Advanced Nitrile Manufacturing via Selective Phosphonic Anhydride Dehydration for Global Pharma Supply Chains
Advanced Nitrile Manufacturing via Selective Phosphonic Anhydride Dehydration for Global Pharma Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the relentless demand for higher purity intermediates and more sustainable synthetic routes. A pivotal advancement in this domain is detailed in patent CN101208296A, which discloses a highly selective method for preparing nitriles of formula (II) through an elimination reaction. Nitriles serve as indispensable building blocks in modern organic synthesis, acting as versatile precursors for a vast array of heterocyclic compounds essential in pharmaceutical and agrochemical applications. However, the historical challenge has always been balancing reactivity with selectivity. This patent introduces a transformative approach utilizing phosphonic anhydrides, specifically cyclic variants like propanephosphonic anhydride (T3P), to achieve dehydration of carboxamides or carboxylic acids under remarkably mild conditions. For R&D directors and procurement strategists alike, this technology represents a significant leap forward, offering a pathway to high-purity pharmaceutical intermediates while mitigating the risks associated with traditional, harsh dehydrating agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of nitriles has relied heavily on reagents such as phosphoryl chloride (POCl3), thionyl chloride (SOCl2), or carbodiimides like dicyclohexylcarbodiimide (DCC). While these reagents can effect the desired transformation, they are fraught with significant drawbacks that impact both yield and downstream processing costs. POCl3 and SOCl2 are aggressive dehydrating agents that often lack chemoselectivity, reacting indiscriminately with other functional groups present in complex molecular scaffolds. Furthermore, the use of DCC, while effective for amide coupling, presents severe challenges in nitrile synthesis due to the formation of dicyclohexylurea as a stoichiometric byproduct. This urea derivative is notoriously difficult to remove, often requiring labor-intensive chromatographic separation, which is economically unfeasible on a commercial scale. Additionally, the harsh thermal conditions often required by these traditional methods can lead to partial epimerization of chiral centers, compromising the stereochemical integrity of valuable drug candidates.
The Novel Approach
In stark contrast, the methodology outlined in CN101208296A utilizes alkylphosphonic anhydrides to solve these persistent problems. The core of this innovation lies in the use of 2,4,6-substituted 1,3,5,2,4,6-trioxatriphosphinane 2,4,6-trioxides, as depicted in the structural formula below. These reagents act as ideal, highly selective dehydrating agents that facilitate the conversion of N-alkylcarboxamides or carboxylic acid ammonium salts into the corresponding nitriles with exceptional efficiency.
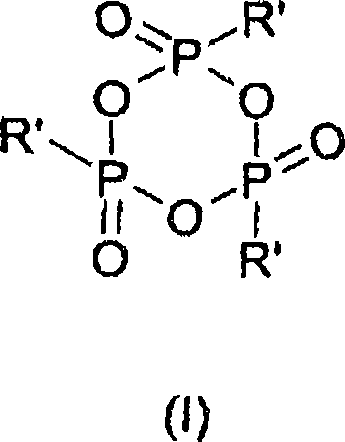
The novel approach leverages the unique reactivity of the phosphonic anhydride bond, which activates the substrate gently yet effectively. Unlike the chaotic reactivity of POCl3, this system operates with maximal regio- and stereoselectivity, ensuring that even sensitive functional groups remain untouched. The patent data indicates that this method is not only theoretically superior but practically robust, capable of delivering yields typically ranging from 90% to 100%, with selectivity often exceeding 99%. This level of performance transforms the synthesis of nitriles from a problematic bottleneck into a reliable, scalable unit operation suitable for the rigorous demands of commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Phosphonic Anhydride-Catalyzed Dehydration
To fully appreciate the value proposition for R&D teams, one must understand the mechanistic elegance of this transformation. The reaction proceeds through the activation of the carboxylic acid or amide oxygen by the electrophilic phosphorus center of the anhydride. This activation generates a highly reactive mixed anhydride or acyl-phosphonate intermediate in situ. In the presence of a base, such as triethylamine or pyridine, the subsequent elimination of the phosphonic acid moiety occurs smoothly to form the carbon-nitrogen triple bond. Crucially, this mechanism avoids the formation of highly acidic or basic micro-environments that typically trigger racemization. The cyclic structure of the preferred reagents, such as T3P, imposes steric constraints that further enhance selectivity, preventing unwanted side reactions that plague linear anhydrides.
Furthermore, the impurity profile generated by this process is inherently cleaner than that of conventional methods. The primary byproduct is a phosphonic acid derivative which, as noted in the patent description, is generally well water-soluble. This physicochemical property is a game-changer for purification strategies. Instead of relying on expensive silica gel chromatography to separate organic-soluble urea byproducts (as seen with DCC), manufacturers can employ simple aqueous workups. The reaction mixture can be hydrolyzed, and the phases separated, leaving the high-purity nitrile in the organic layer. This mechanistic feature directly translates to reduced processing time and lower solvent consumption, addressing key concerns for cost reduction in pharmaceutical manufacturing while maintaining stringent quality standards.
