Advanced Hydrocortisone Manufacturing: Novel Rearrangement and Protection Strategies for Commercial Scale
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of critical corticosteroids, with hydrocortisone remaining a cornerstone therapy for inflammatory and autoimmune conditions. Patent CN1070196A discloses a transformative approach to synthesizing hydrocortisone (Formula I), addressing long-standing challenges in steroid functionalization and protecting group chemistry. This technology introduces a novel rearrangement reaction of halohydrin intermediates in the presence of alcohols, followed by highly selective protection strategies that significantly streamline the manufacturing process. By shifting away from harsh oxidative conditions and non-selective protection methods, this route offers a compelling value proposition for manufacturers aiming to optimize their API supply chains. The core innovation lies in the efficient conversion of 11-hydroxy precursors to 11-keto intermediates and the subsequent precise construction of the C20-C21 side chain.
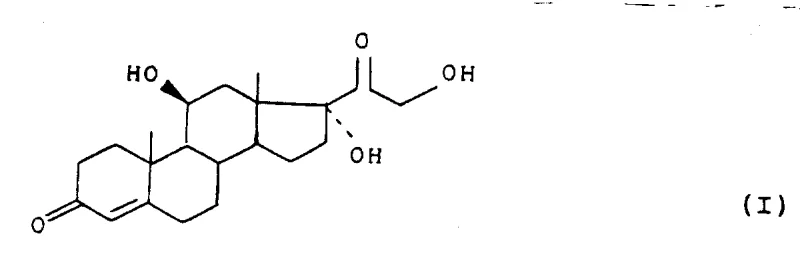
Traditional methods for corticosteroid synthesis have often been plagued by complex protection-deprotection sequences and low-yielding oxidation steps. Conventional routes frequently rely on enol ether or ketone acetal protections for the 3-oxo group, which suffer from poor selectivity, often resulting in difficult-to-separate mixtures of 3-monoprotected and 3,17-diprotected by-products. Furthermore, historical rearrangement reactions required severe conditions that promoted degradation of the sensitive steroid nucleus, leading to substantial material loss and increased waste generation. The necessity for distinct 11-hydroxylation steps in many legacy processes further complicates the supply chain, introducing biological variability or expensive chemical oxidants. These limitations create significant bottlenecks in commercial production, driving up costs and extending lead times for high-purity active pharmaceutical ingredients.
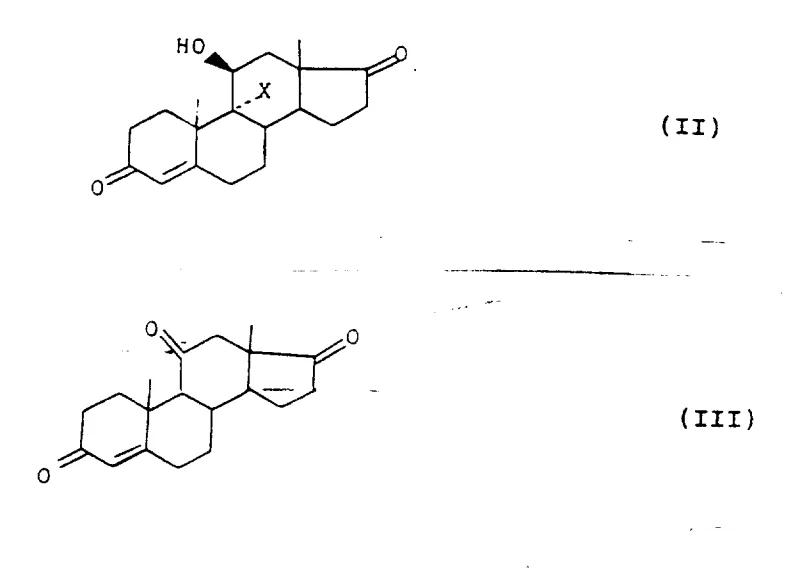
The novel approach detailed in the patent data overcomes these hurdles through a sophisticated sequence of chemical transformations centered on stability and selectivity. The process initiates with a mild rearrangement of a halohydrin precursor (Formula II) to form the 11-keto compound (Formula III), a step that proceeds efficiently under reflux conditions with lower boiling cosolvents like ethyl acetate. Crucially, the method employs thioketal protection using dithioglycol or similar mercaptans, which provides exceptional stability under both acidic and alkaline reaction conditions encountered in subsequent steps. This selectivity ensures that only the 3-position is protected, leaving the 17-position available for functionalization, thereby eliminating the formation of unwanted diprotected impurities. The ability to potentially operate the protection and rearrangement in a 'one-pot' manner further exemplifies the process intensification potential of this technology.
