Advanced Bifunctional Thiourea Organocatalysts: Enabling High-Precision Asymmetric Synthesis
The landscape of asymmetric synthesis is undergoing a paradigm shift towards sustainable and efficient organocatalysis, driven by the urgent need to eliminate toxic heavy metals from pharmaceutical supply chains. Patent CN102167698B introduces a groundbreaking class of difunctional thiourea organic small molecule catalysts that leverage the rigid chiral framework of Cinchona alkaloids combined with versatile amino acid derivatives. This innovation addresses critical limitations in traditional catalytic systems by offering a robust, bifunctional architecture capable of simultaneous activation of reaction partners through hydrogen bonding and Lewis base interactions. The structural versatility of these catalysts, defined by the general formula (I), allows for precise modulation of steric hindrance and electronic effects, ensuring exceptional stereoselectivity across a broad range of asymmetric transformations.
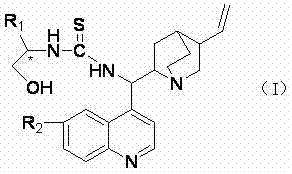
For R&D directors seeking reliable pharmaceutical intermediate supplier partnerships, understanding the mechanistic superiority of these organocatalysts is paramount. Unlike conventional methods that often rely on expensive transition metal complexes requiring rigorous removal protocols, this technology utilizes abundant natural products as starting materials. The introduction of multiple hydrogen bond donors within the thiourea motif creates a highly organized transition state environment, facilitating precise chiral induction. This patent not only details the molecular design but also validates the practical applicability of these catalysts in demanding reactions such as asymmetric Michael additions, where high enantiomeric excess is non-negotiable for regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to asymmetric catalysis have long been dominated by transition metal complexes, which, despite their efficacy, present significant logistical and safety challenges for large-scale manufacturing. These conventional systems frequently necessitate catalyst loadings as high as 20 mol% to 30 mol% to achieve acceptable conversion rates, leading to substantial increases in production costs and waste generation. Furthermore, the presence of residual heavy metals in the final active pharmaceutical ingredient (API) poses severe regulatory hurdles, requiring additional, costly purification steps to meet stringent ppm limits. The reliance on scarce precious metals also introduces supply chain volatility, making long-term planning difficult for procurement managers who prioritize cost reduction in organocatalyst manufacturing.
The Novel Approach
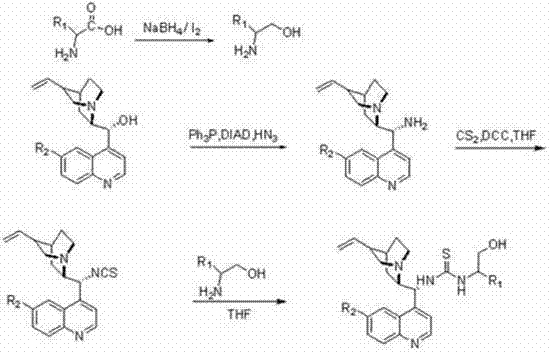
The novel synthetic route disclosed in the patent offers a transformative solution by utilizing cheap and easy-to-obtain Cinchona alkaloids as the chiral backbone. This approach fundamentally simplifies the catalyst architecture while enhancing performance through a bifunctional design that mimics enzymatic activity. By integrating a thiourea moiety with the quinuclidine nitrogen of the alkaloid, the catalyst achieves dual activation capabilities that significantly lower the energy barrier for reaction without the need for excessive catalyst loading. The synthetic pathway is remarkably straightforward, involving the conversion of amino acids to amino alcohols and their subsequent coupling with cinchona-derived isothiocyanates, ensuring that the process is amenable to commercial scale-up of complex polymer additives and fine chemicals alike.
Mechanistic Insights into Bifunctional Thiourea Catalysis
The exceptional performance of these catalysts stems from their unique ability to function as both Lewis bases and hydrogen bond donors simultaneously. The tertiary amine nitrogen within the quinuclidine ring acts as a Lewis base, effectively activating nucleophilic reagents by increasing their electron density. Concurrently, the thiourea group serves as a potent hydrogen bond donor, coordinating with electrophilic substrates such as nitroalkenes to lower their LUMO energy levels. This synergistic dual activation brings the reactants into close proximity within a well-defined chiral pocket, enforcing a specific trajectory for bond formation that results in high stereoselectivity. The rigidity of the Cinchona scaffold prevents conformational flexibility that could otherwise lead to racemic background reactions, ensuring that the chiral information is efficiently transferred to the product.
