Revolutionizing Primary Amine Production with Advanced Ternary Nickel-Ruthenium Catalysts
Revolutionizing Primary Amine Production with Advanced Ternary Nickel-Ruthenium Catalysts
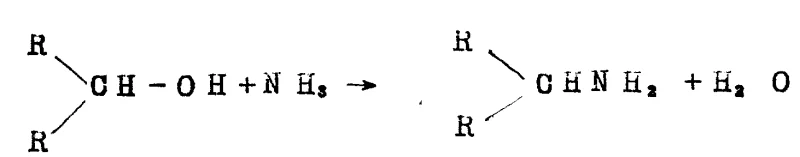
The landscape of fine chemical synthesis is constantly evolving, driven by the relentless pursuit of higher purity, improved yields, and more sustainable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN88101658A, which introduces a sophisticated method for producing amines through reductive amination. This technology addresses a longstanding challenge in the industry: the difficulty of selectively producing primary amines without generating significant quantities of secondary and tertiary amine by-products. By leveraging a novel ternary catalytic composition comprising nickel, ruthenium, and a specific third transition metal, this process offers a transformative approach to amine synthesis that is particularly relevant for the manufacture of pharmaceutical intermediates and specialty chemicals.
The core innovation lies in the precise engineering of the catalyst surface. Traditional methods often rely on simple nickel or cobalt catalysts, which, while active, lack the necessary selectivity for high-value applications. The patented process described in CN88101658A utilizes a synergistic combination of metals supported on a robust carrier, such as maca-alumina. This configuration not only enhances the conversion rates of alcohols, aldehydes, or ketones but also fundamentally alters the reaction pathway to favor the formation of the primary amine. For R&D directors and process engineers, this represents a significant opportunity to streamline synthesis routes and reduce the burden on downstream purification systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of amines via reductive amination has been plagued by selectivity issues. Conventional catalysts, typically based on nickel or cobalt, often facilitate a cascade of reactions where the desired primary amine, once formed, acts as a nucleophile and reacts further with the remaining alcohol or aldehyde substrate. This leads to the formation of secondary and tertiary amines, which are often difficult and expensive to separate from the target product. In many cases, this lack of selectivity results in complex distillation trains, high energy consumption, and significant material loss. Furthermore, older catalyst formulations frequently suffer from rapid deactivation or require harsh operating conditions that compromise the integrity of sensitive functional groups present in complex organic molecules.
The Novel Approach
The methodology outlined in patent CN88101658A breaks this paradigm by introducing a ternary metal system. By incorporating ruthenium and a promoter metal from the second or third transition series—such as palladium, rhenium, or iridium—into a nickel base, the catalyst achieves a level of specificity previously unattainable. This advanced formulation effectively suppresses the consecutive alkylation reactions that lead to by-product formation. The result is a process that delivers high yields of primary amines directly from the reactor, minimizing the need for extensive purification. This approach is not merely an incremental improvement but a fundamental shift in how reductive amination is approached, offering a cleaner, more efficient pathway for synthesizing critical amine building blocks.
Mechanistic Insights into Ternary Catalyst Reductive Amination
The success of this catalytic system can be attributed to the unique electronic and geometric properties imparted by the multi-metal composition. The presence of ruthenium and the third transition metal modifier likely alters the adsorption characteristics of the intermediate imine species on the catalyst surface. It is hypothesized that these promoters facilitate the rapid hydrogenation of the imine to the primary amine while simultaneously reducing the affinity of the primary amine for the active sites, thereby preventing it from undergoing further reaction. This delicate balance ensures that the primary amine desorbs from the catalyst before it can react again, preserving the integrity of the product. Such mechanistic control is essential for maintaining high purity standards required in pharmaceutical and electronic chemical applications.
Furthermore, the versatility of this catalytic system is demonstrated by its compatibility with a broad spectrum of substrates. As illustrated by the general formula for suitable alkanols, the process accommodates various aliphatic structures, ranging from simple short-chain alcohols to more complex long-chain variants. The ability to handle diverse substrates without compromising selectivity makes this technology highly adaptable. Whether the feedstock is a simple alkanol or a functionalized alcohol containing ether or amine groups, the ternary catalyst maintains its efficacy. This flexibility allows manufacturers to utilize a single catalytic platform for multiple product lines, simplifying inventory management and operational protocols.
