Optimizing Pemetrexed Disodium Production: A Novel Protected Intermediate Strategy for Commercial Scale
Optimizing Pemetrexed Disodium Production: A Novel Protected Intermediate Strategy for Commercial Scale
The global demand for oncology therapeutics continues to drive rigorous innovation in the synthesis of critical active pharmaceutical ingredients (APIs), particularly for multi-target antifolates like Pemetrexed disodium. As a cornerstone treatment for malignant pleural mesothelioma and non-small cell lung cancer, the supply chain integrity and purity profile of Pemetrexed are paramount. Recent advancements detailed in patent CN110305134B introduce a transformative approach to synthesizing the key intermediate, N-{4-[2-(2-acetylamino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoyl}-L-glutamic acid diethyl ester tosylate (Intermediate III-1). This technical insight report analyzes the strategic shift from conventional direct condensation to a protected amino-group strategy, offering significant advantages for R&D directors seeking purity, procurement managers targeting cost efficiency, and supply chain leaders requiring robust scalability.
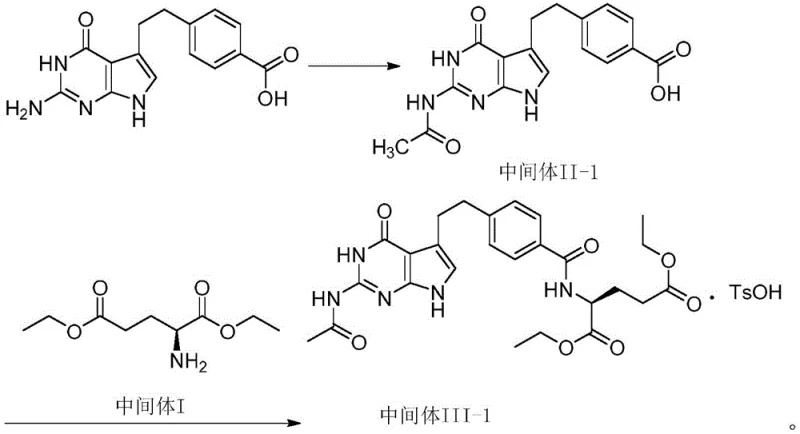
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
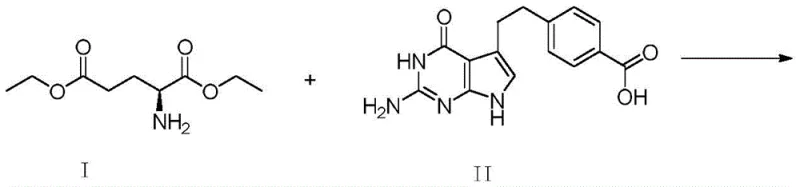
Historically, the industrial synthesis of Pemetrexed disodium has relied on the direct condensation of 4-[2-(2-amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoic acid with L-glutamic acid diethyl ester. While conceptually straightforward, this conventional pathway suffers from severe chemoselectivity issues due to the presence of multiple reactive sites on the starting material. Specifically, the primary amino group on the pyrrolopyrimidine ring is highly nucleophilic and prone to uncontrolled side reactions during the coupling process. This lack of selectivity leads to the formation of a persistent byproduct known as Impurity V, which can accumulate to levels between 5% and 10% in the crude reaction mixture. To meet stringent pharmacopeial standards, manufacturers are forced to subject the intermediate to two or three rounds of intensive recrystallization. This repetitive purification not only drastically reduces the overall process yield but also escalates solvent consumption and processing time, creating a significant bottleneck in cost reduction in API manufacturing.
The Novel Approach
The innovative methodology disclosed in the patent addresses these chemoselectivity challenges through a proactive protection-deprotection strategy. By converting the starting benzoic acid derivative into its N-acetyl protected form (Intermediate II-1) prior to the coupling step, the reactive amino group is effectively masked. This structural modification prevents the amino group from participating in the condensation reaction, thereby directing the coupling exclusively to the carboxylic acid moiety. Consequently, the formation of Impurity V is virtually eliminated at the source. This allows the resulting Intermediate III-1 to proceed to the next stage without the need for extensive recrystallization, streamlining the workflow. The result is a process that not only enhances product stability against oxidative discoloration but also delivers a substantial increase in throughput, making it a superior choice for commercial scale-up of complex oncology intermediates.
Mechanistic Insights into Acylation Protection and CDI Coupling
The core of this technological breakthrough lies in the precise manipulation of functional group reactivity. The initial step involves the acylation of the primary amine on the pyrrolopyrimidine scaffold using acetic anhydride in an acetic acid medium. Mechanistically, the lone pair electrons on the nitrogen atom of the amino group act as a nucleophile, attacking the electrophilic carbonyl carbon of the acetic anhydride. This reaction is conducted under mild thermal conditions, typically between 40°C and 50°C, which ensures complete conversion to the acetamide while preserving the integrity of the sensitive heterocyclic ring system. The resulting acetamide group is significantly less nucleophilic than the original amine due to the electron-withdrawing nature of the adjacent carbonyl, effectively deactivating the nitrogen towards further unwanted alkylation or acylation during subsequent steps.
