Revolutionizing Primary Amine Production: A Deep Dive into Silver-Catalyzed N-H Insertion Technology
Revolutionizing Primary Amine Production: A Deep Dive into Silver-Catalyzed N-H Insertion Technology
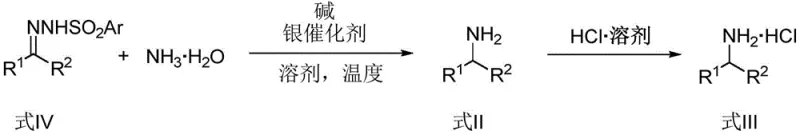
The landscape of organic synthesis is constantly evolving, driven by the relentless pursuit of more efficient, cost-effective, and sustainable methodologies for constructing essential chemical building blocks. A groundbreaking development in this arena is detailed in Chinese Patent CN115448841A, which discloses a novel method for synthesizing primary amines utilizing inexpensive ammonia water as the nitrogen source. This technology represents a paradigm shift from traditional amination strategies, leveraging a sophisticated transition metal silver catalytic system to facilitate carbene N-H insertion reactions. By employing either diazo compounds or benzenesulfonyl hydrazones as carbene precursors, this innovative approach enables the direct construction of arylmethylamines, aliphatic amines, and α-amino acid esters with remarkable efficiency. The significance of this patent lies not only in its chemical elegance but also in its profound implications for industrial scalability, offering a streamlined pathway that circumvents the high costs and safety hazards associated with conventional reagents.
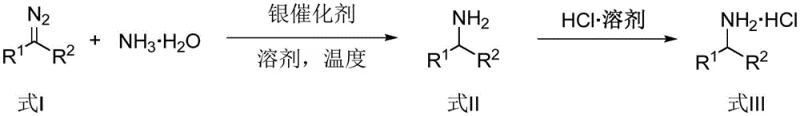
For R&D directors and process chemists, the ability to utilize aqueous ammonia directly is a substantial technical breakthrough. Historically, the presence of water in carbene insertion reactions has been a formidable challenge due to the competitive O-H insertion reaction, which often leads to alcohol byproducts and reduced yields. Furthermore, ammonia typically acts as a poison to many transition metal catalysts through strong coordination. However, the specific silver catalyst system described in this patent, TpBr3Ag, demonstrates exceptional tolerance and selectivity. It successfully navigates these thermodynamic and kinetic hurdles, ensuring that the carbene intermediate preferentially inserts into the N-H bond of ammonia rather than reacting with the solvent or water molecules. This high level of chemoselectivity is critical for maintaining product purity and minimizing downstream purification burdens, which are key metrics for any viable commercial process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of primary aliphatic and aromatic amines has relied heavily on methods such as the reductive amination of carbonyl compounds or the hydrogenation of nitriles. While these methods are well-established, they suffer from significant drawbacks that impact both economic viability and operational safety. Reductive amination often requires expensive reducing agents like sodium cyanoborohydride or high-pressure hydrogenation equipment, which increases capital expenditure and operational complexity. Moreover, controlling the selectivity to stop at the primary amine stage without over-alkylation to secondary or tertiary amines can be challenging, leading to complex impurity profiles that require rigorous chromatographic separation. Similarly, nitrile hydrogenation typically demands harsh reaction conditions, including elevated temperatures and pressures, along with specialized catalysts that may be sensitive to poisoning. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115448841A offers a streamlined, one-pot solution that dramatically simplifies the synthetic workflow. By utilizing N-tosylhydrazones or diazo compounds as stable and accessible carbene precursors, the process avoids the need for pre-formed organometallic reagents or high-pressure infrastructure. The reaction proceeds under mild thermal conditions, typically ranging from 60°C to 100°C, which significantly reduces energy consumption compared to traditional high-temperature hydrogenation processes. Furthermore, the use of ammonia water as a bulk industrial commodity eliminates the dependency on specialized, costly nitrogen sources. This approach not only lowers the raw material cost baseline but also enhances the safety profile of the operation by avoiding the handling of anhydrous ammonia gas or explosive azide reagents. The versatility of this method is further evidenced by its compatibility with a wide range of functional groups, allowing for the synthesis of complex amine structures without the need for extensive protecting group strategies.
Mechanistic Insights into TpBr3Ag-Catalyzed Carbene N-H Insertion
The core of this technological advancement lies in the unique properties of the TpBr3Ag catalyst, a tris(pyrazolyl)borate silver complex. Mechanistically, the reaction initiates with the decomposition of the diazo compound or the in situ generation of a diazo species from the tosylhydrazone precursor in the presence of a base. The silver center coordinates with the diazo functionality, facilitating the extrusion of nitrogen gas to generate a highly electrophilic silver-carbene intermediate. This transient species is the key driver of the transformation. In a typical aqueous environment, such a reactive intermediate would be rapidly quenched by water molecules to form ketones or alcohols via O-H insertion. However, the steric and electronic environment provided by the brominated pyrazolyl ligands on the silver atom modulates the reactivity of the carbene. This modulation creates a kinetic preference for the nucleophilic attack by ammonia, despite the vast excess of water present in the ammonia solution. The result is a highly selective N-H insertion event that forms the C-N bond directly.
