. The solid base component serves to scavenge the acid byproduct generated during the acylation, driving the equilibrium forward and ensuring complete conversion of the starting materials without the need for excessive reagent excesses that complicate purification.
Furthermore, the impurity control mechanism is inherently built into the physical properties of the catalyst system, which prevents the formation of polymeric byproducts often seen in homogeneous catalysis. The heterogeneous nature of the solid base ensures that active sites are accessible yet contained, preventing localized hotspots of high basicity that could degrade the sensitive antibiotic scaffold. For R&D directors focused on purity and impurity profiles, this means a cleaner crude product that requires fewer purification steps, directly impacting the overall process mass intensity (PMI). The ability to tune the anionic component of the ionic liquid, such as using hydroxide, acetate, or bicarbonate variants, provides an additional layer of process optimization, allowing manufacturers to fine-tune the reaction kinetics for maximum efficiency and consistent quality across different batch sizes.
How to Synthesize Cefdinir Efficiently
The implementation of this synthesis route involves a straightforward sequence of mixing, reacting, and separating that is highly amenable to standard pharmaceutical manufacturing equipment. The process begins with the suspension of the solid base catalyst in the alkaline ionic liquid, followed by the sequential addition of the cephem nucleus and the side-chain activator under controlled thermal conditions. Detailed standard operating procedures regarding specific mixing rates, addition times, and quality control checkpoints are critical for ensuring reproducibility and safety during scale-up operations. The following guide outlines the standardized synthesis steps derived from the patent data to assist technical teams in evaluating the feasibility of this route for their specific production lines.
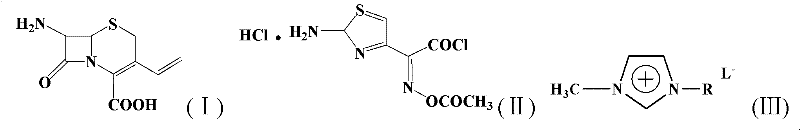
- React 7-amino-3-vinyl-3-cephem-4-carboxylic acid with the acetyl chloride derivative in alkaline ionic liquid using a solid base catalyst at 10-60°C.
- Extract the reaction mixture with toluene to separate the ionic liquid and solid catalyst for reuse.
- Hydrolyze the extract with sulfuric acid, adjust pH, and acidify with hydrochloric acid to precipitate pure cefdinir hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the transition to this ionic liquid-based synthesis offers profound advantages that extend beyond mere technical performance, directly addressing the core concerns of procurement managers and supply chain heads regarding cost stability and operational continuity. The elimination of expensive homogeneous catalysts and the ability to recycle the ionic liquid medium result in a substantial reduction in raw material costs per kilogram of produced intermediate. Moreover, the simplified workup procedure, which relies on basic extraction and precipitation rather than complex chromatography, significantly shortens the production cycle time, allowing facilities to increase throughput without additional capital investment in new reactor trains. This efficiency gain is crucial for maintaining competitive pricing in the global market while ensuring that supply commitments can be met even during periods of high demand.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the recyclability of the catalytic system and the reduction in solvent consumption. By reusing the ionic liquid and solid base across multiple batches, manufacturers can amortize the cost of these specialized materials over a much larger production volume, leading to significant cost savings over the lifecycle of the product. Additionally, the mild reaction conditions reduce energy costs associated with heating and cooling, while the high selectivity minimizes the loss of valuable starting materials to waste streams. This holistic approach to cost optimization ensures that the final cefdinir intermediate is produced with a lower cost basis, providing a buffer against market volatility in raw material pricing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of the required reagents. The solid base catalyst is composed of common industrial materials such as alumina and magnesium hydroxide, which are readily available from multiple global suppliers, reducing the risk of single-source bottlenecks. The stability of the ionic liquid system also allows for longer storage life of prepared reaction media, enabling manufacturers to maintain strategic reserves without degradation concerns. This reliability translates into reduced lead time for high-purity cephalosporins, ensuring that downstream API manufacturers receive their intermediates on schedule and can maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of heterogeneous catalysis, which avoids the mixing and heat transfer issues often encountered with viscous homogeneous systems. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the liability and disposal costs associated with chemical manufacturing. Facilities adopting this technology can demonstrate a commitment to sustainability, which is becoming a key differentiator in vendor selection processes for major pharmaceutical companies. The ease of waste treatment and the potential for solvent recovery further enhance the environmental profile, making this a future-proof solution for large-scale antibiotic production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology for cefdinir production. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and feasibility of adopting this route for industrial applications. Understanding these details is vital for stakeholders evaluating the potential integration of this method into their existing manufacturing portfolios.
Q: How does the ionic liquid system improve impurity control in cefdinir synthesis?
A: The alkaline ionic liquid combined with a solid base catalyst provides a homogeneous-like reaction environment that enhances selectivity, significantly reducing side reactions and ensuring purity levels above 99.5% without complex chromatography.
Q: Can the catalyst system be recycled for industrial production?
A: Yes, the patent specifies that both the ionic liquid and the solid base catalyst can be directly recovered after toluene extraction and reused in subsequent batches, drastically lowering material costs and waste generation.
Q: What are the temperature requirements for this synthesis route?
A: The reaction proceeds efficiently within a mild temperature range of 10°C to 60°C, eliminating the need for extreme cryogenic conditions often required in traditional beta-lactam acylation processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ionic liquid-mediated synthesis route for cefdinir and possess the technical expertise to bring this innovative process to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cefdinir intermediate meets the highest international standards, providing our partners with the confidence they need to rely on us for their critical supply chains.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your production costs and enhance your product quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term business goals in the pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