Impurity control is inherently superior in this system due to the absence of metal-mediated side reactions such as oxidation or homocoupling, which are common pitfalls in transition metal catalysis. The organic nature of the catalyst ensures that degradation products are structurally related to the starting materials, simplifying the impurity profile and facilitating easier purification via standard chromatographic techniques. This mechanistic clarity provides R&D teams with the confidence to apply these catalysts to sensitive substrates without fear of unpredictable metal-ligand interactions. The versatility of the R1 and R2 substituents further allows for fine-tuning the electronic environment to match specific substrate requirements, optimizing yield and enantiomeric excess for diverse chemical transformations including the asymmetric Michael addition demonstrated in the patent embodiments.
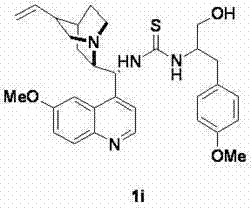
How to Synthesize Bifunctional Thiourea Catalysts Efficiently
The preparation of these high-performance catalysts follows a logical and scalable three-step sequence that avoids hazardous reagents and extreme conditions. The process begins with the reduction of chiral alpha-amino acids to their corresponding amino alcohols using a mild sodium borohydride and iodine system, preserving the chiral integrity of the starting material. Simultaneously, the Cinchona alkaloid backbone is functionalized at the C9 position via an azide substitution followed by conversion to an isothiocyanate using carbon disulfide and DCC. The final convergence step involves the nucleophilic attack of the amino alcohol on the isothiocyanate, forming the stable thiourea linkage that defines the catalyst's active site. Detailed standardized synthesis steps see the guide below.
- Reduce chiral alpha-amino acids to corresponding amino alcohols using sodium borohydride and iodine in THF.
- Convert Cinchona alkaloids to 9-amino derivatives via azide substitution, followed by reaction with carbon disulfide to form isothiocyanates.
- Couple the cinchona isothiocyanate with the chiral amino alcohol in THF to yield the final bifunctional thiourea catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this technology offers profound advantages by decoupling catalyst production from the volatile markets of precious metals. The reliance on Cinchona alkaloids, which are harvested from renewable botanical sources, ensures a stable and predictable supply chain that is less susceptible to geopolitical disruptions compared to mined metal ores. For procurement managers, this translates into significant cost savings not just in the raw material acquisition but also in the downstream processing, as the elimination of metal scavengers reduces the overall bill of materials. The simplified workflow reduces the operational complexity of the manufacturing suite, allowing for faster batch turnover and improved asset utilization rates without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic impact of switching to this organocatalytic system is driven by the drastic reduction in catalyst loading requirements and the elimination of expensive metal removal technologies. By avoiding the need for specialized equipment to handle toxic heavy metals, facilities can operate with lower capital expenditure and reduced maintenance costs associated with corrosion and contamination control. The use of commodity chemicals like sodium borohydride and carbon disulfide further drives down the variable costs per kilogram of catalyst produced, creating a compelling business case for adoption in high-volume production environments.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available natural product feedstocks that have established global distribution networks. Unlike specialized ligands that may have single-source suppliers with long lead times, Cinchona alkaloids and standard amino acids are stocked by multiple vendors worldwide, mitigating the risk of stockouts. This redundancy allows supply chain heads to negotiate better terms and maintain buffer stocks more easily, ensuring continuous production schedules even during periods of market fluctuation or logistical bottlenecks.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower, aligning with modern green chemistry principles and increasingly strict regulatory frameworks regarding waste disposal. The absence of heavy metals simplifies wastewater treatment protocols and reduces the classification of waste streams as hazardous, leading to substantial savings in disposal fees. Furthermore, the robustness of the reaction conditions allows for seamless translation from gram-scale laboratory optimization to multi-ton commercial production, ensuring that process parameters remain consistent and controllable regardless of batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering a clear picture of performance expectations. Understanding these nuances helps stakeholders make informed decisions about integrating this methodology into their existing process workflows.
Q: What represents the primary advantage of this bifunctional catalyst over traditional metal-based systems?
A: The primary advantage lies in its metal-free composition, which eliminates the risk of heavy metal contamination in pharmaceutical products, alongside its ability to activate both nucleophiles and electrophiles simultaneously for superior stereocontrol.
Q: Can the steric and electronic properties of this catalyst be tuned for specific reactions?
A: Yes, the modular design allows for significant tunability by varying the R1 group on the amino alcohol moiety and selecting different Cinchona alkaloid backbones, enabling precise optimization for diverse substrate scopes.
Q: Is this catalyst suitable for large-scale industrial asymmetric Michael additions?
A: Absolutely. The synthesis utilizes cheap, readily available raw materials like Cinchona alkaloids and avoids complex purification steps, making it highly scalable and cost-effective for commercial manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bifunctional Thiourea Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced organocatalysts play in the development of next-generation pharmaceutical intermediates and fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst meets the highest standards of enantiomeric purity and chemical consistency required by global regulatory bodies.
We invite you to collaborate with us to unlock the full potential of this bifunctional thiourea technology for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current manufacturing processes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our supply solutions can enhance your operational efficiency and reduce your total cost of ownership.
