Advanced Regioselective Synthesis of Hydrocortisone Butyrate for Commercial Scale-up
Advanced Regioselective Synthesis of Hydrocortisone Butyrate for Commercial Scale-up
The pharmaceutical industry constantly seeks robust synthetic routes for potent corticosteroids, particularly for topical applications where purity profiles are critical. Patent CN101891797B introduces a transformative methodology for the preparation of steroidal 17α-esters, specifically targeting compounds like hydrocortisone butyrate. This technology addresses the longstanding challenge of regioselectivity in steroid functionalization, moving away from non-selective hydrolysis methods that generate difficult-to-separate impurity profiles. By leveraging a cyclic ester intermediate strategy coupled with a novel directed hydrolysis system, this process achieves exceptional conversion rates and purity levels. For R&D teams and procurement specialists, understanding this pathway is essential for securing a reliable steroid intermediate supplier capable of delivering high-quality active pharmaceutical ingredients (APIs) consistently.
The core innovation lies in the precise control over the 17-position versus the 21-position during esterification and subsequent hydrolysis. Traditional methods often struggle with the thermodynamic stability of 21-esters, leading to significant byproduct formation. The approach detailed in CN101891797B circumvents this by first protecting the 17,21-dihydroxy system as a cyclic orthoester. This temporary protection allows for subsequent selective deprotection under mild conditions. The structural complexity of the target molecule, as seen in the chemical architecture below, demands such precision to ensure therapeutic efficacy and safety compliance.
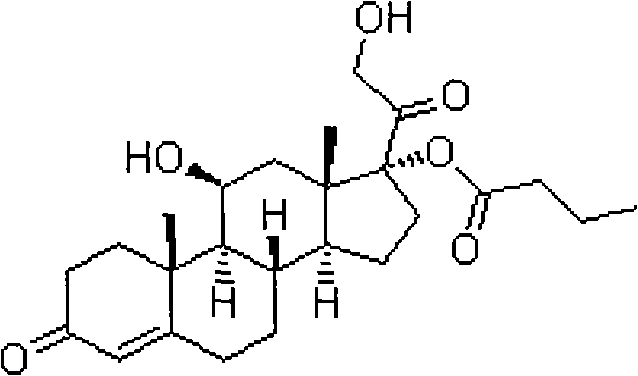
Hydrocortisone butyrate serves as a prime example of the utility of this technology. As a medium-potency adrenocortical hormone, it is widely utilized for treating eczema, dermatitis, and allergic skin conditions. The commercial viability of such compounds depends heavily on the ability to manufacture them without excessive waste or costly purification steps. The patent outlines a route that not only improves yield but also simplifies the downstream processing, making it an attractive option for cost reduction in API manufacturing. This sets the stage for a deeper analysis of why conventional methods fail and how this novel approach succeeds in an industrial setting.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 17α-esters from 17α,21-dihydroxy steroids has been plagued by selectivity issues. Conventional direct esterification or hydrolysis of cyclic esters using strong mineral acids like sulfuric acid often results in a statistical mixture of isomers. Data indicates that sulfuric acid hydrolysis can lead to a 17α-ester to 21-ester ratio of merely 7:3, which is commercially unacceptable for high-purity pharmaceutical grades. Furthermore, alternative solid-state methods utilizing silica gel or ion-exchange resins, such as those described in Japanese patents JP60048998 and JP56040700, have failed to completely eliminate the formation of the unwanted 21-ester isomer. These impurities, often present at levels around 10% or higher in older processes, necessitate rigorous and yield-loss-inducing purification steps like repeated recrystallization or preparative chromatography.
