Scalable Production of High-Purity Cefepime Hydrochloride via Optimized Silylation and Crystallization
Scalable Production of High-Purity Cefepime Hydrochloride via Optimized Silylation and Crystallization
The pharmaceutical landscape for fourth-generation cephalosporins demands rigorous adherence to purity standards and cost-efficient manufacturing protocols to meet global healthcare needs. Patent CN102276631A introduces a transformative preparation method for Cefepime Hydrochloride, addressing critical bottlenecks found in both traditional laboratory techniques and existing industrial production technologies. This innovation focuses on the strategic optimization of the 7-amino-cephalosporanic acid (7-ACA) protection phase and the subsequent crystallization of key intermediates, delivering a pathway that significantly enhances operational safety and product yield. By implementing wet feeding protocols for 7-ACA and utilizing N,O-bis(trimethylsilyl)acetamide (BSA) as a superior protective agent, the process mitigates the risks associated with dry powder handling while accelerating reaction kinetics. For R&D directors and procurement specialists seeking a reliable antibiotic intermediate supplier, this methodology represents a substantial leap forward in process chemistry, offering a robust framework for the commercial scale-up of complex beta-lactam antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefepime Hydrochloride has been plagued by inefficiencies that hinder cost reduction in antibiotic manufacturing and compromise supply chain reliability. Traditional routes often rely on expensive raw materials and cumbersome protection-deprotection sequences that result in suboptimal yields, with some prior art reporting total recovery rates as low as 24.7 percent. These legacy methods frequently necessitate the use of harsh conditions or transition metal catalysts that introduce difficult-to-remove impurities, thereby complicating the purification process and increasing the burden on quality control laboratories. Furthermore, conventional crystallization techniques often fail to produce uniform crystal morphologies, leading to poor filtration characteristics and inconsistent solvent retention in the final cake. Such variability not only extends production cycles but also elevates the risk of batch rejection, creating significant volatility for supply chain heads managing inventory levels for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN102276631A fundamentally reengineers the synthetic route by introducing a wet feeding mechanism for 7-ACA and a streamlined silylation protocol using BSA. This novel approach eliminates the hazards of dust generation during raw material charging, drastically improving the industrial hygiene environment and protecting operator health. By replacing traditional protective groups with BSA, the reaction time for the protection step is reduced to merely 2 hours at room temperature, which dramatically increases throughput capacity without sacrificing conversion efficiency. Additionally, the process incorporates a sophisticated crystallization strategy for the 7-MPCA intermediate involving stepwise solvent addition and room temperature grain growing, which ensures the formation of high-quality crystals that are easy to wash and filter. This holistic optimization reduces the dependency on activated carbon for decolorization, directly translating to lower material costs and simplified waste treatment procedures for the facility.
Mechanistic Insights into BSA-Mediated Silylation and Crystallization Control
The core chemical innovation lies in the efficient silylation of the 7-ACA substrate using N,O-bis(trimethylsilyl)acetamide (BSA), which acts as a potent silylating agent to protect both the amino and carboxyl functionalities simultaneously. In the presence of dichloromethane as the solvent, BSA reacts rapidly with 7-ACA to form a bis-silylated intermediate that is highly nucleophilic and stable under the reaction conditions. This activation allows for the subsequent quaternization with N-methylpyrrolidine and iodotrimethylsilane to proceed with high regioselectivity, minimizing the formation of unwanted isomers or degradation products. The use of N,N-diethylaniline as a base further facilitates the reaction by scavenging generated acids, maintaining a neutral environment that preserves the integrity of the sensitive beta-lactam ring. This mechanistic precision ensures that the resulting 7-MPCA intermediate possesses a high degree of chemical purity, which is critical for the success of the final coupling step with the AE active ester.
Following the synthesis of the intermediate, the patent describes a controlled crystallization mechanism that is vital for impurity management and physical property optimization. By adding acetone gradually to the reaction mixture at room temperature and allowing for a dedicated grain-growing period of 30 minutes, the process promotes the formation of larger, more uniform crystals rather than amorphous precipitates. This controlled nucleation and growth phase allows impurities to remain in the mother liquor rather than being occluded within the crystal lattice, significantly enhancing the purity profile of the isolated solid. The improved crystal morphology also facilitates efficient washing with fresh acetone, removing residual solvents and by-products effectively. Consequently, the final Cefepime Hydrochloride product requires less aggressive decolorization treatments, preserving the yield and ensuring that the final API meets stringent pharmacopeial specifications for heavy metals and organic volatiles.
