Advanced Synthetic Route for High-Purity Cefepime Hydrochloride Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics like Cefepime Hydrochloride, a fourth-generation cephalosporin known for its broad spectrum and stability against beta-lactamases. Patent CN101735251A introduces a significant technological advancement in this domain by detailing a refined synthetic method that addresses historical inefficiencies in producing the key intermediate, 7-MPCA, and the final active pharmaceutical ingredient. This innovation centers on a strategic modification of the alkylation step using a pre-formed quaternary ammonium salt intermediate and employs phase transfer catalysis to optimize the subsequent acylation reaction. For global procurement and R&D teams, this patent represents a viable route to enhance production economics while maintaining the rigorous quality standards required for sterile injectable formulations. By shifting away from direct alkylation methods that often suffer from uncontrolled reactivity, this process offers a more predictable and scalable solution for the commercial manufacture of high-value beta-lactam antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Cefepime often rely on the direct reaction of 7-aminocephalosporanic acid (7-ACA) derivatives with N-methylpyrrolidine, a process fraught with chemical challenges due to the inherent properties of the amine. The nitrogen atom in N-methylpyrrolidine possesses a high electron cloud density, making it excessively reactive and prone to generating numerous side products during the electrophilic substitution on the cephalosporin nucleus. These side reactions not only consume valuable raw materials but also complicate the downstream purification processes, leading to lower overall yields and potential impurity profiles that are difficult to control. Furthermore, conventional methods frequently struggle with the acylation step, where the reaction between the amine intermediate and the activated ester can be sluggish or incomplete without effective phase management, resulting in suboptimal conversion rates. The cumulative effect of these inefficiencies is a manufacturing process that is costly, environmentally burdensome due to waste generation, and less reliable for meeting the high-volume demands of the global antibiotic market.
The Novel Approach
The methodology disclosed in the patent fundamentally alters the reaction dynamics by introducing a pre-formation step for the quaternary ammonium salt intermediate before it encounters the cephalosporin nucleus. By reacting N-methylpyrrolidine with iodotrimethylsilane separately to create a stable quaternary salt solution, the process effectively tames the high reactivity of the nitrogen atom, allowing for a controlled electrophilic substitution on the silyl-protected 7-ACA. This strategic intervention minimizes side reactions and has been shown to increase the yield of the first synthetic step by approximately 7%, a substantial gain in industrial chemistry. Additionally, the novel approach integrates a phase transfer catalyst during the N-acylation reaction in the second step, which dramatically improves the interaction between the aqueous and organic phases. This enhancement boosts the conversion rate by roughly 5%, ensuring that the valuable AE-active ester is utilized efficiently and that the final product achieves a purity level exceeding 99%, thereby streamlining the path to commercial-grade Cefepime Hydrochloride.
Mechanistic Insights into Silyl Protection and Phase Transfer Catalysis
The core chemical ingenuity of this process lies in the dual application of silyl protection and phase transfer catalysis to manage reactivity and solubility issues simultaneously. Initially, 7-ACA undergoes protection of both its carboxylic acid and amino groups using hexamethyldisilazane (HMDS) and iodotrimethylsilane (TMSI), creating a shielded intermediate that prevents unwanted polymerization or degradation of the sensitive beta-lactam ring during the harsh alkylation conditions. Simultaneously, the formation of the N-methylpyrrolidine quaternary ammonium salt acts as a 'masked' nucleophile; upon mixing with the protected 7-ACA in the presence of N,N-diethylaniline, it facilitates a clean electrophilic attack at the 3-position of the cephem nucleus. This mechanism bypasses the kinetic traps associated with free amine alkylation, ensuring that the quaternary ammonium side chain is installed with high regioselectivity. Following deprotection and salt formation, the resulting 7-MPCA is ready for the critical acylation step where the side chain defining Cefepime's activity is attached.
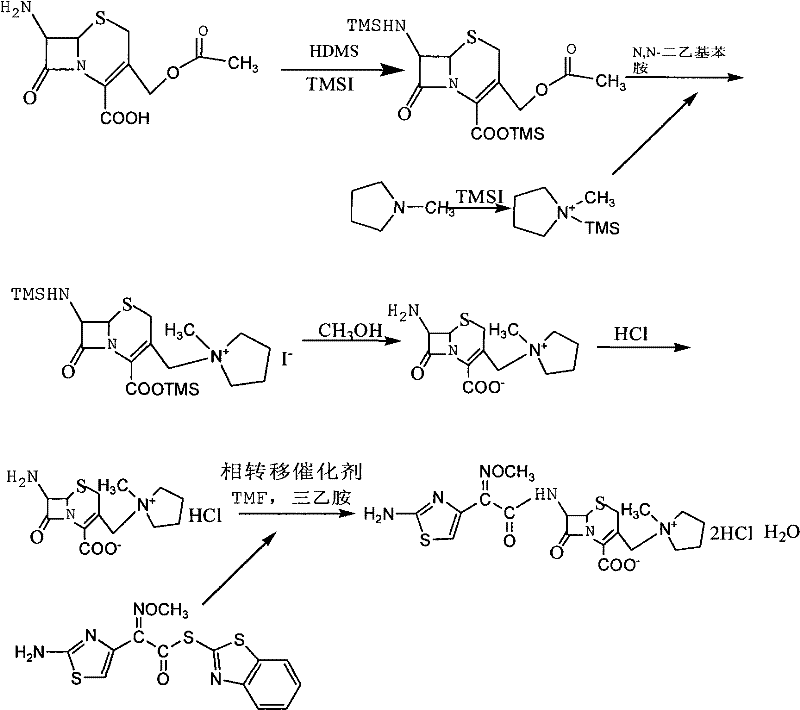
In the second stage of the synthesis, the reaction environment shifts to a biphasic system comprising water and an organic solvent such as tetrahydrofuran or methylene chloride. The addition of a phase transfer catalyst, such as tetramethylammonium chloride or tributylamine, is the pivotal mechanistic element here. These catalysts function by transporting the anionic species of the 7-MPCA from the aqueous phase into the organic phase where the AE-active ester is dissolved. This transport mechanism drastically increases the local concentration of reactants in the organic layer, overcoming the diffusion limitations that typically plague biphasic reactions. The result is a rapid and complete N-acylation reaction that proceeds with high fidelity, minimizing the formation of hydrolysis byproducts of the active ester. This mechanistic control is essential for maintaining the stereochemical integrity of the Z-oxime group in the side chain, which is critical for the biological activity of the final antibiotic.
