Advanced Cefuroxime Acid Synthesis: Technical Upgrade and Commercial Scalability
Advanced Cefuroxime Acid Synthesis: Technical Upgrade and Commercial Scalability
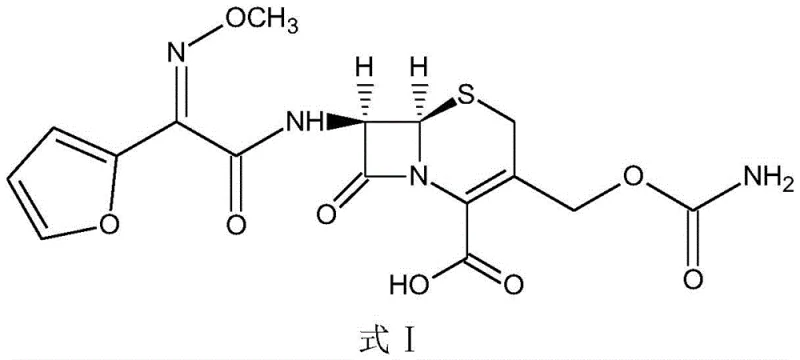
The pharmaceutical industry continuously seeks robust manufacturing processes for critical beta-lactam antibiotics, and recent intellectual property developments highlight significant strides in this domain. Specifically, patent CN112679525B discloses a refined preparation method for cefuroxime acid that addresses long-standing stability and crystallization challenges inherent in traditional synthesis routes. This technical breakthrough utilizes deacetyl 7-aminocephalosporanic acid (D-7ACA) as a primary starting material, streamlining the production pathway while enhancing the physical properties of the final active pharmaceutical ingredient. By integrating early-stage decolorization and a novel multi-step crystallization protocol, the method ensures superior product fluidity and stability, which are paramount for downstream formulation. For global supply chain stakeholders, understanding these mechanistic improvements is essential for evaluating the reliability of potential cefuroxime acid suppliers and the feasibility of large-scale commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of cefuroxime acid has been plagued by significant technical bottlenecks that compromise both yield and product quality. Traditional methods often rely on 7-aminocephalosporanic acid (7-ACA) or D-7ACA, but the latter has frequently been associated with the formation of extremely fine crystals that are unsuitable for efficient centrifugal operation. In many prior art processes, such as those described in CN106432267A, the reliance on liquid ester solvents creates difficulties in recovery and amplification, leading to excessive energy consumption and prolonged working hours. Furthermore, conventional crystallization techniques often involve dissolving the product in an alkali solution for decolorization late in the process, which exposes the sensitive beta-lactam ring to degradation risks, resulting in higher color grades and reduced stability over time. These inefficiencies create substantial supply chain vulnerabilities, making cost reduction in API manufacturing difficult to achieve without sacrificing purity standards.
The Novel Approach
The innovative methodology outlined in the patent data introduces a paradigm shift by reordering the purification and crystallization sequences to maximize stability and operational efficiency. A key differentiator is the strategic relocation of the decolorization step to the preparation of the intermediate MDCC, thereby eliminating the need to dissolve the final cefuroxime acid in alkali for purification. This modification significantly mitigates the risk of product degradation and color growth, ensuring a higher quality output that meets stringent pharmacopoeia standards. Additionally, the process employs a controlled, multi-stage addition of alkali solution during the final crystallization phase, which promotes the growth of larger, cuboid crystals measuring approximately 4μm by 1μm. This improvement in crystal morphology not only enhances product fluidity and ease of splitting but also facilitates faster filtration and drying, directly contributing to reduced lead time for high-purity cephalosporins in a commercial setting.
Mechanistic Insights into D-7ACA Acylation and Crystallization Control
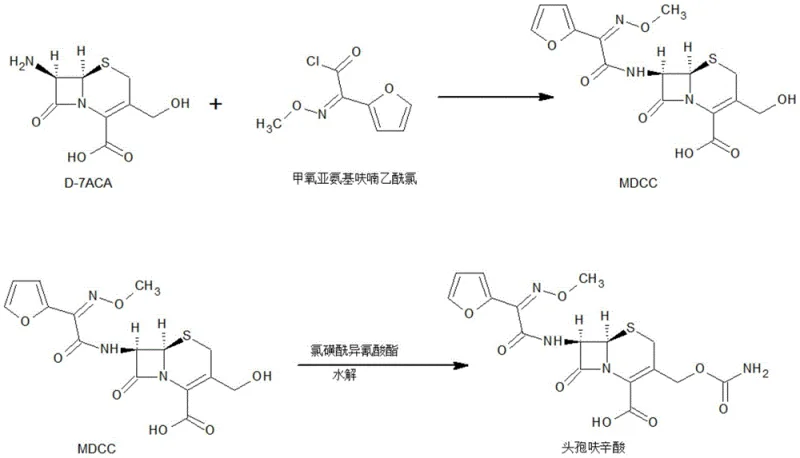
The core of this synthesis lies in the precise chemical transformation of D-7ACA into the MDCC intermediate, followed by a highly controlled aminomethyl acylation. The reaction begins with the acylation of D-7ACA using methoxyimino furan acetyl chloride in a dichloromethane system, where pH is meticulously maintained between 6.0 and 8.0 to optimize nucleophilic attack while preserving the integrity of the beta-lactam ring. Following this, the intermediate undergoes a critical reaction with chlorosulfonyl isocyanate at cryogenic temperatures ranging from -40°C to -25°C. This low-temperature environment is crucial for suppressing side reactions and ensuring the selective formation of the carbamoyl group at the 3-position of the cephem nucleus. The subsequent hydrolysis step converts the chlorosulfonyl intermediate into the target cefuroxime acid suspension, setting the stage for the unique crystallization protocol that defines the patent's novelty.
