Advanced Synthetic Route for High-Purity Cefepime Hydrochloride Manufacturing
The pharmaceutical industry continuously demands more efficient and robust synthetic routes for critical antibiotics like Cefepime, a fourth-generation cephalosporin known for its broad spectrum and stability against beta-lactamases. Patent CN101735251A introduces a transformative methodology that addresses longstanding challenges in the manufacturing of Cefepime Hydrochloride, specifically focusing on the optimization of the 3-position modification and the subsequent acylation steps. This technical breakthrough leverages a novel quaternary ammonium salt intermediate strategy combined with phase transfer catalysis to drastically enhance reaction control. By shifting away from traditional direct alkylation methods which often suffer from uncontrolled reactivity, this process offers a streamlined pathway that is highly attractive for industrial scale-up. For R&D directors and procurement specialists alike, understanding this mechanism is key to securing a reliable cefepime intermediate supplier capable of delivering consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for modifying the 3-position of the cephalosporin nucleus often rely on the direct reaction of 7-ACA derivatives with N-methylpyrrolidine. However, the nitrogen atom in N-methylpyrrolidine possesses a high electron cloud density, rendering it excessively reactive and difficult to control during electrophilic substitution. This inherent instability frequently leads to a plethora of side reactions, resulting in complex impurity profiles that are costly and time-consuming to remove. Furthermore, conventional acylation steps often struggle with poor mass transfer between immiscible phases, leading to incomplete conversions and lower overall yields. These inefficiencies create significant bottlenecks in cost reduction in antibiotic manufacturing, as extensive purification protocols are required to meet stringent pharmacopeial standards.
The Novel Approach
The innovative approach detailed in the patent circumvents these issues by pre-forming a stable quaternary ammonium salt intermediate from N-methylpyrrolidine and iodotrimethylsilane before introducing it to the protected 7-ACA solution. This strategic modification effectively tames the reactivity of the nitrogen species, allowing for a clean and selective electrophilic substitution that minimizes byproduct formation. Additionally, the introduction of a phase transfer catalyst in the subsequent N-acylation step ensures efficient interaction between the 7-MPCA intermediate and the AE-active ester within the organic phase. This dual-strategy not only boosts the yield of the critical 7-MPCA intermediate by approximately 7% but also enhances the final acylation conversion by roughly 5%, demonstrating a clear path toward high-purity cefepime hydrochloride production.
Mechanistic Insights into Quaternary Ammonium Mediated Substitution
The core of this synthetic advancement lies in the precise manipulation of electronic properties during the 3-position substitution. Initially, 7-aminocephalosporanic acid (7-ACA) undergoes dual protection of its carboxylic and amino groups using hexamethyldisilazane (HMDS) and iodotrimethylsilane (TMSI), creating a robust silylated scaffold. Simultaneously, N-methylpyrrolidine is converted into a quaternary ammonium salt, which acts as a controlled electrophile rather than a volatile nucleophile. When these two components meet in the presence of N,N-diethylaniline, the reaction proceeds via a highly ordered electrophilic substitution mechanism. This controlled environment prevents the chaotic polymerization or degradation often seen in direct alkylation, ensuring that the cephalosporin ring system remains intact while the desired side chain is installed with high fidelity.
Following the formation of the 7-MPCA intermediate, the process moves to the acylation stage where mechanistic efficiency is paramount. The addition of phase transfer catalysts, such as tetramethyl ammonium chloride or tributylamine, creates a molecular bridge between the aqueous and organic layers. This facilitates the transport of anionic species into the organic phase where the AE-active ester resides, significantly accelerating the nucleophilic attack on the activated ester carbonyl. The result is a rapid and near-complete conversion that minimizes the residence time of sensitive intermediates, thereby reducing the risk of hydrolysis or epimerization. This level of mechanistic control is essential for the commercial scale-up of complex cephalosporins, ensuring batch-to-batch consistency.
