Advanced Purification Technology for High-Purity Pemetrexed Disodium Commercial Manufacturing
The pharmaceutical industry continuously demands higher standards for bulk drug purity, particularly for potent antifolate agents used in oncology treatments. Patent CN102206218A introduces a groundbreaking purification method for pemetrexed disodium that addresses critical limitations in existing manufacturing processes. This technology leverages a sophisticated combination of salting-out techniques and organic solvent crystallization performed strictly at normal temperature to achieve exceptional purity profiles. By effectively controlling both organic and inorganic impurities, this process ensures that the final active pharmaceutical ingredient meets the rigorous identification thresholds required by global regulatory bodies. The strategic implementation of this method allows manufacturers to produce high-purity pemetrexed disodium that rivals imported preparations in quality while optimizing production efficiency. For R&D teams and procurement specialists, understanding this technological leap is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent, high-quality raw materials for life-saving medications.
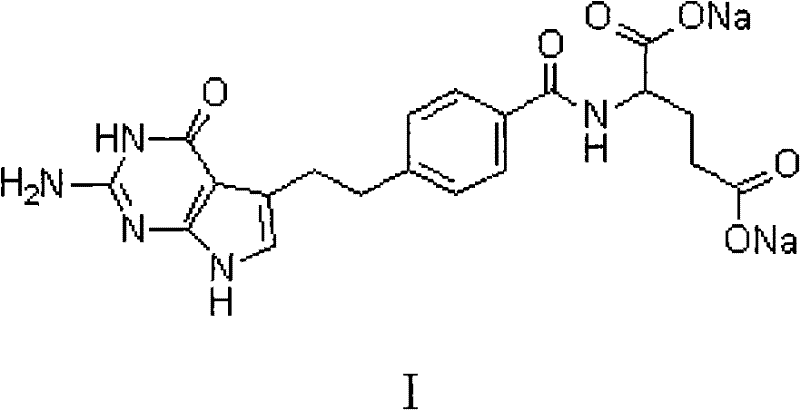
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification strategies for pemetrexed disodium have historically struggled to balance yield with impurity removal, often resulting in products that fail to meet strict regulatory standards. Conventional mixed solvent crystallization methods typically require heating steps, which unfortunately aggravate the hydrolysis and oxidation of the product, leading to an increase in degradation impurities that compromise drug safety. Furthermore, standard salting-out processes conducted solely in aqueous phases are ineffective at removing organic impurities with lower polarity than the target molecule, leaving behind significant contaminants that exceed the 0.1% identification limit. These deficiencies necessitate additional processing steps that drive up costs and extend lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates. The inability of prior art to simultaneously control organic impurity quantity and inorganic salt content has been a persistent challenge, forcing manufacturers to accept lower yields or suboptimal purity levels that hinder commercial viability.
The Novel Approach
The innovative process described in the patent data overcomes these historical hurdles by integrating a dual-stage crystallization strategy that operates entirely under normal temperature conditions ranging from 5°C to 39°C. This method first utilizes a controlled salting-out step with sodium chloride to precipitate the product while leaving highly water-soluble impurities in the solution, followed by a precise mixed solvent crystallization using ethanol or similar organic solvents. By avoiding thermal stress, the novel approach prevents the formation of heat-induced degradation products, ensuring the chemical stability of the pemetrexed disodium structure throughout the purification cycle. The sequential application of these techniques allows for the drastic reduction of organic impurity numbers from fifteen down to four, achieving a level of cleanliness that aligns with the quality of originator drugs. This breakthrough represents a significant advancement in cost reduction in API manufacturing, as it simplifies the workflow while enhancing the overall quality and safety profile of the final bulk drug substance.
Mechanistic Insights into Salting-Out and Mixed Solvent Crystallization
The core mechanism driving the success of this purification technology lies in the differential solubility properties of pemetrexed disodium compared to its associated impurities under specific ionic strength and solvent polarity conditions. In the initial salting-out phase, the addition of sodium chloride to a concentration of 8% to 20% creates an environment where the target molecule precipitates efficiently while retaining polar organic impurities in the aqueous mother liquor. This step is critical for removing inorganic contaminants and setting the stage for the subsequent organic solvent treatment, which targets non-polar or less polar organic byproducts that remain after the first crystallization. The careful regulation of pH between 7.5 and 8.5 ensures that the molecule remains in its optimal ionic state for crystallization, preventing the formation of unwanted salts or degradation products that could arise from acidic or basic conditions. This precise control over the physicochemical environment is what enables the process to achieve such high levels of purity without sacrificing yield, making it a robust solution for commercial scale-up of complex pharmaceutical intermediates.
Impurity control is further enhanced by the second crystallization step, where the introduction of a water-miscible organic solvent like ethanol reduces the dielectric constant of the medium, forcing the pemetrexed disodium to crystallize out while leaving residual organic impurities dissolved. The patent data indicates that maintaining the organic solvent concentration between 70% and 90% is vital; concentrations below this range result in insufficient precipitation and yield loss, while higher concentrations risk co-precipitating impurities. By operating at normal temperature, the process avoids the kinetic energy that typically drives side reactions such as hydrolysis, which is a common failure point in heated crystallization methods. This mechanistic understanding allows process chemists to fine-tune parameters for maximum efficiency, ensuring that the maximum single organic impurity content is consistently controlled below the 0.1% threshold required for medicine registration. Such rigorous control is essential for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for reprocessing or rejection of batches due to out-of-specification impurity profiles.
