Advanced Chemical Synthesis of Hydrocortisone: Enhancing Purity and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways for critical corticosteroids like hydrocortisone, balancing purity requirements with economic feasibility. Patent CN101397323A introduces a transformative approach to the preparation of hydrocortisone and its derivatives, shifting away from the limitations of traditional biosynthetic methods. This innovation utilizes 17-hydroxyl-4,9-diene-pregna-3,20-diketone as a strategic starting material, enabling a concise sequence of 9,11-transformations and 21-functionalizations. For R&D directors and procurement specialists, this patent represents a pivotal opportunity to optimize the supply chain for adrenal cortex hormone drugs. By leveraging existing intermediates already produced within the facility, the method not only streamlines the operational workflow but also facilitates the simultaneous production of triamcinolone and anecortave acetate series, thereby maximizing asset utilization and reducing the overall cost burden associated with sterile hormone manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of hydrocortisone has relied heavily on microbial fermentation or convoluted chemical syntheses that present significant scalability challenges. Traditional biosynthetic routes, such as those utilizing Absidia coerulea or Curvularia lunata, are fundamentally constrained by the poor aqueous solubility of steroidal substrates, typically ranging from 10-6 to 10-5 Mol/L. This physical limitation creates a mass transfer bottleneck where the substrate cannot effectively contact the intracellular 11-beta-hydroxylase enzymes, leading to sluggish reaction kinetics and suboptimal yields. Furthermore, older chemical synthesis documents describe routes requiring expensive auxiliary materials like dithioglycol for ketone protection, which necessitates additional deprotection steps and generates hazardous sulfur-containing waste streams. These legacy methods often involve lengthy reaction sequences with multiple purification stages, driving up both the operational expenditure and the environmental compliance costs for manufacturers attempting to scale production to meet global demand.
The Novel Approach
In stark contrast, the methodology disclosed in CN101397323A offers a streamlined chemical alternative that bypasses the solubility issues inherent in fermentation. By initiating the synthesis with 17-hydroxyl-4,9-diene-pregna-3,20-diketone, a versatile intermediate, the process achieves a direct functionalization of the steroid nucleus without the need for complex protecting group strategies. This novel route capitalizes on the structural features of the diene system to facilitate selective halogenation and subsequent rearrangement, effectively installing the critical 11-beta-hydroxyl and 21-hydroxyl functionalities in fewer steps. The ability to derive multiple high-value products, including hydrocortisone esters and triamcinolone precursors, from a single intermediate line creates a synergistic manufacturing effect. This approach not only simplifies the process flow but also enhances the economic viability of the operation by distributing fixed costs across a broader product portfolio, making it an attractive option for large-scale commercial production.
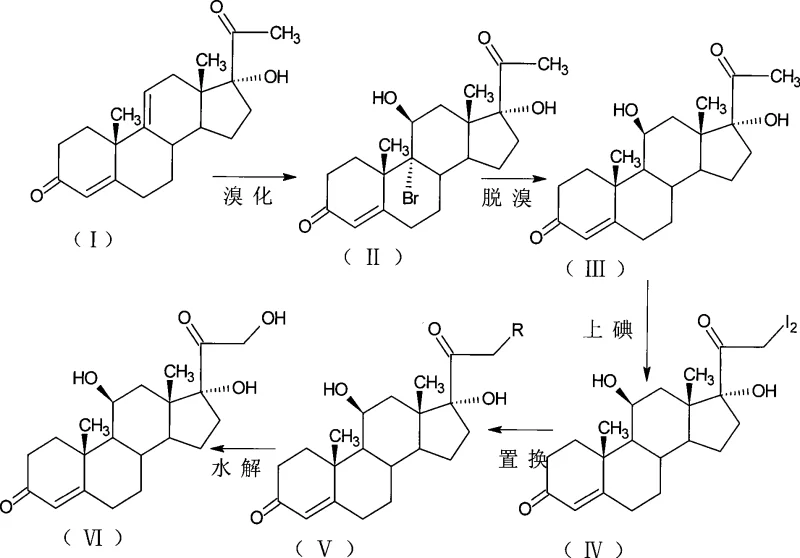
Mechanistic Insights into Steroidal Halogenation and Substitution
The core of this synthetic strategy lies in the precise control of regioselectivity during the halogenation and reduction phases. The initial bromination step targets the 9-alpha position of the 4,9-diene system, utilizing reagents like N-bromosuccinimide (NBS) or dibromomalononitrile in polar aprotic solvents such as tetrahydrofuran. This electrophilic addition is carefully managed at low temperatures, typically between 0°C and 20°C, to prevent over-bromination or degradation of the sensitive enone system. Following this, the debromination step employs mild reducing agents, such as tributyltin hydride or chromium salts, to remove the bromine atom while simultaneously establishing the correct stereochemistry at the 11-position. This sequence effectively mimics the biological hydroxylation but occurs in a homogeneous organic phase where reaction parameters like temperature and concentration can be tightly regulated to minimize the formation of isomeric impurities that often plague fermentation batches.
Subsequent functionalization at the 21-position is achieved through a sophisticated iodination-substitution cascade. The introduction of iodine atoms at the 21-position activates the side chain for nucleophilic attack by alkali metal carboxylates, such as potassium acetate or propionate. This displacement reaction proceeds efficiently in solvents like dimethylformamide (DMF) or pyridine, allowing for the direct formation of hydrocortisone-21-esters. The mechanism ensures that the 21-hydroxyl group is introduced with high fidelity, avoiding the oxidation issues common in other synthetic routes. Finally, mild alkaline hydrolysis cleaves the ester bond to yield the free hydrocortisone alcohol. This modular approach allows manufacturers to halt the synthesis at the ester stage if hydrocortisone acetate or propionate is the desired target, providing flexibility in product output without altering the upstream process conditions.