From an impurity control perspective, this mechanism offers distinct advantages. The primary side reaction to monitor is the dimerization of the carbene precursor or the aforementioned O-H insertion. However, the patent data indicates that by optimizing the molar ratios of ammonia to precursor (typically 50:1 to 100:1), the formation of these byproducts is effectively suppressed. The high concentration of ammonia ensures that the probability of the carbene encountering an ammonia molecule is maximized before it can react with other species. Additionally, the use of a mild base like cesium carbonate helps to maintain the reaction pH and facilitate the decomposition of the hydrazone without promoting unwanted side reactions. This precise control over the reaction microenvironment ensures that the final crude product contains minimal impurities, often allowing for purification via simple crystallization of the hydrochloride salt rather than resource-intensive column chromatography.
How to Synthesize Primary Amines Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The process generally involves charging a reaction vessel with the silver catalyst and base under an inert atmosphere to prevent oxidation of sensitive intermediates. Subsequently, the carbene precursor and aqueous ammonia are introduced into the organic solvent, and the mixture is heated to the specified temperature range. The reaction progress is monitored until the starting material is fully consumed, after which a straightforward workup involving filtration and acidification yields the desired amine salt. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, please refer to the standardized protocol below.
- Prepare the reaction vessel by adding the TpBr3Ag catalyst (10 mol%) and a base such as cesium carbonate (1.5 equiv) under an inert atmosphere.
- Introduce the carbene precursor (diazo compound or N-tosylhydrazone) and aqueous ammonia solution into the reactor containing the dry organic solvent.
- Heat the mixture to 60-80°C for 12-24 hours while protecting from light, then isolate the product via filtration and acidification to obtain the amine hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver-catalyzed amination technology presents a compelling value proposition centered on cost optimization and supply security. The most immediate benefit is the drastic reduction in raw material costs. By replacing expensive amines, azides, or specialized reducing agents with commodity-grade ammonia water, the direct material cost of the synthesis is significantly lowered. Ammonia water is produced on a massive global scale, ensuring a stable and reliable supply chain that is less susceptible to the market volatility often seen with fine chemical reagents. This stability translates directly into more predictable pricing models for long-term contracts, allowing procurement teams to negotiate better terms and secure margins. Furthermore, the simplicity of the reaction setup reduces the burden on manufacturing infrastructure, as it does not require specialized high-pressure reactors or cryogenic cooling systems often needed for alternative amination methods.
- Cost Reduction in Manufacturing: The economic impact of this process extends beyond just raw materials. The elimination of expensive transition metals like palladium or rhodium, which are commonly used in cross-coupling amination reactions, results in substantial savings on catalyst costs. Silver, while a precious metal, is used in relatively low loadings (around 10 mol%) and the ligand system is robust. Moreover, the simplified purification process, which often relies on precipitation and filtration rather than complex distillation or chromatography, reduces solvent consumption and waste disposal costs. This leaner operational footprint contributes to a lower overall cost of goods sold (COGS), making the final API intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: Reliability is a cornerstone of modern supply chain management, and this technology enhances it by diversifying the sourcing of nitrogen inputs. Since ammonia is a fundamental chemical feedstock available from numerous suppliers worldwide, the risk of supply disruption due to single-source dependency is minimized. Additionally, the mild reaction conditions reduce the risk of safety incidents that could halt production. The process is tolerant to a wide range of substrates, meaning that the same catalytic system can be used to manufacture a diverse portfolio of amine intermediates without needing to retool or requalify entirely new chemical processes. This flexibility allows manufacturers to respond more agilely to changing market demands.
- Scalability and Environmental Compliance: Scaling chemical processes from the gram scale to multi-ton production is often fraught with challenges, but this method demonstrates excellent scalability potential. The reaction exotherms are manageable, and the use of common organic solvents like dichloroethane or toluene fits within existing solvent recovery infrastructures. From an environmental standpoint, the atom economy of the reaction is favorable, and the avoidance of stoichiometric metal waste (common in reduction reactions) aligns with green chemistry principles. This compliance with increasingly stringent environmental regulations reduces the regulatory burden and potential fines, ensuring uninterrupted production continuity and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed amination technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing manufacturing workflows.
Q: What are the key advantages of using ammonia water over traditional amination reagents?
A: Using ammonia water significantly reduces raw material costs compared to expensive amines or azides. It also simplifies the supply chain as ammonia is a bulk industrial commodity, enhancing overall process economics and sustainability.
Q: How does the TpBr3Ag catalyst improve chemoselectivity in the presence of water?
A: The TpBr3Ag silver complex effectively promotes carbene N-H insertion over the competing O-H insertion reaction with water. This unique selectivity allows the use of aqueous ammonia directly, avoiding the need for hazardous anhydrous ammonia gas.
Q: Is this method suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the protocol operates under mild temperatures (60-100°C) and uses standard organic solvents, making it highly scalable. The simple workup procedure involving filtration and crystallization further supports robust commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Primary Amine Supplier
The technological potential of silver-catalyzed N-H insertion for primary amine synthesis is immense, offering a pathway to high-value intermediates with superior cost and efficiency profiles. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative patent technologies into commercial reality. As a leading CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of primary amine intermediate meets the highest industry standards required for pharmaceutical applications.
We invite you to explore how this advanced synthesis method can optimize your supply chain and reduce manufacturing costs. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific target molecules. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to cutting-edge synthetic methodologies combined with the reliability of a seasoned manufacturing partner, ensuring your projects move from concept to commercial success without delay.