Following protection, the condensation reaction utilizes N,N-carbonyldiimidazole (CDI) as a coupling agent to activate the carboxylic acid of Intermediate II-1. CDI reacts with the carboxyl group to form a reactive acyl-imidazole intermediate, releasing imidazole as a leaving group. This activated species is then attacked by the amino group of L-diethyl glutamate (Intermediate I). The use of CDI is particularly advantageous in this context as it avoids the introduction of heavy metal catalysts or toxic coupling reagents that could complicate downstream purification. Finally, the addition of p-toluenesulfonic acid facilitates the formation of the stable tosylate salt (Intermediate III-1). This salt formation not only aids in the isolation of the product via crystallization but also locks the molecule into a specific conformation that minimizes degradation, ensuring high-purity pharmaceutical intermediates are maintained throughout storage and transport.
How to Synthesize Pemetrexed Disodium Intermediate Efficiently
The implementation of this synthesis route requires careful control of stoichiometry and temperature to maximize the benefits of the protection strategy. The process begins with the acylation step, where precise molar ratios of acetic anhydride are critical to ensure full protection without excessive reagent waste. Subsequently, the coupling reaction must be monitored to ensure complete activation by CDI before the addition of the glutamate derivative. The final salt formation step serves as both a purification and stabilization mechanism. For detailed operational parameters, stoichiometric ratios, and workup procedures, please refer to the standardized synthesis guide below.
- Perform acylation of 4-[2-(2-amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoic acid using acetic anhydride to form the protected Intermediate II-1.
- Condense Intermediate II-1 with L-diethyl glutamate using N,N-carbonyldiimidazole (CDI) followed by reaction with p-toluenesulfonic acid to obtain Intermediate III-1.
- Hydrolyze Intermediate III-1 in sodium hydroxide solution, adjust pH, and perform salt formation to yield final Pemetrexed disodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this protected intermediate route offers compelling economic and logistical benefits. The primary driver of value is the drastic simplification of the purification train. By eliminating the need for multiple recrystallizations to remove Impurity V, manufacturers can significantly reduce solvent usage, energy consumption for heating and cooling, and labor hours associated with filtration and drying. This leaner process directly translates to lower variable costs per kilogram of produced API. Furthermore, the higher yield reported in the patent data—improving by nearly 40% compared to the unprotected comparative examples—means that less raw material is required to produce the same amount of final product, optimizing the cost of goods sold (COGS) and enhancing margin potential in a competitive generic drug market.
- Cost Reduction in Manufacturing: The elimination of repetitive recrystallization steps removes a major cost center from the production budget. Solvents represent a significant portion of chemical manufacturing expenses, and reducing the number of dissolution-crystallization cycles leads to substantial savings in both material procurement and waste disposal fees. Additionally, the higher reaction yield ensures better atom economy, meaning more of the expensive starting materials are converted into saleable product rather than being lost as mother liquor or degraded impurities.
- Enhanced Supply Chain Reliability: A process that is less sensitive to impurity buildup is inherently more robust. In traditional methods, slight variations in reaction conditions can lead to spikes in Impurity V, causing batch failures or extended processing times to salvage the material. The protected route mitigates this risk, leading to more consistent batch-to-batch quality and predictable production schedules. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream formulation teams receive their materials on schedule without unexpected delays caused by out-of-specification results.
- Scalability and Environmental Compliance: The mild reaction conditions (40-60°C) and the use of common organic solvents like DMF, dichloromethane, and ethanol make this process highly scalable from pilot plant to multi-ton commercial production. The reduced solvent load also aligns with modern green chemistry principles, lowering the environmental footprint of the manufacturing site. Easier waste management and reduced hazardous waste generation simplify regulatory compliance, allowing for smoother audits and sustained operational licenses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Pemetrexed disodium using this advanced intermediate strategy. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the technology's capabilities.
Q: How does the new synthesis route reduce Impurity V in Pemetrexed production?
A: The novel route introduces an acetyl protection group on the amino moiety of the pyrrolopyrimidine core before condensation. This prevents the free amine from participating in side reactions that typically generate Impurity V, effectively eliminating this critical quality issue.
Q: What are the yield improvements associated with this protected intermediate method?
A: By avoiding the formation of difficult-to-remove impurities, the process eliminates the need for multiple recrystallization steps. Patent data indicates this optimization improves the overall yield of the intermediate by nearly 40% compared to conventional unprotected routes.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions are mild (40-60°C) and utilize standard reagents like acetic anhydride and CDI. The simplified purification process and higher stability of the protected intermediate make it highly scalable for industrial API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand that the complexity of oncology drug synthesis demands a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We employ stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Intermediate III-1 meets the highest global standards. Our facility is designed to handle the specific solvent systems and reaction conditions required for this protected synthesis route, ensuring safety and efficiency at every stage.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and high-yield synthesis technology. By optimizing your supply chain with our advanced intermediates, you can accelerate your time-to-market and improve your product's profitability. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our Pemetrexed disodium intermediate can become a cornerstone of your successful API manufacturing strategy.