How to Synthesize Primary Amines Efficiently
Implementing this advanced reductive amination process requires careful attention to catalyst preparation and reaction conditions. The patent details a specific sequential impregnation method to ensure the optimal distribution of the active metal phases on the support. The process begins with the impregnation of the carrier with nickel, followed by calcination to stabilize the nickel oxide phase. Subsequently, the promoters (ruthenium and the third metal) are introduced. This stepwise approach is critical for achieving the synergistic effects observed in the final catalyst. Once prepared, the catalyst is activated under hydrogen flow at elevated temperatures to generate the active metallic species ready for the amination reaction.
- Prepare the catalyst support by impregnating maca-alumina with a nickel compound solution, followed by calcination at 550-600°C to fix the nickel component.
- Sequentially impregnate the calcined support with solutions of a second or third row transition metal (e.g., Pd, Re, Ir) and a ruthenium compound to form the ternary active phase.
- Activate the final catalyst composition by heating above 280°C in a hydrogen atmosphere for at least 3 hours prior to introducing the alcohol and ammonia reactants.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ternary catalyst technology translates into tangible strategic advantages. The primary benefit stems from the drastic improvement in selectivity. By minimizing the formation of secondary and tertiary amine by-products, the process significantly reduces the complexity and cost of downstream purification. In traditional processes, separating close-boiling amine mixtures can be energy-intensive and result in substantial yield losses. The high selectivity of the Ni-Ru-M catalyst system mitigates these issues, leading to a more streamlined production workflow and lower overall manufacturing costs.
- Cost Reduction in Manufacturing: The enhanced selectivity of the ternary catalyst directly impacts the bottom line by reducing waste and energy consumption. Because the reaction produces fewer unwanted by-products, the load on distillation columns and separation units is significantly decreased. This reduction in processing intensity leads to lower utility costs and less solvent usage. Additionally, the high activity of the catalyst allows for potentially milder operating conditions or shorter reaction times, further contributing to operational efficiency and cost savings without compromising product quality.
- Enhanced Supply Chain Reliability: The robustness of the maca-alumina supported catalyst ensures consistent performance over extended periods. In industrial settings, catalyst longevity is a critical factor for supply chain continuity. A catalyst that maintains its activity and selectivity over long cycles reduces the frequency of reactor shutdowns for catalyst replacement or regeneration. This reliability minimizes production disruptions and ensures a steady flow of high-purity amine products, which is essential for meeting the rigorous delivery schedules of downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, suitable for both batch and continuous operations. The use of a heterogeneous catalyst simplifies product separation, as the solid catalyst can be easily filtered or retained in a fixed bed, eliminating the need for complex homogeneous catalyst removal steps. This feature not only facilitates easier scale-up from pilot to commercial production but also aligns with modern environmental standards by reducing the generation of hazardous waste streams associated with catalyst disposal and solvent recovery.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this reductive amination technology. These insights are derived from the specific embodiments and comparative data presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: How does the ternary catalyst improve selectivity compared to traditional nickel catalysts?
A: The addition of ruthenium and a third transition metal (like palladium, rhenium, or iridium) to the nickel base significantly suppresses the over-alkylation reaction. This prevents the newly formed primary amine from reacting further with the alcohol to form unwanted secondary and tertiary amines, thereby drastically increasing the yield of the desired primary amine product.
Q: What types of alcohol substrates are compatible with this reductive amination process?
A: The process is highly versatile and accommodates a wide range of aliphatic and aromatic alcohols. This includes monohydric alkanols (such as isopropanol), dihydric alcohols (glycols like ethylene glycol), and even complex polyether polyols, provided the total carbon count remains within manageable limits for the specific application.
Q: Can this catalyst system be scaled for industrial continuous production?
A: Yes, the catalyst is designed for robustness and can be utilized in both batch autoclaves and continuous fixed-bed reactors. The use of a stable maca-alumina support ensures mechanical strength and thermal stability, which are critical prerequisites for long-cycle industrial operations and commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Primary Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced catalytic technologies in driving the next generation of chemical manufacturing. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the ternary catalyst reductive amination can be successfully translated from the laboratory to the plant floor. We are committed to delivering high-purity primary amines that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to explore how our expertise can optimize your supply chain. By leveraging our deep understanding of catalytic amination processes, we can offer a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can enhance your product quality and operational efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