How to Synthesize Pemetrexed Disodium Efficiently
Implementing this purification protocol requires strict adherence to the specified operational parameters to ensure reproducibility and compliance with quality standards. The process begins with dissolving the crude pemetrexed disodium in water for injection, followed by pH adjustment and the controlled addition of sodium chloride solution to initiate the first crystallization phase. After filtering and washing the initial crystals, the material is redissolved and subjected to the organic solvent crystallization step, where the concentration of ethanol or alternative solvents is carefully managed to optimize purity and yield. Detailed standardized synthesis steps see the guide below, which outlines the precise measurements and timing required to replicate the high-success rates observed in the patent examples. Following these guidelines ensures that manufacturers can consistently produce bulk drug substances that meet the stringent requirements of global regulatory agencies while maintaining cost-effective production workflows.
- Dissolve crude pemetrexed disodium in water, adjust pH to 7.5-8.5, and add sodium chloride solution to reach 8-20% concentration for initial crystallization.
- Filter the initial crystals and wash with sodium chloride solution to remove water-soluble impurities effectively.
- Redissolve the filter cake in water, add a water-miscible organic solvent like ethanol to reach 70-90% concentration, and crystallize at normal temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this purification technology offers substantial strategic benefits that extend beyond mere technical compliance. By eliminating the need for heating steps, the process significantly reduces energy consumption and minimizes the risk of batch failures due to thermal degradation, leading to more predictable production schedules and enhanced supply chain reliability. The ability to effectively purify crude materials with varying initial purity levels means that manufacturers can source raw materials more flexibly without compromising on the quality of the final product, thereby mitigating supply risks associated with raw material variability. Furthermore, the simplification of the purification workflow reduces the overall processing time and equipment usage, contributing to substantial cost savings in the manufacturing of this critical oncology ingredient. These efficiencies translate into a more competitive pricing structure for buyers seeking a reliable agrochemical intermediate supplier or pharmaceutical partner, ensuring long-term availability of this essential medication.
- Cost Reduction in Manufacturing: The elimination of heating steps and the optimization of solvent usage drastically simplify the production process, removing the need for expensive energy inputs and complex temperature control systems. By avoiding the introduction of excessive inorganic salts that require additional removal steps, the process streamlines the workflow and reduces waste disposal costs associated with hazardous byproducts. This qualitative improvement in process efficiency allows for a more lean manufacturing model, where resources are allocated more effectively to maximize output without inflating operational expenditures. Consequently, partners can expect a more favorable cost structure that reflects the inherent efficiencies of this advanced purification methodology.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in crude material quality ensures a steady flow of compliant product, reducing the likelihood of production delays caused by out-of-specification raw materials. Since the process operates at normal temperature, it is less susceptible to equipment failures related to heating elements or cooling systems, further stabilizing the production timeline. This reliability is crucial for maintaining continuous supply lines for critical medications, ensuring that patients have uninterrupted access to their treatments without the risk of shortages. Supply chain heads can therefore plan with greater confidence, knowing that the manufacturing process is designed to withstand common variabilities without compromising delivery commitments.
- Scalability and Environmental Compliance: The use of common solvents like ethanol and sodium chloride facilitates easy scale-up from laboratory to industrial production without requiring specialized or hazardous reagents. The reduction in organic impurity load also simplifies waste treatment processes, aligning with increasingly stringent environmental regulations and reducing the ecological footprint of the manufacturing facility. This scalability ensures that production volumes can be increased to meet market demand without encountering the technical bottlenecks often associated with complex purification chemistries. Additionally, the minimized generation of degradation products contributes to a cleaner production environment, supporting corporate sustainability goals and regulatory compliance efforts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of pemetrexed disodium, providing clarity on how this technology meets industry standards. These insights are derived directly from the patent data and are intended to assist decision-makers in evaluating the feasibility and benefits of adopting this method for their supply chains. Understanding these details is key to establishing a productive partnership with a manufacturer capable of delivering high-quality intermediates.
Q: How does this purification method control organic impurities below 0.1%?
A: By combining salting-out with mixed solvent crystallization at normal temperature, the process effectively removes both polar and non-polar organic impurities without inducing thermal degradation, ensuring maximum single impurity content remains below the 0.1% identification threshold.
Q: Why is normal temperature critical for pemetrexed disodium stability?
A: Heating steps in conventional methods often aggravate hydrolysis and oxidation of the product. Operating within the 5-39°C range prevents the formation of degradation impurities, maintaining the structural integrity and stability of the bulk drug.
Q: Can this process handle crude materials with varying purity levels?
A: Yes, the method is robust enough to purify pemetrexed disodium crude products with content ranging from 80% to 99.5%, consistently reducing total impurities and meeting stringent medicine registration requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of oncology therapeutics like pemetrexed disodium. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production positions us as an ideal partner for bringing this advanced purification technology to market. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the highest international standards for safety and efficacy. Our commitment to quality assurance means that clients can rely on us for a steady supply of high-purity pharmaceutical intermediates that support their drug development and commercialization goals without compromise.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can benefit your bottom line. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to verify our technical competence and commitment to excellence firsthand. Let us collaborate to ensure the successful and sustainable production of this vital medication.
