Advanced Cyanide-Free Synthesis of Aminomethylbenzoic Acid for Global Pharmaceutical Supply Chains
Advanced Cyanide-Free Synthesis of Aminomethylbenzoic Acid for Global Pharmaceutical Supply Chains
The pharmaceutical industry is constantly seeking robust, safe, and scalable synthetic routes for critical intermediates, particularly those used in hemostatic agents and skincare formulations. Patent CN108623488B introduces a groundbreaking methodology for the synthesis of aminomethylbenzoic acid, a key precursor for tranexamic acid and various dermatological applications. This technology addresses long-standing safety and efficiency concerns associated with traditional manufacturing processes by eliminating the use of highly toxic cyanide reagents and hazardous organic solvents. By leveraging a novel thiadiazole-mediated amination strategy, this process offers a pathway to high-purity products with significantly reduced environmental footprints. For global procurement teams and R&D directors, understanding this shift is crucial for securing reliable supply chains that meet increasingly stringent regulatory standards regarding worker safety and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of aminomethylbenzoic acid has relied heavily on routes involving p-cyanobenzyl chloride or similar cyanide-containing starting materials. These conventional pathways present severe drawbacks, primarily centered around the extreme toxicity of the cyanide group (-CN), which poses significant risks to operator safety and requires complex, costly waste treatment protocols. Furthermore, traditional methods often employ benzene or other highly polluting organic solvents to drive the reaction, exacerbating environmental liabilities and increasing the cost of solvent recovery and disposal. From a process efficiency standpoint, these older routes frequently suffer from sluggish reaction kinetics, necessitating prolonged reaction times and harsh conditions that can degrade product quality. The resulting crude products often contain difficult-to-remove impurities, leading to lower overall yields, sometimes dropping as low as 65% in comparative studies, which directly impacts the cost of goods sold and supply reliability.
The Novel Approach
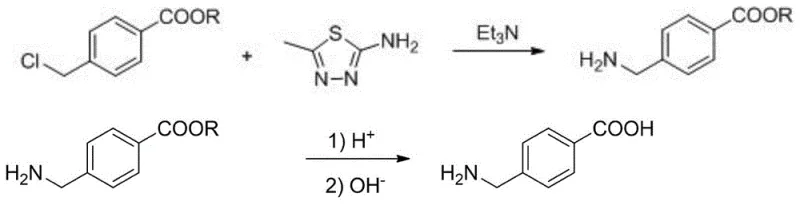
In stark contrast, the method disclosed in patent CN108623488B utilizes 4-halomethyl alkyl benzoate as the starting material, reacting it with 2-amino-5-methyl-1,3,4-thiadiazole and triethylamine in a benign ethanol-water solvent system. This innovative approach completely bypasses the need for toxic cyanide sources, fundamentally altering the safety profile of the manufacturing process. The reaction proceeds under mild thermal conditions, typically between 70°C and 100°C, which reduces energy consumption and minimizes the formation of thermal degradation byproducts. The use of an ethanol-water mixture not only serves as a greener solvent alternative but also enhances the solubility of the reagents, facilitating a more homogeneous and efficient reaction environment. Post-reaction processing is streamlined through simple evaporation and filtration, allowing for the direct isolation of the intermediate ester with high purity, thereby eliminating the need for complex chromatographic separations or extensive washing procedures that plague older methodologies.
Mechanistic Insights into Thiadiazole-Mediated Amination and Hydrolysis
The core innovation of this synthetic route lies in the unique role of 2-amino-5-methyl-1,3,4-thiadiazole combined with triethylamine to effectuate the amination of the benzylic halide. Unlike the classical Delépine reaction which uses hexamethylenetetramine (urotropine), this system appears to facilitate a nucleophilic substitution where the thiadiazole derivative acts as a highly effective nitrogen source or catalyst that promotes the displacement of the halide group. The presence of triethylamine serves to scavenge the generated acid (HX), driving the equilibrium forward and preventing the protonation of the amine product, which could otherwise inhibit further reaction. This synergistic interaction allows the reaction to proceed rapidly, often completing within 0.5 to 3 hours, which is a significant acceleration compared to the multi-hour refluxes required in traditional ammoniation processes. The result is the formation of the 4-aminomethyl alkyl benzoate intermediate with exceptional selectivity, minimizing the formation of secondary or tertiary amine byproducts that typically complicate purification.
Following the isolation of the ester intermediate, the second stage involves acid-catalyzed hydrolysis to yield the final carboxylic acid. The patent specifies the use of concentrated sulfuric acid, phosphoric acid, or nitric acid, with sulfuric acid being particularly effective. The ester bond is cleaved under controlled heating (50-90°C), converting the alkyl benzoate into the free acid. A critical aspect of this step is the work-up procedure; instead of complex extraction sequences, the reaction mixture is simply cooled, diluted with water, and neutralized with a base such as ammonia water or sodium hydroxide. This pH adjustment causes the zwitterionic aminomethylbenzoic acid to precipitate out of the solution as a white solid. This precipitation behavior is indicative of the high purity of the intermediate; because few side products were formed in the first step, the final crystallization yields a product with purity levels consistently exceeding 94%, often reaching 99% without the need for recrystallization.