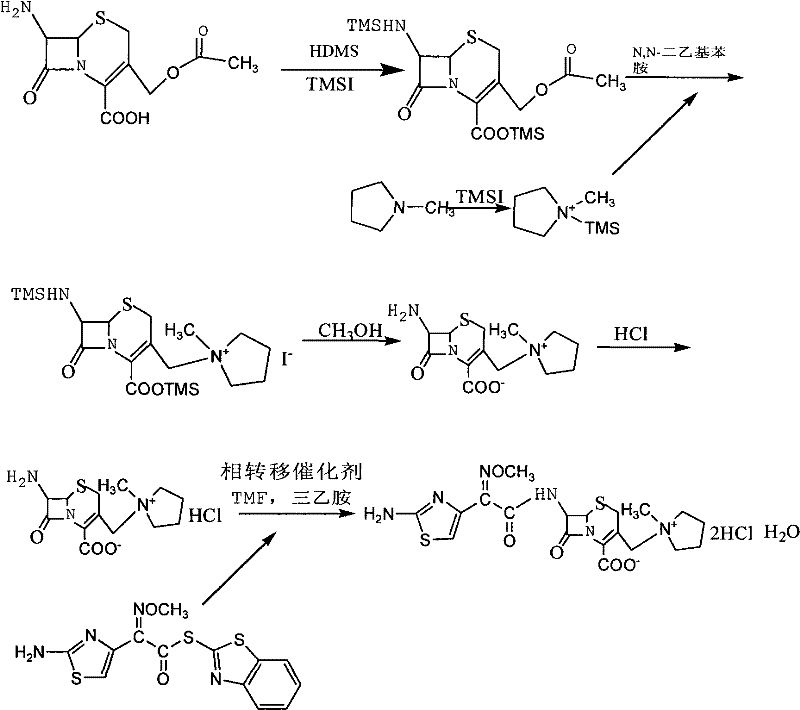
How to Synthesize 7-MPCA and Cefepime Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the patented method. The process begins with the silylation of 7-ACA followed by the separate preparation of the quaternary salt, which are then combined under inert conditions to form the 7-MPCA intermediate. Subsequent deprotection and acylation steps utilize common organic solvents like methylene chloride and tetrahydrofuran, making the process adaptable to existing infrastructure. For detailed operational parameters, stoichiometric ratios, and specific workup procedures, please refer to the standardized guide below which outlines the critical control points for successful implementation.
- Protect 7-ACA with HMDS and TMSI, then react with a pre-formed N-methylpyrrolidine quaternary ammonium salt intermediate to synthesize 7-MPCA.
- Perform N-acylation of 7-MPCA with AE-active ester in an organic phase using a phase transfer catalyst to enhance conversion rates.
- Complete the synthesis by salifying with hydrochloric acid and crystallizing the final Cefepime Hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patented methodology offers substantial advantages in terms of operational efficiency and resource utilization. By fundamentally altering the reaction kinetics to favor the desired product, the process inherently reduces the generation of hazardous waste and the consumption of raw materials associated with reprocessing off-spec batches. The use of readily available starting materials such as 7-ACA and standard silating agents ensures that the supply chain remains resilient against market fluctuations. Furthermore, the simplified purification profile means that manufacturing cycles can be shortened, directly contributing to reducing lead time for high-purity beta-lactams and enhancing overall throughput without the need for capital-intensive new equipment.
- Cost Reduction in Manufacturing: The elimination of excessive side reactions translates directly into lower production costs by maximizing the yield of valuable intermediates. Since the process achieves high purity in a single crystallization step, the need for expensive chromatographic purification or multiple recrystallizations is significantly diminished. This streamlining of the downstream processing workflow results in substantial savings on solvent usage, energy consumption, and labor hours, providing a competitive edge in pricing strategies for generic antibiotic production.
- Enhanced Supply Chain Reliability: The robustness of the quaternary ammonium salt strategy ensures that the synthesis is less susceptible to minor variations in reaction conditions, leading to more predictable output volumes. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers who depend on consistent quality. By mitigating the risk of batch failures due to uncontrolled reactivity, suppliers can offer more secure delivery schedules and maintain higher inventory turnover rates, strengthening the partnership between chemical manufacturers and pharmaceutical clients.
- Scalability and Environmental Compliance: The process utilizes standard organic solvents and avoids the use of heavy metal catalysts, aligning well with modern green chemistry principles and environmental regulations. The simplicity of the workup, involving straightforward extraction and crystallization, makes the technology highly scalable from pilot plant to multi-ton commercial production. This ease of scale-up reduces the technical risk associated with technology transfer, allowing for rapid deployment of capacity to meet surging global demand for fourth-generation cephalosporins.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthetic route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is vital for evaluating the feasibility of integrating this technology into your existing production portfolio.
Q: How does the quaternary ammonium salt intermediate improve the synthesis of Cefepime?
A: By pre-forming the quaternary ammonium salt from N-methylpyrrolidine and iodotrimethylsilane, the process overcomes the high electron cloud density and excessive reactivity of the nitrogen atom. This controlled reactivity significantly reduces side reactions during the electrophilic substitution at the 3-position of the cephalosporin nucleus, leading to a marked improvement in both yield and product purity compared to direct alkylation methods.
Q: What is the role of the phase transfer catalyst in the acylation step?
A: In the second step involving the reaction between 7-MPCA and the AE-active ester, the phase transfer catalyst facilitates the migration of reactive species between the aqueous and organic phases. This enhancement in mass transfer efficiency increases the reaction conversion rate by approximately 5%, ensuring a more complete reaction and simplifying the downstream purification process.
Q: What are the purity specifications achievable with this patented method?
A: The optimized synthetic route described in patent CN101735251A allows for the production of Cefepime Hydrochloride with exceptional purity levels, consistently reaching above 99% in a single crystallization step. This high purity is attributed to the suppression of side products in the first step and the efficient conversion in the second step, minimizing the burden on final purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101735251A are fully realized in practical manufacturing settings. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Cefepime intermediate meets the highest international standards, providing our clients with the confidence needed for regulatory filings.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive value and efficiency for your organization.
