Advanced Palladium-Catalyzed Decarboxylative Thioesterification for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking safer, more efficient pathways to construct critical molecular scaffolds. Patent CN110483351B introduces a transformative approach to synthesizing thioesters, a vital class of intermediates used extensively in drug discovery and agrochemical development. Unlike conventional methods that rely on hazardous gaseous reagents, this invention utilizes a palladium-catalyzed decarboxylative coupling strategy. By employing oxothioglycolic acid compounds as stable solid precursors, the process achieves high-efficiency catalytic conversion under mild conditions. This technological breakthrough not only simplifies the operational workflow by removing the need for external bases in many instances but also aligns perfectly with the principles of green chemistry. For R&D teams and procurement specialists, this represents a significant opportunity to optimize synthesis strategies for complex drug molecules while mitigating safety risks associated with traditional carbonylation techniques.
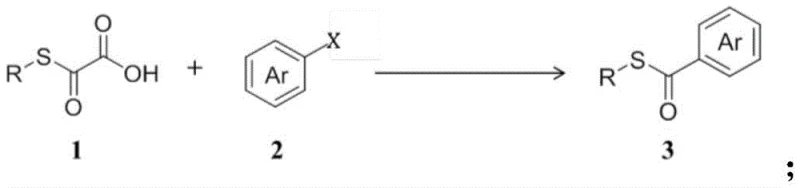
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of thioester bonds has predominantly relied on carbonyl intercalation systems involving thiols and carbon monoxide. While chemically effective, these traditional protocols present severe industrial challenges. The requirement for carbon monoxide, a highly toxic and flammable gas, necessitates specialized high-pressure reactor infrastructure and rigorous safety monitoring systems. Furthermore, these reactions often demand elevated temperatures to overcome activation barriers, which can lead to thermal degradation of sensitive functional groups commonly found in advanced pharmaceutical intermediates. The logistical burden of storing and handling pressurized CO cylinders, combined with the potential for catalyst poisoning by sulfur species under harsh conditions, creates a bottleneck in scaling up production. These factors collectively increase both the capital expenditure for facility setup and the operational costs related to safety compliance and waste management.
The Novel Approach
The methodology described in CN110483351B fundamentally shifts the paradigm by replacing gaseous CO with solid oxothioglycolic acid derivatives. This innovation allows the reaction to proceed at ambient pressure, drastically reducing the engineering controls required for safe operation. The process operates effectively at room temperature for aryl iodides and unsaturated compounds, and at moderate heating (around 100°C) for aryl bromides, preserving the integrity of thermally labile moieties. Moreover, the system exhibits remarkable atom economy and functional group tolerance, successfully accommodating substrates with esters, ketones, aldehydes, and heterocycles without the need for extensive protecting group strategies. By simplifying the post-reaction workup and eliminating the need for additional alkali additives in many cases, this novel approach streamlines the purification process, offering a cleaner and more sustainable route for the commercial scale-up of complex thioesters.
Mechanistic Insights into Palladium-Catalyzed Decarboxylative Coupling
The core of this synthetic advancement lies in the intricate palladium catalytic cycle that facilitates the decarboxylation and subsequent bond formation. The reaction initiates with the oxidative addition of the aryl halide or the activation of the unsaturated bond to the zero-valent palladium center. In the presence of the bidentate phosphine ligand Xantphos, the palladium complex stabilizes the intermediate species, preventing premature catalyst deactivation which is a common issue in sulfur-containing reactions. The oxothioglycolic acid compound then coordinates to the metal center, undergoing a crucial decarboxylation step that releases carbon dioxide and generates a reactive acyl-palladium species. This transient intermediate is key to the process, as it mimics the reactivity of a carbonyl-palladium complex formed from CO gas but without the associated hazards. The final thioester product is released via reductive elimination, regenerating the active Pd(0) catalyst to continue the cycle. This mechanistic pathway ensures high chemical selectivity and minimizes the formation of homocoupling byproducts.
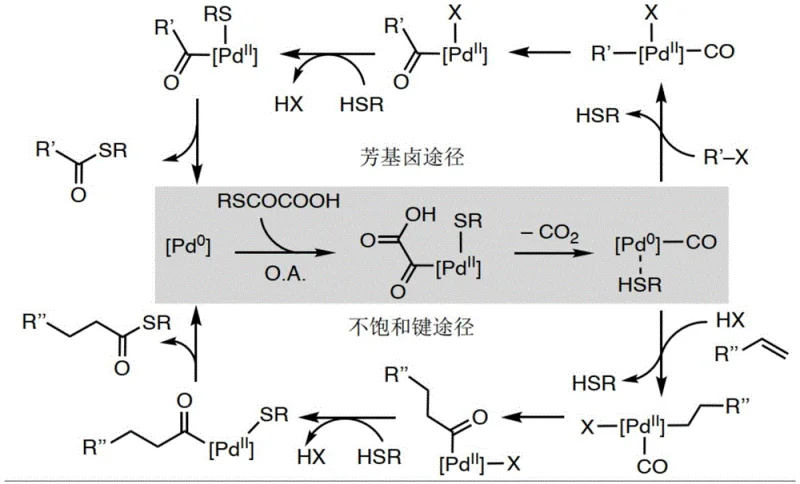
Impurity control is inherently managed through the mild reaction conditions and the specific choice of ligands. The use of Xantphos, with its large bite angle, promotes the reductive elimination step, which is often the rate-determining step in such cross-couplings. This kinetic acceleration reduces the residence time of reactive intermediates, thereby limiting opportunities for side reactions such as beta-hydride elimination or protodehalogenation. Additionally, the protocol's compatibility with a wide range of solvents, including polar aprotic solvents like DMA for aryl halides and non-polar solvents like toluene for unsaturated bonds, allows for fine-tuning the solubility of reactants and products. This solubility management is critical for maintaining a homogeneous reaction phase, which further enhances reproducibility and batch-to-b consistency. For quality control teams, this means a more predictable impurity profile, simplifying the validation process for regulatory submissions.
