Revolutionizing Isoxazole Production: A Green Aqueous Micellar Catalysis Strategy for Commercial Scale
Introduction to Green Isoxazole Synthesis Technology
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by both regulatory pressure and the economic necessity of reducing waste. Patent CN113582937A introduces a groundbreaking methodology for the synthesis of isoxazole compounds, a privileged scaffold found in numerous bioactive molecules ranging from antibiotics to anti-inflammatory agents. This technology leverages the power of micellar catalysis using TPGS-750-M, a water-soluble vitamin E derivative, to facilitate the cycloaddition of aldoximes and alkynes in an aqueous medium. By replacing traditional volatile organic compounds (VOCs) and expensive transition metal catalysts with a benign aqueous system, this invention addresses critical pain points in modern chemical synthesis, offering a pathway to high-purity intermediates with a drastically reduced environmental footprint.
The core innovation lies in the ability to perform these transformations under exceptionally mild conditions, specifically at room temperature, which contrasts sharply with the energy-intensive protocols of the past. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes for complex heterocyclic targets. The method utilizes N-chlorosuccinimide (NCS) as an oxidant in conjunction with a mild base, creating a reactive environment within the hydrophobic core of the surfactant micelles. This not only enhances reaction rates but also improves selectivity, minimizing the formation of difficult-to-remove impurities that often plague traditional metal-catalyzed approaches.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoxazole ring has relied heavily on methodologies that pose significant challenges for industrial scale-up and environmental compliance. Traditional routes often involve the use of homogeneous metal catalysts such as palladium, copper, or gold, which are not only prohibitively expensive but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing trace metals to meet stringent regulatory limits requires additional purification steps, such as scavenging or recrystallization, which inevitably lower overall yield and increase processing time. Furthermore, many classical syntheses require harsh reaction conditions, including high temperatures and the use of toxic organic solvents like dichloromethane or DMF, generating substantial hazardous waste streams that are costly to treat and dispose of.
Another critical drawback of conventional methods is their limited functional group tolerance. Strong acids or bases often required in older protocols can lead to the degradation of sensitive moieties present in complex drug candidates, necessitating protective group strategies that add unnecessary steps to the synthesis. The reliance on stoichiometric amounts of hazardous reagents further exacerbates safety concerns in a manufacturing setting. Consequently, there is a pressing demand for a robust, metal-free alternative that can deliver high purity isoxazole derivatives without compromising on safety or sustainability, a gap that previous technologies have failed to adequately fill.
The Novel Approach
The methodology described in CN113582937A fundamentally reimagines the synthesis landscape by utilizing water as the primary reaction solvent, mediated by the designer surfactant TPGS-750-M. This approach creates a "nanoreactor" environment where organic reactants are concentrated within the micellar core, effectively mimicking the efficiency of organic solvents while maintaining the safety profile of water. The reaction proceeds smoothly at room temperature, eliminating the need for energy-intensive heating or cooling systems. As illustrated in the general reaction scheme below, the coupling of aldoximes and alkynes is achieved with high regioselectivity and excellent yields, demonstrating the versatility of this system across a broad range of substrates.
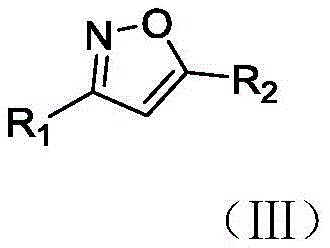
By employing N-chlorosuccinimide (NCS) and a simple amine base like triethylamine, the process avoids the use of precious metals entirely. This metal-free characteristic is a game-changer for supply chain managers, as it removes the volatility associated with the pricing and availability of catalysts like palladium. Moreover, the aqueous workup is straightforward; the product can be extracted with standard organic solvents like ethyl acetate, leaving the surfactant in the water phase for potential recovery and reuse. This simplicity translates directly into operational efficiency, reducing the complexity of downstream processing and allowing for a more streamlined production workflow that aligns perfectly with green chemistry principles.
Mechanistic Insights into Micellar Catalysis and Oxidative Cyclization
To fully appreciate the technical superiority of this method, one must understand the mechanistic role of the TPGS-750-M surfactant. TPGS-750-M is a amphiphilic molecule derived from tocopherol (Vitamin E), featuring a hydrophobic tail and a hydrophilic polyethylene glycol head. In water, these molecules self-assemble into nanomicelles above a critical concentration. These micelles possess a lipophilic interior capable of solubilizing the organic aldoxime and alkyne substrates, effectively bringing them into close proximity despite the aqueous bulk medium. This local concentration effect significantly accelerates the reaction kinetics, allowing the transformation to occur rapidly at ambient temperatures where it might otherwise be sluggish or impossible in pure water.
The oxidative cyclization is initiated by N-chlorosuccinimide (NCS), which activates the aldoxime to form a nitrile oxide intermediate or a similar electrophilic species in situ. Within the confined space of the micelle, this reactive intermediate undergoes a [3+2] cycloaddition with the alkyne. The basic conditions provided by triethylamine facilitate the deprotonation steps necessary for the cycle to complete. Crucially, the micellar environment stabilizes these transient species, preventing side reactions that typically occur in bulk solution. The preparation of the surfactant itself, as shown in the synthesis scheme below, involves the esterification of tocopherol succinate with methoxy polyethylene glycol, ensuring that the catalyst is both biodegradable and non-toxic, further enhancing the green credentials of the overall process.
