Advanced Delbin Reaction Technology for Commercial Aminomethylbenzoic Acid Production
Advanced Delbin Reaction Technology for Commercial Aminomethylbenzoic Acid Production
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with economic viability, particularly for critical intermediates like aminomethylbenzoic acid (CAS 56-91-7). A significant technological breakthrough in this domain is detailed in patent CN112409196A, which discloses a novel preparation process based on the Delbin reaction mechanism. This method fundamentally shifts the paradigm from traditional, energy-intensive ammoniation processes to a more controlled quaternization-hydrolysis sequence. By leveraging urotropine as a nitrogen source, the process achieves exceptional purity levels exceeding 99.8% while mitigating the formation of stubborn dimer impurities that plague conventional synthesis. For R&D directors and procurement strategists, understanding this shift is crucial for securing a reliable aminomethylbenzoic acid supplier capable of meeting stringent quality standards without inflating production costs.
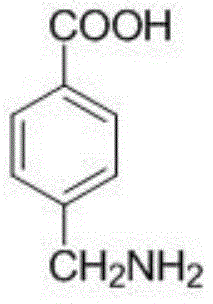
The structural integrity of the final product is paramount for downstream drug synthesis, and the molecular architecture shown above represents the target specification achieved through this optimized pathway. The ability to consistently produce this specific isomer with minimal regio-isomeric contamination is a key differentiator in the competitive landscape of pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of aminomethylbenzoic acid has relied heavily on the direct ammoniation of halogenated methylbenzoic acids, a route fraught with thermodynamic and kinetic inefficiencies. In the most common legacy process, p-methyl benzoic acid is chlorinated under harsh illumination conditions to generate p-chloromethyl benzoic acid, which is then subjected to ammoniation using massive excesses of ammonium bicarbonate and ammonia water. This approach suffers from severe drawbacks, primarily the generation of aminomethylbenzoic acid dimers as persistent by-products, which drastically lowers the overall yield and complicates purification. Furthermore, the reliance on excessive aqueous ammonia necessitates a subsequent, energy-intensive step of heating under reduced pressure to strip residual ammonia, creating a significant burden on utility costs and environmental compliance systems. Alternative routes utilizing bromination reagents, while offering marginally better yields, introduce prohibitive raw material costs due to the high price of bromine, rendering them economically unfeasible for large-scale commercial adoption.
The Novel Approach
The innovative process described in the patent data circumvents these historical bottlenecks by employing the Delbin reaction, a sophisticated transformation that utilizes urotropine (hexamethylenetetramine) as the aminating agent. Instead of a direct, chaotic nucleophilic substitution with ammonia, the process first forms a stable quaternary ammonium salt intermediate through the reaction of p-chloromethylbenzoic acid with urotropine in an aqueous medium. This intermediate is subsequently hydrolyzed under acidic conditions using hydrogen chloride gas in an ethanol solvent system. This two-stage mechanism effectively suppresses the formation of dimer by-products, as the bulky urotropine moiety prevents intermolecular coupling during the substitution phase. The result is a streamlined workflow that eliminates the need for excessive ammonia stripping, reduces solvent consumption through efficient recycling protocols, and delivers a crude product that requires only simple water recrystallization to reach pharmaceutical-grade purity.
Mechanistic Insights into Urotropine-Mediated Delbin Reaction
The core of this technological advancement lies in the precise mechanistic control offered by the urotropine-mediated pathway, which transforms a typically messy alkylation into a highly selective synthesis. The reaction initiates with the nucleophilic attack of the tertiary amines within the urotropine cage on the benzylic carbon of the p-chloromethylbenzoic acid. This step is kinetically favored at moderate temperatures around 50°C, ensuring complete conversion of the starting material while preventing thermal degradation of the carboxylic acid functionality. The resulting quaternary ammonium salt is isolable and stable, serving as a protected form of the amine that shields the reactive center from side reactions. Upon exposure to anhydrous hydrogen chloride gas in ethanol, the quaternary salt undergoes acid-catalyzed hydrolysis. The acidic environment protonates the nitrogen atoms of the urotropine cage, destabilizing the C-N bonds and releasing the free primary amine along with formaldehyde and ammonium chloride by-products. This hydrolysis step is critical; conducting it in ethanol rather than water enhances the solubility of the organic intermediate while facilitating the precipitation of inorganic salts, thereby simplifying the workup procedure.
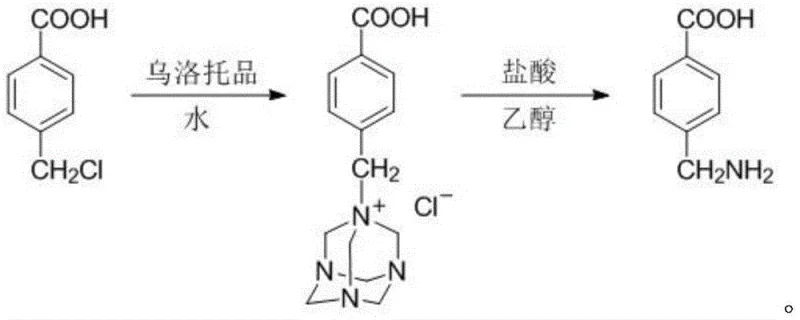
As illustrated in the reaction scheme above, the transformation proceeds through a distinct cationic intermediate before yielding the final aminomethylbenzoic acid hydrochloride species, which is then neutralized to the free base. This mechanistic clarity allows for rigorous impurity control; because the dimerization pathway is sterically blocked by the urotropine group, the impurity profile is significantly cleaner than that of direct ammoniation. For quality control teams, this means fewer unknown peaks in HPLC chromatograms and a much more predictable crystallization behavior during the final purification stage. The ability to tune the pH precisely during the hydrolysis (pH < 3) and neutralization (pH 8) steps further ensures that the zwitterionic nature of the amino acid is managed effectively to maximize recovery yields between 85% and 88%.