How to Synthesize Thioesters Efficiently
Implementing this synthesis route requires precise adherence to the optimized conditions outlined in the patent to ensure maximum yield and purity. The general procedure involves charging a reaction vessel with the oxothioglycolic acid substrate, the electrophilic partner (aryl halide or alkene), the palladium catalyst, and the phosphine ligand under an inert argon atmosphere. The choice of solvent and temperature is dictated by the specific reactivity of the electrophile; aryl iodides and alkenes react smoothly at 25°C, whereas aryl bromides benefit from heating to 100°C. For reactions involving unsaturated bonds, the addition of sodium iodide acts as a crucial promoter to facilitate the formation of the active palladium species. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for successful technology transfer.
- Prepare the reaction mixture by combining oxothioglycolic acid compound (1.5 equiv), aryl halide or unsaturated compound (1.0 equiv), Pd(PPh3)4 catalyst (5 mol%), and Xantphos ligand (6 mol%) in an inert atmosphere.
- Add the appropriate solvent: N,N-dimethylacetamide for aryl iodides/bromides or toluene with sodium iodide additive for unsaturated bond-containing compounds.
- Stir the mixture at 25°C for aryl iodides and unsaturated compounds, or heat to 100°C for aryl bromides, for 8 hours under argon protection before purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The transition from gas-phase carbonylation to solid-phase decarboxylation removes the dependency on specialized high-pressure infrastructure, allowing production to be conducted in standard glass-lined or stainless steel reactors available in most multipurpose chemical plants. This flexibility significantly lowers the barrier to entry for manufacturing partners and reduces the lead time associated with booking specialized high-pressure suites. Furthermore, the stability of the oxothioglycolic acid starting materials ensures a robust supply chain, as these solids are easier to store, transport, and handle compared to compressed gas cylinders, mitigating risks related to logistics disruptions.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes the need for expensive gas detection systems, scrubbing units, and high-pressure containment vessels, leading to substantial capital expenditure savings. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, while the high atom economy minimizes raw material waste. The simplified workup procedure, often requiring only basic extraction and chromatography, reduces solvent usage and labor hours, collectively driving down the cost of goods sold for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing stable solid reagents instead of hazardous gases enhances the reliability of the supply chain by reducing regulatory hurdles related to the transport of dangerous goods. The broad substrate scope means that a single standardized protocol can be applied to synthesize a diverse library of thioester derivatives, allowing for agile response to changing market demands. This versatility ensures that production schedules are less likely to be impacted by the need for process re-validation when switching between different molecular targets.
- Scalability and Environmental Compliance: The process aligns with increasingly stringent environmental regulations by avoiding the release of toxic CO and minimizing the generation of heavy metal waste through efficient catalyst loading. The ability to run reactions at ambient pressure simplifies the scale-up process from laboratory to pilot plant to commercial production, as heat and mass transfer issues are less pronounced compared to high-pressure gas-liquid reactions. This ease of scale-up ensures a continuous supply of materials without the prolonged delays often encountered when optimizing complex high-pressure processes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this palladium-catalyzed decarboxylative thioesterification method. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on reaction parameters and substrate compatibility.
Q: What are the safety advantages of this thioester synthesis method compared to traditional carbonylation?
A: Traditional thioester synthesis often requires toxic carbon monoxide gas under high pressure and temperature. This patented method utilizes stable oxothioglycolic acid solids as the carbonyl source, eliminating the need for hazardous CO gas handling and high-pressure equipment, thereby significantly enhancing operational safety.
Q: Can this palladium-catalyzed method tolerate complex functional groups found in drug molecules?
A: Yes, the method demonstrates excellent functional group compatibility. It successfully synthesizes thioesters from substrates containing esters, ketones, aldehydes, halogens, and even complex biomolecular structures like steroids and protected amino acids, making it highly suitable for late-stage functionalization of pharmaceutical intermediates.
Q: What catalyst system is optimal for reacting with aryl bromides versus aryl iodides?
A: For aryl iodides, the reaction proceeds efficiently at room temperature (25°C) using Pd(PPh3)4 and Xantphos. For the less reactive aryl bromides, the same catalyst system is effective but requires elevated temperatures (optimally 100°C) to achieve high conversion rates, ensuring versatility across different halide substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Supplier
The innovative synthesis route detailed in CN110483351B underscores the evolving landscape of fine chemical manufacturing, where safety and efficiency are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such advanced methodologies to market. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that ensure every batch meets the highest international standards. We understand the critical nature of supply continuity for API intermediates and are committed to delivering consistent quality.
We invite procurement leaders and R&D directors to collaborate with us to evaluate the feasibility of this green synthesis route for your specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data for our existing thioester portfolio and discuss route feasibility assessments for your custom synthesis needs, ensuring a secure and cost-effective supply chain for your future developments.