The Novel Approach
The methodology disclosed in CN101891797B represents a paradigm shift by introducing a 'directed reagent' concept during the hydrolysis phase. Instead of relying solely on solvent effects or brute-force acid catalysis, this process incorporates saturated aqueous ammonium chloride solution into the hydrolysis mixture. This additive acts as a suppressor factor, kinetically favoring the cleavage of the 21-position ester bond while preserving the 17α-ester linkage. The result is a dramatic shift in the product distribution, heavily favoring the desired 17α-isomer. This strategic intervention allows manufacturers to bypass the limitations of thermodynamic equilibrium that hinder traditional acid-catalyzed reactions, thereby streamlining the production workflow and enhancing overall process efficiency significantly.
Mechanistic Insights into Selective Hydrolysis and Cyclic Ester Formation
The reaction mechanism begins with the formation of a cyclic orthoester intermediate. By dissolving the starting material, such as hydrocortisone, in a cyclic ester solvent like dioxane and reacting it with an orthoester (e.g., triethyl orthoformate) in the presence of a sulfonic acid catalyst, a stable 17,21-cyclic structure is formed. This step is crucial as it temporarily masks the reactivity difference between the two hydroxyl groups. The use of mild catalysts like p-toluenesulfonic acid (PTS) ensures that the sensitive steroid backbone remains intact, preventing degradation or rearrangement side reactions that are common under harsher acidic conditions. The cyclic intermediate serves as a unified precursor that can be processed in bulk before the critical differentiation step occurs.
The true brilliance of the mechanism is revealed during the hydrolysis step. When the cyclic ester is dissolved in a hydrolysis solvent such as methanol and treated with the ammonium chloride solution followed by a hydrolyzing agent like aluminum chloride aqueous solution, a selective cleavage occurs. The ammonium ions likely coordinate with the oxygen atoms or influence the local proton activity in a way that destabilizes the 21-ester bond relative to the 17-ester bond. This directed hydrolysis ensures that the final product is predominantly the 17α-ester. The reaction scheme below illustrates this transformation from the diol starting material (Compound 1) through the cyclic intermediate (Compound 2) to the final 17α-ester product (Compound 3), highlighting the structural changes at the D-ring of the steroid nucleus.
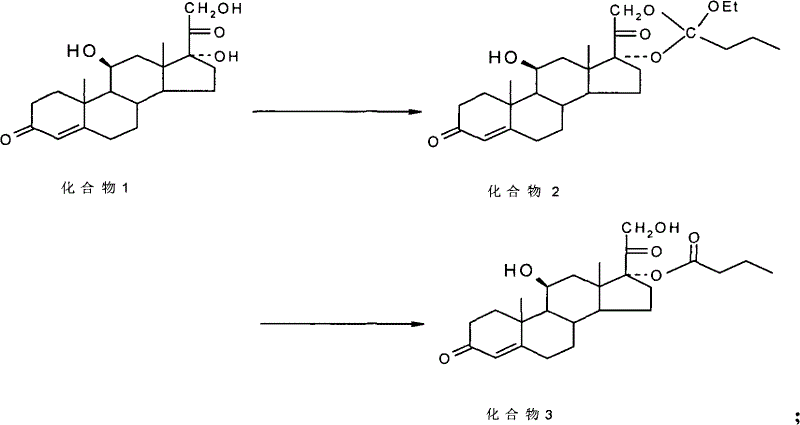
Impurity control is inherently built into this mechanistic pathway. By suppressing the formation of the 21-ester isomer at the source, the burden on purification is drastically reduced. The patent data reports that major impurity levels (specifically the 21-ester) can be controlled to as low as 0.21% to 0.32% after simple activated carbon decolorization and refining. This level of purity is achieved without the need for complex chiral separations or extensive column chromatography, which are often bottlenecks in steroid manufacturing. The mechanism effectively converts a separation problem into a synthesis problem, solving it through chemical selectivity rather than physical isolation.
How to Synthesize Hydrocortisone Butyrate Efficiently
Implementing this synthesis route requires careful attention to solvent quality and reagent stoichiometry to maximize the benefits of the selective hydrolysis. The process is designed to be operationally simple, utilizing standard reactor equipment and commonly available reagents. The initial cyclization is performed at ambient temperatures (around 20°C), which reduces energy consumption and thermal stress on the equipment. Following the reaction, the workup involves a straightforward neutralization and precipitation step, allowing for the isolation of the cyclic ester in high purity before the final hydrolysis. This modular approach allows for quality control checks at the intermediate stage, ensuring that only high-quality material proceeds to the final step.