Mechanistic Insights into Zinc-Mediated Side Chain Functionalization
The heart of this synthetic strategy involves the precise construction of the dihydroxyacetone side chain at the C17 position, achieved through a specialized organometallic transformation. The protected 11-keto intermediate (Formula IV) is treated with trihaloacetic acid esters, such as methyl trichloroacetate, in the presence of zinc powder and a Lewis acid catalyst like titanium tetrachloride or zinc chloride. This reaction generates a reactive organozinc species that attacks the C17 carbonyl, effectively extending the carbon skeleton to introduce the C20 and C21 atoms in a single operation. The use of Lewis acids is critical here, as they activate the carbonyl group and facilitate the formation of the carbon-carbon bond under controlled temperatures, typically ranging from -15°C to ambient temperature. This mechanistic pathway avoids the use of highly pyrophoric reagents often seen in alternative Grignard-type approaches, enhancing operational safety on a large scale.
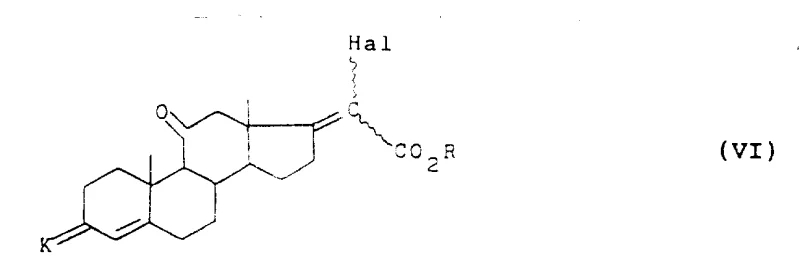
Following the side chain extension, the process incorporates a nucleophilic substitution where a phenol derivative displaces the halogen atom at the C20 position, forming an ether linkage (Formula VII). This step is conducted in an alkaline medium using bases such as sodium amide or alkali metal hydroxides in solvents like butanone or tetrahydrofuran. The resulting phenoxy intermediate serves as a masked precursor to the final hydroxyl group. Subsequent reduction using hydride reagents like diisobutylaluminium hydride (DIBAL-H) converts the ester functionality to a primary alcohol while maintaining the integrity of the steroid backbone. The final stages involve the strategic removal of the thioketal protecting group using iodine in an alkaline or oxidative medium, followed by epoxidation of the 17(20)-double bond and acidic hydrolysis to reveal the native 17-alpha-hydroxyl and 21-hydroxyl groups of hydrocortisone. This sequence demonstrates remarkable chemoselectivity, preserving the sensitive 11-beta-hydroxyl and 3-keto functionalities throughout the synthesis.
How to Synthesize Hydrocortisone Efficiently
The synthesis of hydrocortisone via this novel pathway requires careful control of reaction parameters to maximize yield and purity. The process begins with the preparation of the halohydrin starting material, which undergoes rearrangement in the presence of glycols or alcohols to establish the 11-keto motif. Following this, the 3-oxo group is selectively masked as a thioketal, a step that is pivotal for preventing side reactions at the A-ring during subsequent basic treatments. The standardized protocol involves reacting the protected intermediate with methyl trichloroacetate and zinc dust under inert atmosphere, followed by quenching and extraction. Detailed standard operating procedures for each unit operation, including specific solvent ratios, temperature ramps, and workup techniques, are essential for reproducibility. For the complete step-by-step synthesis guide including exact molar equivalents and purification methods, please refer to the technical documentation below.
