Advanced Manufacturing of Pemetrexed Disodium via Novel Bromo-Intermediate Route
Introduction to Patent CN103086912A
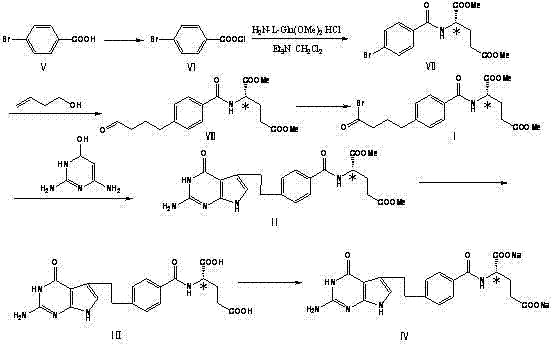
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex antineoplastic agents, and patent CN103086912A presents a significant breakthrough in the manufacturing of Pemetrexed Disodium. This document details a robust chemical synthesis method for N-[4-[2-(2-amino-4,7-dihydro-4-oxo-1H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoyl]-L-glutamic acid disodium salt, commonly known as Pemetrexed. The core innovation lies in the utilization of a novel intermediate, N-[3-bromo-4-(butyraldehyde-4-yl)-benzoyl]-L-glutamic acid dimethyl ester, which serves as a pivotal building block. For R&D Directors, this route offers a compelling alternative to legacy methods by promising higher yields and superior impurity profiles through milder reaction conditions. Furthermore, the strategic design of this synthesis pathway directly addresses critical supply chain concerns by eliminating the need for exotic reagents and hazardous high-pressure operations, thereby facilitating a smoother transition from laboratory bench to commercial scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pemetrexed has been plagued by significant technical and economic hurdles that hinder efficient commercialization. As illustrated in prior art such as Route 1 (EP 0589720), traditional methodologies often depend heavily on expensive and moisture-sensitive reagents like hexamethyldisilazane and iodotrimethylsilane.
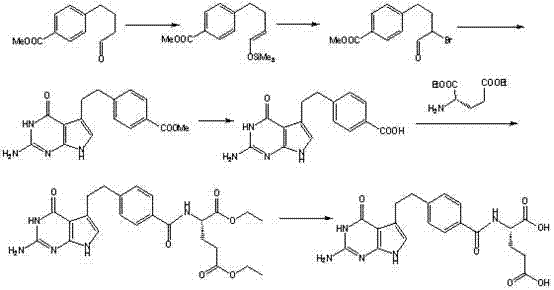
These reagents not only drive up the raw material costs but also introduce complexity in handling and storage, requiring strictly anhydrous conditions that are difficult to maintain in large reactors. Additionally, other reported routes, such as Route 3 (EP 0432677), involve intermediates containing acetylene bonds that necessitate high-pressure hydrogenation for reduction. This requirement introduces substantial safety risks and capital expenditure (CAPEX) barriers, as it mandates the use of specialized high-pressure reactors and rigorous safety protocols, making these methods less attractive for cost-sensitive generic manufacturing environments.
The Novel Approach
In stark contrast, the novel approach detailed in the patent circumvents these bottlenecks by employing a strategy centered on accessible starting materials and mild transformation steps. The new route initiates with a palladium-catalyzed coupling reaction to install the carbon chain, followed by a controlled bromination to activate the aromatic ring for subsequent cyclization. This methodology completely avoids the use of costly silylating agents and eliminates the need for dangerous high-pressure hydrogenation equipment. By shifting the synthetic logic to a bromo-aldehyde intermediate strategy, the process achieves a remarkable balance between operational simplicity and chemical efficiency. This allows manufacturers to utilize standard glass-lined or stainless steel reactors without the need for specialized high-pressure infrastructure, significantly lowering the barrier to entry for production and enhancing the overall economic viability of the supply chain.
Mechanistic Insights into Palladium-Catalyzed Coupling and Cyclization
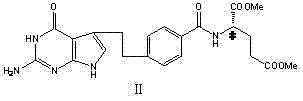
The heart of this synthetic advancement lies in the precise execution of the palladium-catalyzed coupling and the subsequent cyclization mechanism. The process begins with the reaction of 4-bromobenzoyl-L-glutamic acid dimethyl ester with 3-butene-1-ol under the influence of a palladium acetate catalyst system. This step, occurring in a polar solvent like DMF with lithium halides and phase-transfer catalysts, facilitates the formation of the critical aldehyde intermediate (VIII). The presence of weak alkaline reagents such as lithium acetate ensures that the reaction proceeds smoothly at moderate temperatures (50-70°C), preventing the degradation of the sensitive ester functionalities. This careful control of reaction parameters is essential for maintaining the integrity of the molecule before it undergoes the crucial activation step.