- Dissolve hydrocortisone in dioxane, add triethyl orthoformate and p-toluenesulfonic acid catalyst to form the cyclic ester intermediate at 20°C.
- Neutralize the reaction mixture with saturated sodium bicarbonate solution and isolate the cyclic ester via filtration and washing.
- Dissolve the cyclic ester in methanol, add saturated ammonium chloride solution and aluminum chloride aqueous solution to effect selective hydrolysis to the 17α-ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates directly into tangible operational improvements and risk mitigation. The primary advantage lies in the substantial increase in overall yield, which reportedly reaches approximately 98% in optimized embodiments. In the context of high-value steroid intermediates, a yield improvement of this magnitude significantly lowers the cost of goods sold (COGS) by maximizing the output from expensive starting materials like hydrocortisone. Furthermore, the reduction in byproduct formation means that less raw material is wasted in purification streams, aligning with green chemistry principles and reducing waste disposal costs associated with hazardous solvent streams.
- Cost Reduction in Manufacturing: The elimination of complex separation processes required to remove 21-ester impurities leads to significant cost savings. Traditional methods often require multiple recrystallizations or chromatographic steps to meet pharmacopeial standards, which are time-consuming and solvent-intensive. By achieving high selectivity chemically, this process reduces the number of unit operations, thereby lowering labor, energy, and solvent recovery costs. The use of recyclable solvents like dioxane further enhances the economic profile by allowing for solvent recovery loops that minimize fresh solvent purchase requirements.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents such as ammonium chloride, aluminum chloride, and common orthoesters ensures a robust supply chain. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents used in this process are commodity chemicals available globally. This availability reduces the risk of supply disruptions and allows for flexible sourcing strategies. Additionally, the mild reaction conditions reduce the wear and tear on manufacturing equipment, leading to higher asset availability and fewer unplanned maintenance downtimes.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from laboratory to pilot plant scales (e.g., 200L reactors) without loss of efficiency. The mild temperature profile (20°C) simplifies thermal management in large-scale reactors, removing the need for extreme heating or cooling infrastructure. From an environmental perspective, the ability to recover and reuse the cyclic ester solvent (dioxane) and the reduction in hazardous waste generation due to higher selectivity make this process more compliant with increasingly stringent environmental regulations. This facilitates smoother regulatory approvals and long-term sustainability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective esterification technology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this method improve regioselectivity compared to traditional acid hydrolysis?
A: Traditional sulfuric acid hydrolysis often yields a 7:3 mixture of 17α-ester to 21-ester. This patented method utilizes ammonium chloride as a directed reagent during hydrolysis, which acts as a suppressor factor, shifting the equilibrium heavily towards the desired 17α-ester and reducing 21-ester impurities to below 0.32%.
Q: What are the critical reaction conditions for the cyclic ester formation?
A: The process requires dissolving the steroid in a cyclic ester solvent like dioxane at mild temperatures (around 20°C). The use of orthoesters (e.g., triethyl orthoformate) and sulfonic acid catalysts allows for gentle cyclization, avoiding the harsh conditions that typically degrade sensitive steroid backbones.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the patent demonstrates scalability in 200L reactors with yields reaching up to 98.1%. The use of recyclable solvents like dioxane and mild hydrolysis conditions makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Butyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of effective topical corticosteroids. Our technical team has extensively analyzed the pathway described in CN101891797B and possesses the expertise to execute this selective hydrolysis protocol with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of hydrocortisone butyrate meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for steroid intermediates. By leveraging our process engineering capabilities, we can help you achieve significant efficiencies and cost reductions in your API manufacturing. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your product development and commercialization goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