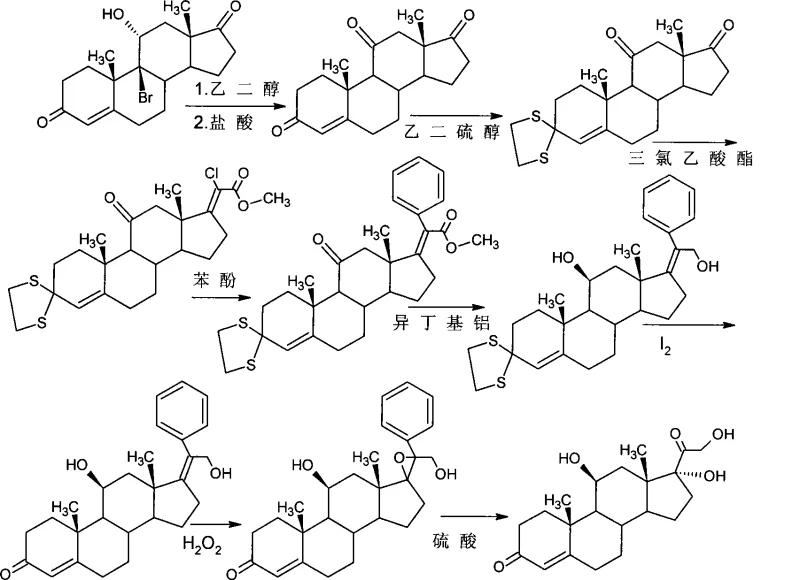
How to Synthesize Hydrocortisone Efficiently
The implementation of this synthesis requires strict adherence to the specified reaction conditions to ensure optimal yield and purity profiles suitable for pharmaceutical applications. The process begins with the dissolution of the starting diene in a selected organic solvent, followed by the controlled addition of the brominating agent under inert atmosphere. Detailed standard operating procedures regarding temperature ramping, quenching protocols, and crystallization parameters are essential for reproducibility. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and workup procedures required to execute this transformation successfully, please refer to the standardized synthesis guide below.
- Perform selective bromination of the starting diene compound using N-bromosuccinimide (NBS) or dibromomalononitrile in organic solvents like THF or acetone at controlled low temperatures.
- Execute debromination using reducing agents such as tributyltin hydride or chromium salts to generate the key 11-beta-hydroxy intermediate.
- Conduct 21-position iodination followed by nucleophilic substitution with alkali metal carboxylates and final hydrolysis to yield high-purity hydrocortisone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this chemical synthesis route offers profound strategic benefits over traditional fermentation-based sourcing. The primary advantage stems from the decoupling of production capacity from biological constraints, such as fungal strain viability and fermentation tank turnover times. By utilizing a purely chemical pathway, manufacturers can achieve consistent batch-to-batch quality and significantly reduce the lead time associated with biological incubation periods. This reliability is crucial for maintaining continuous supply lines for critical anti-inflammatory medications, especially during periods of high market demand or raw material volatility. Furthermore, the use of commodity chemicals and standard reactor equipment lowers the barrier to entry for scaling production, ensuring that supply can be rapidly expanded without the need for specialized bioreactor infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive protecting groups and the reduction in total synthetic steps directly translate to lower raw material consumption and reduced waste disposal costs. By avoiding the use of costly auxiliaries like dithioglycol and minimizing the number of isolation steps, the overall cost of goods sold is substantially decreased. Additionally, the ability to produce multiple steroid derivatives from a common intermediate allows for better inventory management and economies of scale, further driving down the unit cost of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Relying on chemical synthesis mitigates the risks associated with biological variability, such as contamination or strain mutation, which can disrupt fermentation batches. The robustness of the chemical reagents and the simplicity of the reaction conditions ensure a stable and predictable production schedule. This stability is vital for long-term supply agreements, as it guarantees the availability of high-purity hydrocortisone regardless of seasonal or biological fluctuations that might affect traditional manufacturing methods.
- Scalability and Environmental Compliance: The process utilizes standard organic solvents and reagents that are readily available in the global chemical market, facilitating easy scale-up from pilot to commercial tonnage. Moreover, the avoidance of heavy biological waste streams and the potential for solvent recovery systems contribute to a greener manufacturing profile. This alignment with environmental regulations reduces the compliance burden and associated costs, making the operation more sustainable and resilient against tightening environmental policies in key manufacturing regions.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the process capabilities, impurity profiles, and regulatory considerations associated with this patented method. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals.
Q: What are the primary advantages of this chemical route over traditional fermentation?
A: Unlike fermentation which suffers from low steroid solubility (10^-6 to 10^-5 Mol/L) and intracellular enzyme limitations, this chemical route utilizes homogeneous reaction conditions that significantly improve conversion rates and simplify downstream purification.
Q: How does this process impact the cost of hydrocortisone manufacturing?
A: The process eliminates the need for expensive protecting groups like dithioglycol used in older chemical methods and leverages existing company intermediates, resulting in substantial raw material cost savings and reduced processing steps.
Q: Can this synthesis route be adapted for hydrocortisone esters?
A: Yes, the pathway produces hydrocortisone-21-carboxylates as stable intermediates, allowing for the direct isolation of esters like hydrocortisone acetate or propionate without additional esterification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-demand corticosteroids like hydrocortisone. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global pharmaceutical partners. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify the absence of genotoxic impurities and residual solvents. Our facility is equipped to handle the specific reagents and conditions required for this halogenation-substitution cascade, guaranteeing a consistent supply of high-quality intermediates and APIs.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for your product portfolios. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and economic efficiency in the competitive steroid market.