How to Synthesize Aminomethylbenzoic Acid Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management, particularly during the exothermic quaternization and gas absorption phases. The patent outlines a scalable protocol that begins with dissolving urotropine in water followed by the batch-wise addition of the chloro-precursor to manage heat evolution. Following the reaction and solvent removal, the critical hydrolysis step demands precise temperature control between 10-15°C to prevent side reactions while introducing HCl gas. The subsequent neutralization and mother liquor recycling strategy are designed to maximize atom economy, allowing manufacturers to extract value from what would otherwise be waste streams. For detailed operational parameters and safety guidelines regarding the handling of hydrogen chloride gas and ethanol solvents, please refer to the standardized synthesis instructions provided below.
- Dissolve urotropine in water and add p-chloromethylbenzoic acid in batches at room temperature, then heat to 50°C for reaction followed by solvent evaporation.
- Add ethanol to the residue, cool to 10-15°C, and introduce hydrogen chloride gas to adjust pH below 3 for hydrolysis.
- Neutralize with sodium hydroxide ethanol solution to pH 8, cool to precipitate crude product, and recycle mother liquor for additional yield.
- Combine crude products and recrystallize using 25 times volume of pure water to obtain refined aminomethylbenzoic acid with >99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this Delbin-based process represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of the energy-intensive ammonia stripping step translates directly into reduced utility consumption, lowering the variable cost per kilogram of the final API intermediate. Moreover, by avoiding the use of expensive brominating agents and relying on cost-effective chlorinated precursors, the raw material bill of materials is significantly optimized without sacrificing yield. The process's inherent ability to suppress dimer formation reduces the burden on downstream purification, meaning less solvent is consumed for recrystallization and less product is lost during waste treatment. These factors combine to create a manufacturing profile that is not only more economical but also more resilient to fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic simplification of the post-reaction workup. Traditional methods require substantial energy input to remove excess ammonia, often involving vacuum distillation at elevated temperatures which strains equipment and increases electricity or steam costs. By replacing aqueous ammonia with solid urotropine and gaseous HCl, this new process removes that entire energy sink. Additionally, the high selectivity of the reaction minimizes the formation of hard-to-remove dimer impurities, reducing the need for multiple recrystallization cycles or expensive chromatographic purification. This streamlined workflow allows for a substantial reduction in overall production costs, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis routes that rely on hazardous or volatile reagents. The use of urotropine, a stable and widely available solid, mitigates the risks associated with storing and transporting large volumes of concentrated ammonia water. Furthermore, the process demonstrates excellent scalability, with the patent examples showing consistent yields across multiple batches when mother liquors are recycled. This robustness ensures that suppliers can maintain steady output levels even during periods of high demand, reducing the risk of stockouts for downstream pharmaceutical manufacturers who rely on just-in-time delivery models for their own production schedules.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner alternative to legacy methods. The avoidance of excessive ammonia emissions reduces the load on scrubber systems and minimizes the risk of atmospheric pollution. The solvent system, primarily water and ethanol, is relatively benign and easily recoverable, aligning with green chemistry principles. The ability to recycle mother liquors for up to three batches before concentration further minimizes liquid waste generation. For facilities operating under strict environmental regulations, adopting this technology simplifies compliance reporting and reduces the costs associated with waste disposal, thereby enhancing the long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Delbin reaction process for aminomethylbenzoic acid production. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process validation and vendor qualification.
Q: How does the Delbin reaction improve yield compared to traditional ammoniation?
A: Traditional ammoniation often produces significant amounts of aminomethylbenzoic acid dimer by-products due to uncontrolled nucleophilic attacks. The Delbin reaction utilizes urotropine to form a stable quaternary ammonium intermediate, which sterically hinders dimerization and ensures a cleaner conversion to the primary amine upon acid hydrolysis, resulting in yields of 85-88%.
Q: What are the energy efficiency benefits of this process?
A: Conventional methods require excessive ammonia water or ammonium salts, necessitating energy-intensive heating under reduced pressure to strip residual ammonia. This patented process avoids large volumes of aqueous ammonia entirely, eliminating the high-energy ammonia removal step and significantly reducing overall utility consumption.
Q: Why is p-chloromethylbenzoic acid preferred over brominated analogues?
A: While bromination routes can offer slightly higher reactivity, the cost of bromine reagents is substantially higher than chlorine-based reagents. This process demonstrates that p-chloromethylbenzoic acid, when activated via the Delbin mechanism, provides an optimal balance of reactivity and cost-effectiveness suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminomethylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the Delbin reaction is critical for maintaining a competitive edge in the pharmaceutical intermediates sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in laboratory patents are faithfully reproduced on an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of aminomethylbenzoic acid meets the exacting standards required for drug substance synthesis. Our commitment to process excellence means we can offer a supply of high-purity aminomethylbenzoic acid that is both consistent and compliant with international regulatory requirements.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this optimized manufacturing route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic advantages of switching to our Delbin-derived material. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing you to validate our capabilities and secure a partnership that drives value and efficiency for your organization.