Following the formation of the aldehyde, the synthesis proceeds to the key cyclization event where the pyrrolo[2,3-d]pyrimidine core is constructed. As depicted in the condensation scheme, the bromo-intermediate (I) reacts with 2,4-diamino-6-hydroxypyrimidine in the presence of a condensing agent like sodium acetate.
This condensation reaction is conducted in a mixed solvent system of acetonitrile and water at a mild temperature range of 35-50°C. From a mechanistic standpoint, maintaining this low temperature window is vital for impurity control, specifically for preserving the chiral integrity of the L-glutamic acid moiety. Harsh conditions often lead to racemization, generating the unwanted D-isomer which is difficult to separate and detrimental to drug efficacy. By optimizing the pH and temperature during this cyclization, the process ensures high stereochemical purity, resulting in a final product that meets the rigorous quality standards required for oncology therapeutics without the need for complex chiral resolution steps downstream.
How to Synthesize Pemetrexed Disodium Efficiently
The synthesis of Pemetrexed Disodium via this novel route involves a sequence of highly optimized steps designed for reproducibility and scale-up. The process begins with the preparation of the key bromo-intermediate through controlled bromination, followed by the condensation with the pyrimidine base to form the protected precursor. Finally, hydrolysis and salt formation yield the active pharmaceutical ingredient. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification techniques such as recrystallization and tosic acid salification, are outlined below to guide technical implementation.
- Perform palladium-catalyzed coupling of 4-bromobenzoyl-L-glutamic acid dimethyl ester with 3-butene-1-ol to form the aldehyde intermediate.
- Execute controlled bromination of the aldehyde intermediate using 5,5-dibromo-2,2-dimethyl-4,6-dicarbapentaborane-1,3-diox to generate the key bromo-intermediate.
- Conduct condensation with 2,4-diamino-6-hydroxypyrimidine followed by hydrolysis and salt formation to yield Pemetrexed Disodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers transformative advantages that extend far beyond simple yield improvements. The primary value driver is the drastic simplification of the raw material portfolio. By replacing exotic, high-cost reagents like hexamethyldisilazane with commodity chemicals such as 3-butene-1-ol and standard brominating agents, the direct material costs are significantly reduced. Furthermore, the elimination of high-pressure hydrogenation steps removes a major bottleneck in production scheduling, as it negates the need for booking time on specialized, often scarce, high-pressure reactor units. This flexibility allows for more agile production planning and reduces the dependency on specific equipment configurations that can delay batch turnover.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from both material savings and operational efficiencies. The avoidance of expensive silylating agents and the use of common solvents like dichloromethane and acetonitrile streamline the solvent recovery processes, reducing waste disposal costs. Additionally, the mild reaction conditions (20-50°C) lower energy consumption compared to processes requiring extreme heating or cooling, contributing to a leaner overall cost structure that enhances competitiveness in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. The reliance on 4-bromobenzoic acid derivatives and standard amino acid estors ensures that sourcing risks are minimized, as these commodities are produced by multiple global suppliers. This diversification of the supply base protects against single-source disruptions and price volatility, ensuring a consistent flow of intermediates necessary for uninterrupted API production and reliable delivery to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing reaction conditions that are easily transferred from pilot plants to multi-ton commercial facilities. The absence of heavy metal catalysts in the final steps (or their easy removal) and the use of manageable solvents simplify wastewater treatment and environmental compliance. This 'green' aspect of the chemistry not only reduces regulatory burdens but also aligns with the increasing corporate sustainability goals of major pharmaceutical buyers, making the manufacturer a more attractive partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pemetrexed synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method resolves historical manufacturing challenges.
Q: How does this novel route improve upon conventional Pemetrexed synthesis methods?
A: Unlike conventional routes that rely on expensive silylating agents (like HMDS) or dangerous high-pressure hydrogenation steps, this method utilizes mild bromination and standard condensation reactions. This significantly lowers operational risks and raw material costs while maintaining high stereochemical purity.
Q: What are the critical quality attributes controlled in this synthesis?
A: The process is designed to preserve the L-configuration of the glutamic acid moiety. By avoiding harsh acidic or basic conditions during the cyclization step (maintaining 35-50°C), the method minimizes racemization, ensuring the final API meets stringent enantiomeric excess specifications required for oncology drugs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights suitability for mass industrialized production. The use of common solvents like dichloromethane and acetonitrile, along with the elimination of specialized high-pressure equipment, makes the technology highly scalable and easier to transfer to multi-ton manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to monitor critical quality attributes, such as enantiomeric excess and residual solvent levels, at every stage of the synthesis. This commitment to quality assurance guarantees that our Pemetrexed intermediates and APIs consistently meet the demanding requirements of global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and reliability in your oncology drug portfolio.