How to Synthesize 7-MPCA and Cefepime Efficiently
The practical execution of this synthesis requires precise control over temperature and stoichiometry to maximize the benefits of the patented intermediates. The process begins with the silylation of 7-ACA under reflux conditions, followed by the separate preparation of the quaternary ammonium salt at controlled temperatures to prevent decomposition. Once the 7-MPCA intermediate is isolated via crystallization, it serves as the substrate for the final coupling reaction. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures required to replicate this high-yield process, are outlined in the guide below.
- Protect 7-ACA with HMDS and TMSI, then react with a pre-formed N-methylpyrrolidine quaternary ammonium salt intermediate to synthesize 7-MPCA.
- React 7-MPCA with AE-active ester in a biphasic system using a phase transfer catalyst to facilitate N-acylation.
- Perform acidification and crystallization to isolate the final Cefepime Hydrochloride product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible economic and operational benefits that extend beyond simple yield metrics. The primary advantage lies in the significant reduction of raw material waste; by suppressing side reactions in the initial alkylation step, the consumption of expensive starting materials like 7-ACA and N-methylpyrrolidine is optimized, directly lowering the variable cost of goods sold. Furthermore, the enhanced conversion rates in the acylation step mean that less AE-active ester is required per kilogram of final product, contributing to substantial cost savings in the most expensive portion of the synthesis. The process also simplifies the purification workflow, as the higher crude purity reduces the burden on downstream crystallization and washing steps, leading to faster batch cycle times and increased throughput capacity without the need for additional capital investment in equipment.
- Cost Reduction in Manufacturing: The elimination of excessive side reactions translates directly into improved material efficiency, allowing manufacturers to achieve higher output from the same input of raw materials. By avoiding the need for complex chromatographic purifications often required to remove alkylation byproducts, the process significantly lowers solvent consumption and waste disposal costs. This streamlined approach ensures that the cost reduction in pharmaceutical intermediates manufacturing is realized through both yield improvement and operational simplification, making the final API more price-competitive in tender markets.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents such as HMDS and common phase transfer catalysts mitigates the risk of supply disruptions associated with exotic or highly specialized catalysts. The robustness of the reaction conditions, which tolerate standard industrial solvents and temperatures, ensures consistent batch-to-batch performance, a critical factor for maintaining uninterrupted supply to downstream formulation partners. This reliability reduces lead time for high-purity pharmaceutical intermediates by minimizing the occurrence of failed batches or out-of-specification results that would otherwise delay shipment schedules.
- Scalability and Environmental Compliance: The synthetic route is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing unit operations like reflux, extraction, and crystallization that are easily transferred from pilot plant to multi-ton production scales. The reduction in side products also implies a cleaner waste stream, facilitating easier compliance with increasingly stringent environmental regulations regarding effluent treatment. This scalability ensures that suppliers can rapidly respond to surges in demand for fourth-generation cephalosporins without compromising on quality or environmental safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for Cefepime Hydrochloride. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on yield expectations, purity profiles, and process mechanics. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement specialists assessing supplier capabilities.
Q: How does the quaternary ammonium salt intermediate improve the synthesis of 7-MPCA?
A: By pre-forming the quaternary ammonium salt from N-methylpyrrolidine and iodotrimethylsilane, the method overcomes the high electron cloud density and excessive reactivity of the nitrogen atom, significantly reducing side reactions and increasing the yield of the first step by approximately 7%.
Q: What is the role of the phase transfer catalyst in the acylation step?
A: The phase transfer catalyst facilitates the movement of reactants into the organic phase during the N-acylation reaction between 7-MPCA and AE-active ester, enhancing the conversion rate by about 5% and improving overall product yield.
Q: What purity levels can be achieved with this synthetic method?
A: This optimized process allows for the production of Cefepime Hydrochloride with a purity exceeding 99%, meeting stringent pharmaceutical standards for fourth-generation cephalosporin antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and pure synthetic routes in the production of life-saving antibiotics like Cefepime. Our technical team has extensively analyzed advanced methodologies, including the quaternary ammonium salt intermediate approach, to ensure our manufacturing processes align with the highest standards of efficiency and quality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver consistent results. Our commitment to stringent purity specifications and the operation of rigorous QC labs ensures that every batch of Cefepime Hydrochloride we supply meets the exacting requirements of global regulatory bodies, providing our partners with absolute confidence in product quality.
We invite international pharmaceutical companies and generic drug manufacturers to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your transition to a more efficient Cefepime supply source is seamless, compliant, and economically advantageous.