Impurity control is rigorously managed through the physical chemistry of crystallization rather than relying solely on chemical scavengers. By avoiding the dissolution of the final product in alkaline conditions, the process prevents the hydrolysis of the beta-lactam ring which typically generates open-ring impurities and colored byproducts. The multi-dropwise addition of alkali solution during crystallization allows for a gradual adjustment of the system pH to between 1.0 and 2.0, fostering a supersaturation environment that favors the growth of well-defined crystals over rapid nucleation. This controlled growth mechanism effectively traps fewer impurities within the crystal lattice, resulting in a product with superior color stability, as evidenced by data showing color grades remaining below Y6 even after 12 months of storage. Such robust impurity management is critical for a reliable pharmaceutical intermediates supplier aiming to meet the exacting specifications of global regulatory bodies.
How to Synthesize Cefuroxime Acid Efficiently
Implementing this synthesis route requires strict adherence to the thermal and pH parameters defined in the patent examples to ensure reproducibility and safety. The process is divided into two main stages: the preparation of the MDCC intermediate and the subsequent conversion to cefuroxime acid, each demanding precise operational control. Operators must manage the exothermic nature of the acylation reactions and maintain cryogenic conditions during the isocyanate step to prevent thermal runaway. While the general workflow is straightforward, the specific timing of alkali addition and temperature ramps during crystallization are critical process parameters that determine the final crystal habit. For detailed technical execution, the standardized synthesis steps see the guide below which outlines the specific reagent ratios and timing sequences required for optimal yield.
- Prepare MDCC intermediate by acylating D-7ACA with methoxyimino furan acetyl chloride, including an early decolorization step.
- React MDCC with chlorosulfonyl isocyanate at low temperatures (-40 to -25°C) to form the aminomethyl acylated intermediate.
- Hydrolyze the intermediate and perform multi-stage crystallization by dropwise addition of alkali solution to control pH and crystal growth.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing process offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The elimination of complex solvent recovery steps associated with liquid esters and the reduction of unit operations streamline the production workflow, leading to substantial cost savings in terms of energy and labor. By improving the crystal size and fluidity, the process enhances the efficiency of downstream handling, including filtration, drying, and packaging, which reduces the overall cycle time per batch. These operational efficiencies translate into a more competitive cost structure without compromising the high-purity standards required for antibiotic production, making it an attractive option for cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The process achieves significant economic benefits by removing the need for late-stage decolorization of the final product, which traditionally requires additional reagents and processing time. By shifting purification to the intermediate stage, the overall consumption of activated carbon and solvents is optimized, and the risk of yield loss during final handling is minimized. Furthermore, the high weight yield reported in the patent examples indicates a highly efficient conversion of raw materials, reducing the cost of goods sold per kilogram of active ingredient. This efficiency allows for a more resilient pricing model that can withstand fluctuations in raw material costs while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The robustness of the crystallization process ensures consistent batch-to-batch quality, which is vital for maintaining uninterrupted supply to downstream formulation partners. The improved physical properties of the crystals, such as better flowability and larger particle size, reduce the likelihood of processing blockages or delays during manufacturing, thereby enhancing overall equipment effectiveness. This reliability minimizes the risk of production stoppages and ensures that delivery schedules can be met consistently, a key factor for partners seeking a reliable pharmaceutical intermediates supplier for long-term contracts.
- Scalability and Environmental Compliance: The simplified workflow and reduced solvent usage contribute to a smaller environmental footprint, aligning with increasingly strict global regulations on industrial emissions and waste disposal. The process avoids the use of difficult-to-recover liquid esters, favoring solvents that are easier to manage and recycle on a commercial scale. This environmental compliance not only mitigates regulatory risk but also supports the sustainability goals of modern pharmaceutical companies, facilitating the commercial scale-up of complex beta-lactams with greater ease and lower waste treatment costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical principles underlying the process. Understanding these details helps stakeholders assess the feasibility of integrating this technology into their existing supply chains and quality systems.
Q: How does the new process improve cefuroxime acid stability?
A: The process moves the decolorization step to the MDCC intermediate stage, avoiding product degradation during the final alkali dissolution. Additionally, multi-stage crystallization produces larger, more stable crystals.
Q: What are the critical temperature controls in this synthesis?
A: The aminomethyl acylation reaction requires strict temperature control between -40°C and -25°C to ensure reaction efficiency and prevent side reactions, followed by hydrolysis at 10-20°C.
Q: Why is D-7ACA preferred over 7-ACA for this route?
A: Using D-7ACA shortens the process route and reduces raw material costs compared to 7-ACA, while the specific crystallization method overcomes the historical issue of fine, hard-to-filter crystals associated with D-7ACA routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefuroxime Acid Supplier
The technical potential of this D-7ACA based route represents a significant opportunity for optimizing the production of second-generation cephalosporins. NINGBO INNO PHARMCHEM, as a specialized CDMO partner, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative processes to market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of cefuroxime acid meets the highest international standards for safety and efficacy. We are committed to leveraging our technical expertise to support the commercialization of advanced synthesis routes that deliver both quality and value.
We invite procurement leaders to engage with our technical procurement team to discuss how this optimized process can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of this method tailored to your volume needs. We encourage you to contact us to obtain specific COA data and route feasibility assessments, ensuring that your partnership is built on a foundation of transparency, technical excellence, and mutual growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