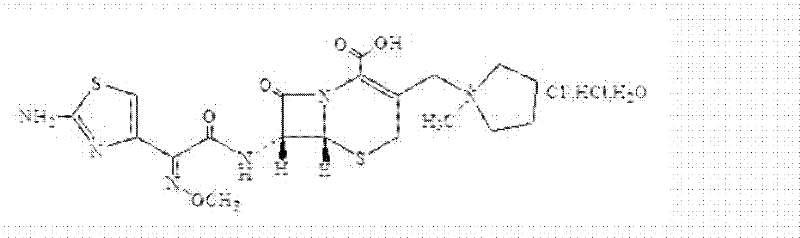
How to Synthesize Cefepime Hydrochloride Efficiently
The synthesis of Cefepime Hydrochloride via this optimized route involves a sequence of precise unit operations designed to maximize yield and safety while minimizing environmental impact. The process begins with the dissolution of 7-ACA in dichloromethane using a wet feeding technique, followed by protection with BSA and subsequent reaction with iodotrimethylsilane and N-methylpyrrolidine to generate the quaternary ammonium salt. After acidification and extraction, the critical 7-MPCA intermediate is isolated through a controlled crystallization process involving stepwise acetone addition, which is then coupled with AE active ester in a mixed solvent system of dichloromethane and DMF. The detailed standardized synthesis steps, including specific reagent ratios, temperature controls, and stirring times required to replicate this high-efficiency pathway, are outlined in the technical guide below.
- Dissolve 7-ACA in dichloromethane using wet feeding, protect with BSA, and react with N,N-Diethylaniline and Iodotrimethylsilane at controlled temperatures.
- Add N-Methylpyrrolidine dropwise at 0-5°C to form the quaternary ammonium intermediate, followed by methanol addition and acidification.
- Crystallize the 7-MPCA intermediate using stepwise acetone addition and room temperature grain growing, then couple with AE active ester and purify.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling economic and operational benefits that directly impact the bottom line. The elimination of expensive and hazardous reagents, combined with the reduction in reaction times, leads to a substantial decrease in overall manufacturing costs, making the final product more competitive in the global marketplace. The robustness of the crystallization process ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results and enhancing the reliability of supply for downstream formulation partners. Furthermore, the simplified purification steps reduce the consumption of auxiliary materials like activated carbon and solvents, aligning with modern sustainability goals and reducing the environmental footprint of the manufacturing facility.
- Cost Reduction in Manufacturing: The implementation of BSA as a protective agent significantly shortens the reaction cycle time compared to traditional methods, allowing for higher equipment utilization rates and increased annual production capacity without additional capital investment. By reducing the amount of activated carbon required for decolorization due to the higher purity of the intermediate, the process lowers the consumption of consumables and minimizes waste disposal costs associated with spent carbon filtration. The wet feeding of 7-ACA also reduces material loss due to dusting, ensuring that every gram of expensive starting material is accounted for in the final product yield, thereby optimizing the cost of goods sold.
- Enhanced Supply Chain Reliability: The improved crystal quality of the 7-MPCA intermediate ensures excellent filtration and drying characteristics, which drastically reduces the turnaround time between batches and prevents bottlenecks in the production schedule. This operational efficiency translates into shorter lead times for high-purity antibiotic intermediates, enabling suppliers to respond more agilely to fluctuations in market demand. The robustness of the process against minor variations in reaction conditions ensures a steady and predictable output, providing procurement teams with the confidence to plan long-term contracts and secure stable pricing for their API requirements.
- Scalability and Environmental Compliance: The entire technology is designed with industrial scalability in mind, utilizing common solvents and straightforward unit operations that can be easily transferred from pilot scale to multi-ton commercial production. The reduction in solvent usage and the minimization of hazardous waste streams through efficient recycling and reduced decolorization needs support compliance with increasingly stringent environmental regulations. This green chemistry approach not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for environmentally conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cefepime Hydrochloride using the optimized method described in patent CN102276631A. These insights are derived directly from the patent's background technology and beneficial effects sections, providing clarity on how this specific route overcomes historical challenges in beta-lactam synthesis. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for their own supply chains or for procurement officers assessing the quality credentials of potential vendors.
Q: What are the key advantages of using BSA in Cefepime synthesis?
A: Using N,O-bis(trimethylsilyl)acetamide (BSA) as a protective agent significantly reduces reaction time to approximately 2 hours compared to conventional methods, improving transformation efficiency and reducing overall processing costs while maintaining high product integrity.
Q: How does the new crystallization process improve product quality?
A: The novel process incorporates stepwise addition of recrystallization solvent and a room temperature grain growing phase, which results in superior crystal formation, enhanced washing efficiency, and obviously improved intermediate quality, thereby reducing the need for extensive decolorization.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial suitability by employing wet feeding of 7-ACA to improve hygiene and safety, simplifying operational steps, and utilizing robust crystallization techniques that ensure consistent batch-to-batch reproducibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced process technologies like the one detailed in CN102276631A to deliver exceptional value to our global partners in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of optimized silylation and crystallization are fully realized in large-scale manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Cefepime Hydrochloride meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to explore how this innovative synthesis route can drive efficiency and cost savings in your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain planning initiatives, ensuring a seamless partnership from development to commercial supply.
