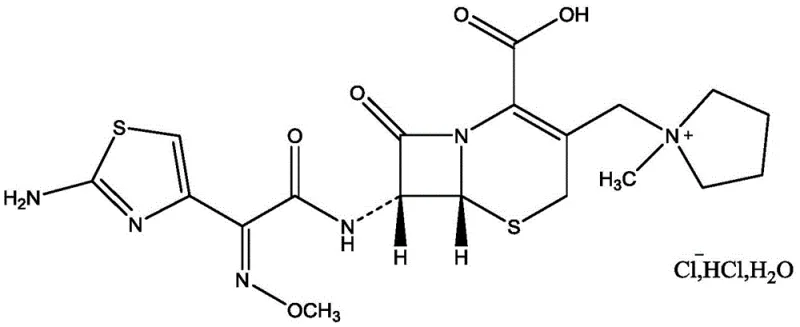
Advanced Purification Technology for Cefepime Hydrochloride: Scaling High-Purity Antibiotic Production
Advanced Purification Technology for Cefepime Hydrochloride: Scaling High-Purity Antibiotic Production
The pharmaceutical industry constantly seeks robust methodologies to enhance the purity and stability of critical beta-lactam antibiotics, particularly when dealing with complex salt forms that pose purification challenges. Patent CN109776572B introduces a groundbreaking approach to the purification of cefepime hydrochloride, specifically addressing the technical hurdles associated with converting cefepime arginine hydrochloride into the final active pharmaceutical ingredient. This innovation is pivotal for manufacturers aiming to optimize their production lines for high-purity API intermediates, as it offers a pathway to recycle off-specification materials that would otherwise be discarded, thereby aligning with modern sustainability goals and cost-efficiency mandates. The core of this technology lies in a sophisticated complexation-crystallization sequence that effectively removes arginine residues and color impurities without relying on expensive chromatographic columns. By leveraging specific complexing agents like sodium dodecyl benzene sulfonate, the process achieves a level of separation precision that traditional activated carbon adsorption methods cannot match. For R&D directors and procurement managers alike, understanding the nuances of this patent is essential for evaluating potential partnerships with a reliable API intermediate supplier who can deliver consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of cefepime hydrochloride has relied heavily on activated carbon adsorption followed by chromatographic column separation or simple crystallization from organic solvents. While these methods can achieve acceptable purity levels for laboratory-scale synthesis, they often fail to meet the rigorous demands of large-scale industrial production due to significant yield losses and high operational costs. Traditional activated carbon treatment, for instance, is non-selective and can adsorb a portion of the active pharmaceutical ingredient along with the impurities, leading to reduced overall recovery rates that impact the economic viability of the manufacturing process. Furthermore, chromatographic separation, although effective for high-resolution purification, introduces substantial bottlenecks in terms of throughput and solvent consumption, making it less desirable for cost reduction in pharmaceutical manufacturing where margin compression is a constant pressure. The reliance on these conventional techniques also limits the ability to effectively recycle raw materials that do not meet initial quality standards, resulting in increased waste generation and higher environmental compliance burdens for production facilities. Additionally, the stability of the beta-lactam ring can be compromised during prolonged exposure to the conditions required for these older purification methods, potentially leading to the formation of degradation products that complicate the impurity profile.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes a targeted complexation strategy followed by a controlled two-stage crystallization process to overcome the inefficiencies of legacy methods. By introducing a specific complexing agent into the dissolved cefepime arginine hydrochloride solution, the process selectively precipitates arginine complexes, allowing for their physical removal via filtration before the final product crystallization begins. This pre-purification step significantly reduces the impurity load entering the crystallization phase, which in turn enhances the quality of the final crystals and minimizes the need for reprocessing. The method employs a dispersing agent mixture, such as acetone and ethanol, to manage crystal growth kinetics, ensuring that the resulting particles are uniform and free from agglomeration, which is critical for downstream processing and formulation. Moreover, the ability to adjust pH levels precisely at different stages allows for the optimization of solubility parameters, ensuring that the cefepime hydrochloride remains stable while impurities are systematically excluded from the crystal lattice. This approach not only improves the yield and purity of the final product but also simplifies the overall workflow, making it highly suitable for the commercial scale-up of complex antibiotics where consistency and efficiency are paramount.
Mechanistic Insights into Complexation-Crystallization Purification
The mechanistic foundation of this purification technology rests on the selective interaction between the complexing agent and the arginine moiety of the starting material. When sodium dodecyl benzene sulfonate or dodecyl benzene sulfonic acid is introduced into the aqueous solution of cefepime arginine hydrochloride, it forms an insoluble complex with the arginine component due to electrostatic interactions and hydrophobic effects. This reaction occurs optimally when the molar ratio of the complexing agent to cefepime is maintained between 1.05:1 and 1.10:1, ensuring that there is a slight excess of the agent to drive the complexation equilibrium towards completion without introducing excessive reagent residues. The formation of this precipitate effectively sequesters the arginine, which is a major impurity source in this specific synthesis route, allowing it to be filtered out as a solid cake while the cefepime remains in the filtrate. This step is crucial because it addresses the root cause of impurity rather than merely masking it, leading to a significant reduction in the total impurity content of the final product. The subsequent decolorization step, using activated carbon or clay at controlled temperatures of 10-20°C, further refines the solution by removing organic color bodies and trace particulates without degrading the thermally sensitive beta-lactam structure.