- Perform rearrangement of 9-alpha-halo-11-beta-hydroxy steroids in the presence of alcohol and acid to form the 11-keto intermediate.
- Protect the 3-oxo group selectively using dithioglycol or similar mercaptans under Lewis acid catalysis to form the thioketal derivative.
- React the protected intermediate with trihaloacetic acid esters using Zinc and Lewis acid, followed by phenol substitution, reduction, and epoxidation to yield hydrocortisone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits rooted in process efficiency and raw material optimization. The shift towards milder reaction conditions directly translates to reduced energy consumption and lower demands on reactor metallurgy, as the process does not require extreme temperatures or pressures. The high selectivity of the thioketal protection minimizes the generation of hard-to-remove impurities, which significantly reduces the burden on downstream purification units such as chromatography columns or crystallization tanks. This improvement in crude quality leads to higher overall recovery rates of the final API, effectively lowering the cost of goods sold (COGS) by maximizing the output from each batch of starting material. Furthermore, the elimination of the 11-hydroxylation step simplifies the manufacturing workflow, removing a potential bottleneck that often relies on specialized fermentation capacity or hazardous oxidants.
- Cost Reduction in Manufacturing: The novel rearrangement and protection strategy eliminates the need for expensive and hazardous reagents associated with traditional 11-hydroxylation and non-selective protection methods. By utilizing readily available reagents like zinc powder, titanium tetrachloride, and common thiols, the direct material costs are optimized. The 'one-pot' potential for the initial rearrangement and protection steps further reduces solvent usage and processing time, leading to substantial operational expenditure savings. Additionally, the robustness of the intermediates allows for longer campaign runs with less equipment downtime for cleaning and maintenance.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available chemical reagents rather than biologically derived enzymes or scarce oxidants enhances the resilience of the supply chain. The intermediates generated in this pathway, such as the thioketal-protected 11-keto steroids, are chemically stable and can be stored or transported if necessary, providing flexibility in production scheduling. This stability mitigates the risk of batch failures due to reagent degradation, ensuring a consistent flow of high-quality intermediates to the final synthesis stages. Consequently, manufacturers can maintain tighter delivery schedules and respond more agilely to market demand fluctuations for hydrocortisone.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents and conditions that are manageable in large-scale stainless steel reactors. The avoidance of heavy metal catalysts in the final steps and the use of aqueous workups align with modern environmental, health, and safety (EHS) standards. Waste streams are easier to treat due to the absence of complex biological by-products or highly toxic oxidation residues. This environmental compatibility facilitates regulatory approval and reduces the costs associated with waste disposal and environmental compliance monitoring, making the process sustainable for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this hydrocortisone synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, focusing on the practical aspects of reaction conditions and intermediate stability. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer. The responses highlight the specific advantages of the thioketal protection and the zinc-mediated coupling reaction which differentiate this method from prior art.
Q: What are the advantages of the thioketal protection method described in CN1070196A?
A: The thioketal protection offers significant selectivity compared to traditional ketone acetal or enol ether protections. It prevents the formation of mixed 3,17-protected products, ensuring higher purity of the 3-oxo protected intermediate and simplifying downstream purification processes.
Q: How does the novel rearrangement reaction improve industrial feasibility?
A: The rearrangement of the halohydrin intermediate occurs under much milder conditions than previous methods (such as those in European patent 30368). This reduces thermal stress on the steroid backbone, minimizing degradation by-products and allowing for safer, more economical scale-up in large reactors.
Q: Can this synthesis route avoid the 11-hydroxylation step?
A: Yes, a key feature of this methodology is that it utilizes starting materials or intermediates that already possess the necessary oxygenation pattern or generates it via rearrangement, thereby eliminating the need for a separate, often low-yielding microbial or chemical 11-hydroxylation step found in older synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing pathways for life-saving corticosteroids like hydrocortisone. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and assay. Our facility is equipped to handle the specific reagents and conditions required for this novel synthesis, including the safe handling of Lewis acids and organozinc intermediates.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can enhance your product availability and cost efficiency. Let us be your strategic partner in delivering high-quality hydrocortisone intermediates and APIs to the global market.