How to Synthesize Nitriles Efficiently
The practical implementation of this technology involves a straightforward protocol that balances reaction kinetics with safety. The process typically begins by predisposing the N-alkylamine or carboxylic acid substrate in a suitable aprotic solvent such as ethyl acetate, THF, or dichloromethane. The phosphonic anhydride is then metered in, either as a melt or a solution, allowing for precise control over the exotherm. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by dissolving the carboxylic acid or N-alkylcarboxamide substrate in an aprotic organic solvent such as ethyl acetate or THF, optionally adding a tertiary amine base.
- Meter in the phosphonic anhydride reagent (such as T3P) either as a melt or a solution while maintaining the temperature between 0°C and 120°C depending on substrate sensitivity.
- Stir the mixture for 1 to 12 hours to ensure complete conversion, followed by hydrolysis and simple phase separation to isolate the high-purity nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of phosphonic anhydride-mediated nitrile synthesis offers tangible strategic benefits beyond mere chemical elegance. The shift away from hazardous reagents like POCl3 and difficult-to-remove coupling agents like DCC fundamentally alters the cost structure and risk profile of production. By implementing this technology, organizations can achieve significant cost savings through the elimination of complex purification trains. The ability to isolate products via simple phase separation drastically reduces the consumption of chromatography media and solvents, which are major cost drivers in fine chemical production. Moreover, the high selectivity of the reaction minimizes the formation of hard-to-remove impurities, leading to higher overall throughput and reduced waste disposal costs associated with off-spec material.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzed through the lens of process efficiency. Traditional methods often require stoichiometric amounts of reagents that generate solid byproducts, necessitating filtration and extensive washing steps. In contrast, the phosphonic anhydride route generates water-soluble byproducts that partition into the aqueous phase during workup. This simplification of the downstream processing workflow removes the need for expensive and time-consuming chromatographic purification. Consequently, the overall cost of goods sold (COGS) is drastically simplified, allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized, hazardous reagents that have volatile availability. Phosphonic anhydrides like T3P are commercially available and stable, offering a more secure sourcing strategy compared to reagents that require strict storage conditions or have limited suppliers. Furthermore, the mild reaction conditions (typically 0°C to 120°C) reduce the energy load on manufacturing facilities and minimize the risk of thermal runaways. This operational stability ensures consistent batch-to-batch quality and reduces lead time for high-purity nitriles, enabling manufacturers to respond more agilely to fluctuating market demands.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste generation and solvent usage intensify, the environmental profile of a synthetic route becomes a critical selection criterion. This method excels in scalability because the workup does not generate large volumes of solid waste (like urea precipitates) that are difficult to dispose of. The water-soluble nature of the phosphonic byproducts facilitates easier wastewater treatment protocols. Additionally, the high atom economy and selectivity mean that less raw material is wasted on side products. This aligns perfectly with green chemistry principles, making the commercial scale-up of complex nitriles not only technically feasible but also environmentally sustainable and compliant with increasingly strict global regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dehydration technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making.
Q: Why is phosphonic anhydride preferred over POCl3 for nitrile synthesis?
A: Unlike phosphoryl chloride (POCl3), which is highly reactive and non-selective, phosphonic anhydrides offer exceptional chemoselectivity. They allow for the conversion of carboxylic acids to nitriles without affecting other sensitive functional groups or causing epimerization of chiral centers, which is critical for pharmaceutical intermediate quality.
Q: Does this method prevent racemization in chiral molecules?
A: Yes, the patent data explicitly highlights that this method proceeds with maximal regio- and stereoselectivity. It avoids the drastic conditions associated with traditional reagents like DCC or SOCl2, thereby preventing the epimerization of remotely spaced stereocenters often found in complex drug molecules.
Q: How is the byproduct removed during the workup process?
A: One of the significant commercial advantages is the ease of purification. The secondary products formed from the phosphonic anhydride are generally well water-soluble. This allows for isolation of the desired nitrile through simple hydrolysis and phase separation, eliminating the need for complex chromatographic purification steps required by urea byproducts in DCC methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrile Intermediate Supplier
The transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging technologies like the phosphonic anhydride dehydration process to deliver superior value to our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed in the laboratory are faithfully reproduced on an industrial scale. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped to analyze complex impurity profiles and ensure that every batch meets the exacting standards required by the pharmaceutical industry.
We invite you to explore how this innovative nitrile synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced capabilities can support your next breakthrough in drug development.