Following the removal of the arginine complex, the purification proceeds through a sophisticated crystallization mechanism driven by pH adjustment and solvent manipulation. The pH of the filtrate is lowered to a range of 1.0-3.0 using hydrochloric acid, which protonates the cefepime molecule and drastically reduces its solubility in the aqueous-organic solvent mixture. The addition of a dispersing agent, typically a mixed solvent of acetone and acetonitrile or acetone and ethanol, serves to lower the dielectric constant of the medium and promote nucleation. Seed crystals of cefepime hydrochloride are introduced to control the nucleation rate, preventing the formation of amorphous solids and ensuring the growth of well-defined crystalline structures. A secondary crystallization step involves the addition of a crystallization agent and cooling the mixture to 0-5°C, which maximizes the recovery of the product from the mother liquor. This multi-stage crystallization ensures that any cefepime that remained dissolved during the primary crystallization is recovered, thereby boosting the overall yield while maintaining high purity standards. The rigorous control over temperature and solvent ratios throughout this process guarantees that the impurity profile, including the cefepime E isomer, remains well below regulatory thresholds.
How to Synthesize Cefepime Hydrochloride Efficiently
Implementing this synthesis route requires precise adherence to the operational parameters defined in the patent to ensure reproducibility and quality. The process begins with the dissolution of the raw material in water under controlled temperature conditions, followed by the critical complexation step that defines the efficacy of the purification. Operators must carefully monitor the pH adjustments and the addition rates of the complexing agents to avoid local supersaturation that could lead to oiling out or impure precipitates. The detailed standardized synthesis steps involve specific ratios of solvents to mass, such as a water-to-solid ratio of 2-5:1 mL/g, which are essential for maintaining the correct viscosity and mass transfer characteristics during the reaction.
- Dissolve cefepime arginine hydrochloride in water at 20-25°C and adjust pH to 4.0-6.0 using mineral acid to ensure complete solubility and stability.
- Add a complexing agent such as sodium dodecyl benzene sulfonate at a molar ratio of 1.05-1.10: 1 to precipitate arginine complexes, then filter to remove impurities.
- Perform secondary crystallization by adjusting pH to 1.0-3.0, adding dispersing agents like acetone-ethanol mixtures, and cooling to 0-5°C to harvest high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this purification technology offers substantial benefits that directly address the pain points of procurement managers and supply chain heads in the pharmaceutical sector. The ability to utilize cefepime arginine hydrochloride that does not meet initial quality standards as a raw material represents a significant opportunity for cost reduction in antibiotic manufacturing. By converting off-spec intermediates into high-quality final products, manufacturers can drastically reduce raw material waste and lower the overall cost of goods sold, which is a critical factor in competitive bidding for generic drug contracts. This recycling capability also enhances supply chain reliability by reducing dependence on the availability of pristine starting materials, thereby mitigating the risk of production stoppages due to raw material shortages. Furthermore, the elimination of complex chromatographic steps simplifies the manufacturing process, leading to shorter production cycles and reduced lead time for high-purity antibiotics. The use of common organic solvents like acetone and ethanol, which are easily recyclable, further contributes to operational efficiency and environmental compliance, reducing the burden of hazardous waste disposal.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chromatographic resin and reduces solvent consumption through efficient recycling protocols. By recovering product from mother liquors via secondary crystallization, the overall yield is maximized, which directly translates to lower unit costs. The removal of the arginine moiety through complexation avoids the loss of active ingredient often associated with non-selective adsorption methods, ensuring that every gram of raw material contributes to the final output. This efficiency allows for a more competitive pricing structure without compromising on the quality or purity specifications required by regulatory bodies.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in raw material quality ensures a consistent output, which is vital for maintaining long-term supply agreements with global pharmaceutical companies. The simplified unit operations reduce the complexity of the production line, minimizing the risk of equipment failure or process deviations that could disrupt supply. Additionally, the use of widely available reagents and solvents means that the supply chain is less vulnerable to geopolitical or logistical disruptions affecting specialized chemicals. This stability is crucial for supply chain heads who need to guarantee continuous availability of critical antibiotics to meet market demand.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing standard reactors and filtration equipment that are common in existing pharmaceutical facilities. The energy-saving nature of the low-temperature crystallization steps reduces the carbon footprint of the manufacturing process, aligning with increasingly strict environmental regulations. The ability to treat and recycle solvents minimizes the generation of hazardous waste, simplifying the permitting process and reducing environmental liability. This makes the technology not only economically attractive but also sustainable for long-term industrial adoption.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the patent data to provide clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs.
Q: How does this method handle off-spec cefepime arginine hydrochloride?
A: The process is specifically designed to recycle cefepime arginine hydrochloride that does not meet injection quality standards, converting it into high-purity cefepime hydrochloride through complexation and crystallization, thereby reducing waste and raw material costs.
Q: What is the role of the complexing agent in this purification?
A: The complexing agent, typically sodium dodecyl benzene sulfonate, selectively binds with arginine to form a precipitate. This allows for the effective separation of arginine from the cefepime molecule before the final crystallization step, ensuring high purity and low impurity levels in the final product.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method avoids complex chromatographic separation and uses standard unit operations like dissolution, filtration, and crystallization. The use of recyclable organic solvents and simple pH adjustments makes it highly scalable and energy-efficient for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced purification technologies to meet the evolving demands of the global pharmaceutical market. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the complexation-crystallization method for Cefepime Hydrochloride are translated into reliable industrial reality. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest international standards for antibiotic intermediates. Our infrastructure is designed to handle complex chemical transformations with precision, offering our partners a secure and efficient route to market for their critical drug substances.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational efficiencies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our capabilities can support your strategic goals. By partnering with us, you gain access to a supply chain that is not only robust and compliant but also driven by a commitment to continuous technological improvement and customer success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