How to Synthesize Aminomethylbenzoic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible framework for manufacturing this valuable intermediate. The process begins with the precise mixing of 4-halomethyl alkyl benzoate and triethylamine, followed by the controlled addition of the thiadiazole reagent in an ethanol-water solution. Maintaining the temperature within the 70-100°C range is critical for optimizing the reaction rate while preventing solvent loss. Once the intermediate is isolated via filtration, it is immediately subjected to acid hydrolysis. The detailed operational parameters, including specific molar ratios and solvent concentrations, are essential for achieving the reported high yields. For a comprehensive guide on the exact stoichiometric calculations and safety precautions required for scale-up, please refer to the standardized synthesis steps provided below.
- React 4-halomethyl alkyl benzoate with triethylamine and 2-amino-5-methyl-1,3,4-thiadiazole in an ethanol-water solvent system at 70-100°C to form the ester intermediate.
- Isolate the 4-aminomethyl alkyl benzoate intermediate by evaporating the solvent, cooling, and filtering the precipitated solid.
- Hydrolyze the ester intermediate using concentrated sulfuric acid at 50-90°C, followed by neutralization with alkali to precipitate pure aminomethylbenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical advantages that extend beyond simple yield metrics. The elimination of cyanide-based reagents removes a major regulatory hurdle, simplifying the permitting process for manufacturing facilities and reducing the insurance premiums associated with handling extremely hazardous substances. Furthermore, the shift from toxic solvents like benzene to an ethanol-water system drastically lowers the cost of solvent procurement and waste disposal, as ethanol is cheaper, more readily available, and easier to recover or treat than chlorinated or aromatic solvents. The simplified work-up procedure, which relies on precipitation rather than complex extractions, reduces the demand for specialized equipment and labor hours, contributing to a leaner manufacturing operation. These factors collectively drive down the total cost of ownership for the intermediate, making it a more attractive option for long-term supply contracts.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of expensive and hazardous raw materials. By avoiding the use of cyanide and benzene, manufacturers eliminate the need for specialized containment systems and costly hazardous waste incineration services. Additionally, the high reaction efficiency means that less raw material is wasted on side products, maximizing the atom economy of the process. The ability to isolate the intermediate through simple evaporation and filtration rather than energy-intensive distillation or chromatography further reduces utility costs. Consequently, the overall production cost is significantly lowered, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the strict regulations surrounding the transport and storage of toxic chemicals. Since this new method utilizes widely available and less regulated starting materials like 4-halomethyl benzoates and ethanol, the risk of supply disruption due to regulatory crackdowns or transportation bans is minimized. The robustness of the reaction conditions also means that production is less susceptible to minor fluctuations in temperature or pressure, ensuring consistent batch-to-batch quality. This reliability is crucial for downstream pharmaceutical manufacturers who require just-in-time delivery of high-quality intermediates to maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental issues, but this methodology is inherently designed for green chemistry principles. The use of water as a co-solvent and the absence of heavy metal catalysts mean that the effluent generated is much easier to treat, helping facilities meet increasingly strict environmental discharge standards. The mild reaction temperatures reduce the energy load on the plant, contributing to a lower carbon footprint. These environmental advantages not only ensure compliance with current regulations but also future-proof the supply chain against tightening global environmental laws, making it a sustainable choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: Why is the thiadiazole-mediated method safer than traditional cyanide routes?
A: Traditional methods often utilize p-cyanobenzyl chloride, introducing highly toxic cyanide groups (-CN) into the process. The novel method described in patent CN108623488B completely eliminates the use of cyanide and toxic solvents like benzene, significantly reducing occupational health risks and environmental hazards.
Q: What are the yield and purity improvements of this new synthetic route?
A: Experimental data indicates that this method achieves product yields ranging from 85% to 91% with purity levels between 94% and 99%. This represents a substantial improvement over comparative methods which often struggle with yields as low as 65% due to side reactions and difficult purification steps.
Q: How does this process impact industrial scalability and waste treatment?
A: The process utilizes mild reaction conditions (70-100°C) and common solvents like ethanol and water, avoiding the need for specialized high-pressure equipment. Furthermore, the absence of heavy metals and toxic cyanides simplifies the 'three wastes' treatment, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminomethylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists has extensively analyzed the potential of the thiadiazole-mediated synthesis described in patent CN108623488B and is fully prepared to implement this advanced route for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aminomethylbenzoic acid meets the highest international standards for pharmaceutical intermediates.
We invite procurement leaders and R&D directors to collaborate with us to leverage this superior manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this green synthesis route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of safety, quality, and economic efficiency.