From an impurity control perspective, this mechanism offers distinct advantages. The mildness of the room temperature reaction prevents thermal degradation of the product or starting materials. Additionally, the absence of metal catalysts eliminates the formation of metal-complexed byproducts, resulting in a cleaner crude reaction mixture. This high level of purity simplifies the purification process, often allowing for direct crystallization or simple chromatography, which is vital for maintaining high throughput in a commercial setting. The robustness of the micellar system also means that it tolerates a wide variety of functional groups on both the aldoxime and alkyne components, making it a versatile platform for synthesizing diverse libraries of isoxazole derivatives for drug discovery programs.
How to Synthesize 3,5-Disubstituted Isoxazoles Efficiently
Implementing this green synthesis route in a laboratory or pilot plant setting is straightforward and requires minimal specialized equipment. The process begins with the preparation of the aqueous surfactant solution, followed by the sequential addition of reagents under stirring. The reaction monitoring can be easily performed using standard TLC or HPLC methods. The key to success lies in maintaining the correct ratio of surfactant to water to ensure optimal micelle formation, as well as controlling the addition rate of the base to manage the exotherm, although the reaction is generally mild. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide provided below.
- Prepare a 2 wt.% aqueous solution of the surfactant TPGS-750-M.
- Mix the aldoxime substrate and N-chlorosuccinimide (NCS) in the surfactant solution and stir at room temperature.
- Add the base (triethylamine) and the alkyne compound, continuing reaction at room temperature for 6-16 hours.
- Extract the product with ethyl acetate, purify via column chromatography, and recover the surfactant from the aqueous phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TPGS-750-M mediated synthesis offers compelling economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage is the drastic reduction in raw material costs associated with the elimination of precious metal catalysts. Palladium and gold catalysts are subject to significant market price fluctuations and supply chain bottlenecks; removing them from the process stabilizes the cost of goods sold (COGS) and insulates the manufacturing budget from external market volatility. Furthermore, the use of water as a solvent replaces expensive and regulated organic solvents, leading to substantial savings in solvent procurement and waste disposal fees.
- Cost Reduction in Manufacturing: The economic impact of switching to this aqueous micellar system is profound. By eliminating the need for expensive transition metals and reducing solvent consumption, the direct material costs are significantly lowered. The ability to run reactions at room temperature also reduces energy consumption for heating and cooling, contributing to lower utility bills. Additionally, the simplified workup procedure reduces labor hours and equipment occupancy time, increasing the overall throughput of the manufacturing facility without the need for capital investment in new hardware.
- Enhanced Supply Chain Reliability: Reliance on a stable and abundant supply of raw materials is critical for uninterrupted production. The reagents used in this method, such as NCS, triethylamine, and the surfactant precursors, are commodity chemicals with robust global supply chains. This contrasts sharply with specialized ligands or rare earth metals that may have single-source suppliers. The resilience of the supply chain is further bolstered by the potential to recycle the TPGS-750-M surfactant from the aqueous waste stream, creating a closed-loop system that minimizes dependency on fresh material inputs.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new safety and environmental challenges, but this technology mitigates those risks effectively. The use of water eliminates fire hazards associated with flammable organic solvents, simplifying safety protocols and insurance costs. From a regulatory standpoint, the process generates significantly less hazardous waste, making it easier to comply with increasingly strict environmental regulations such as REACH or EPA guidelines. This "green" designation can also enhance the marketability of the final API to environmentally conscious partners and consumers.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is essential for stakeholders evaluating its integration into existing workflows. The following questions address common inquiries regarding the practical application, scalability, and compatibility of the TPGS-750-M system. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for decision-making.
Q: What are the primary advantages of using TPGS-750-M in isoxazole synthesis?
A: The use of TPGS-750-M allows the reaction to proceed in water rather than toxic organic solvents. It forms nanomicelles that solubilize organic reactants, enabling high efficiency at room temperature without expensive transition metal catalysts.
Q: Can the surfactant be recycled in this process?
A: Yes, the patent demonstrates that the TPGS-750-M surfactant remains in the aqueous phase after extraction and can be recovered and reused, significantly reducing waste and material costs.
Q: Is this method suitable for large-scale pharmaceutical manufacturing?
A: Absolutely. The mild reaction conditions (room temperature), absence of heavy metals, and use of water as a solvent make this process highly scalable, safe, and compliant with strict environmental regulations for API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazole Derivatives Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, partnering with a manufacturer that possesses advanced, sustainable synthesis capabilities is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging innovations like the TPGS-750-M mediated isoxazole synthesis to deliver superior products. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our rigorous QC labs and stringent purity specifications guarantee that every batch of isoxazole derivatives meets the highest industry standards, ready for immediate use in your drug development pipelines.
We invite you to explore how our green chemistry solutions can optimize your supply chain and reduce your overall manufacturing costs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of modern chemical manufacturing with a partner dedicated to innovation, sustainability, and reliability.
